Clear Sky Science · en
Highly selective and practical hydrogenation of functionalized (hetero)arenes
From Flat Molecules to 3D Building Blocks
Modern medicines, plastics, and many everyday chemicals are built from small molecular “Lego bricks.” Most of these bricks are flat, ring-shaped structures called arenes, which chemists love because they are easy to make and modify. But drug hunters and materials scientists increasingly want more three‑dimensional shapes, which often behave better in the body and give plastics improved performance. This article describes a new, practical way to turn flat rings into precise 3D structures using a robust platinum catalyst, opening a simpler route to next‑generation drugs and safer plastic additives.
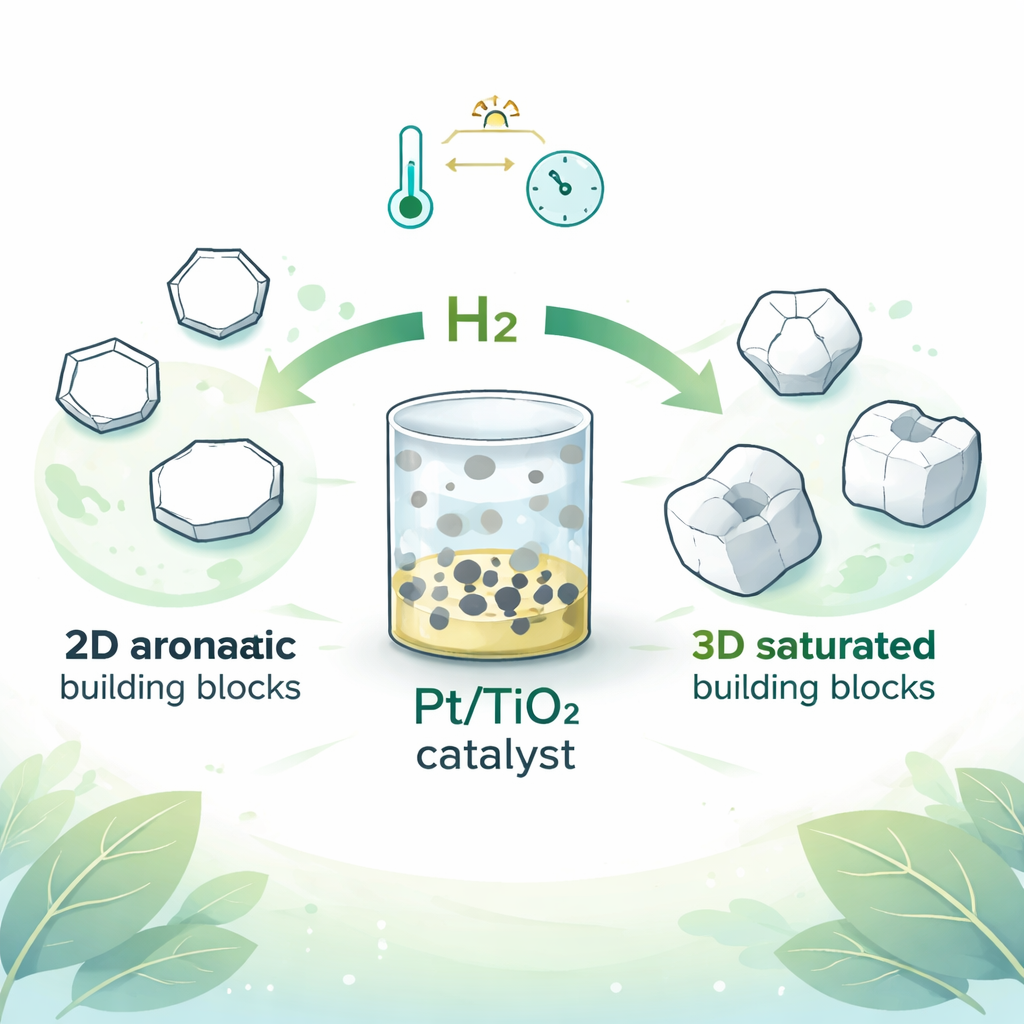
Why Shape Matters in Molecules
Flat aromatic rings are everywhere: in pharmaceuticals, agrochemicals, vitamins, and polymers. Their popularity means they are cheap and widely available. By contrast, their “saturated” cousins—rings whose double bonds have been removed—are far less common commercially, even though they bring important advantages. When a ring becomes saturated and three‑dimensional, chemists gain much finer control over properties such as how a drug fits into a protein pocket or how a plastic bends and softens. For instance, by adjusting the ratio between two mirror‑like 3D arrangements (called cis and trans forms), polymer makers can tune the glass transition temperature, which determines whether a material is stiff or flexible at a given temperature.
The Challenge of Bending Flat Rings
Converting a flat aromatic ring into a 3D saturated ring sounds simple—just add hydrogen. In practice it is very difficult. Aromatic rings are unusually stable, so breaking their “aromaticity” costs a lot of energy. At the same time, real‑world molecules often carry extra chemical groups, like esters or amides, that must survive the process untouched. A catalyst must therefore do three things at once: activate stubborn rings under mild conditions, ignore other sensitive parts of the molecule, and arrange new hydrogen atoms so that one 3D form is strongly preferred over the other. Existing catalysts that manage this are usually complex, sensitive, and hard to recycle, which makes them unattractive for large‑scale industrial use.
A Robust Platinum Catalyst on a Familiar Support
The researchers report a simple heterogeneous catalyst: tiny particles of platinum deposited on titanium dioxide (Pt/TiO2). Using this material, they can hydrogenate a wide variety of multi‑substituted arenes and heteroarenes—rings that also contain atoms like nitrogen or oxygen—under relatively gentle conditions of temperature and hydrogen pressure. Remarkably, the reactions strongly favor one 3D arrangement, often achieving a diastereomer ratio as high as 99 to 1 in favor of the cis form. Unlike many earlier systems, the catalyst is solid, easy to filter off, and reusable. It also leaves delicate groups like esters, boronic esters, and amides intact, which is crucial when the ring is part of a complex drug or functional material.
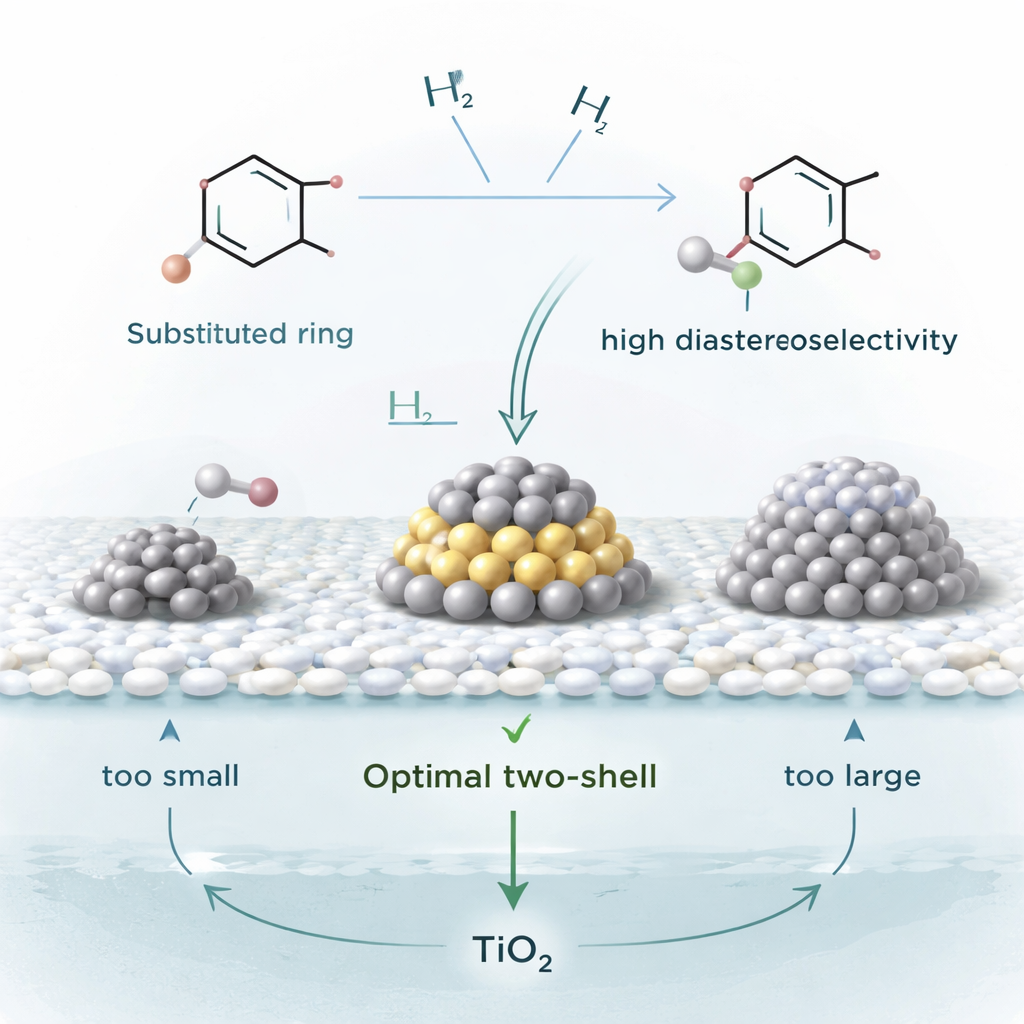
Zooming In on the Sweet Spot
To understand why this catalyst works so well, the team studied a benchmark reaction: turning dimethyl phthalate, a common industrial chemical, into its saturated counterpart. By preparing Pt/TiO2 with different platinum loadings and measuring reaction rates, they discovered that the highest activity occurs when the platinum particles have a very specific intermediate size. Electron microscopy images and computer simulations showed that particles with a so‑called two‑shell structure—big enough to host both the flat ring and hydrogen at the same time, but not so big that the ring binds too weakly—are the true “sweet spot.” Smaller clusters get clogged by the strongly sticking ring, while much larger particles do not grip the ring firmly enough to match the observed behavior.
From Model Reactions to Real‑World Products
Armed with this insight, the scientists explored how broadly the catalyst could be used. They successfully transformed many different substituted benzene derivatives and fused or strained ring systems, usually with high yields and strong preference for the cis products. Crucially for medicinal chemistry, they also applied the method to nitrogen‑containing heteroarenes that serve as building blocks for important drugs, including intermediates related to the antibiotic moxifloxacin. To showcase industrial relevance, they carried out a kilogram‑scale reaction that converts a commercial phthalate plasticizer into a phthalate‑free alternative under solvent‑free conditions, obtaining almost exclusively the desired cis form and demonstrating that the catalyst can be recycled several times.
What This Means for Everyday Chemistry
In plain terms, this work provides chemists with a sturdy, reusable tool for reshaping common flat ring molecules into more three‑dimensional, finely defined forms, without lengthy synthetic detours. By pinpointing exactly which platinum structures do the heavy lifting, the study opens the door to rationally designing even better catalysts. The immediate impact could be faster routes to new drug candidates, safer and more tunable plasticizers, and more efficient use of hydrogen in chemical manufacturing—all achieved with a relatively simple solid catalyst that fits well into existing industrial processes.
Citation: Qu, R., Jena, S., Xiao, L. et al. Highly selective and practical hydrogenation of functionalized (hetero)arenes. Nat Commun 17, 2015 (2026). https://doi.org/10.1038/s41467-026-68537-7
Keywords: arene hydrogenation, platinum catalyst, 3D molecular scaffolds, heteroarenes, plasticizer synthesis