Clear Sky Science · en
Adipocytic sclerostin loop3-LRP4 interaction required by sclerostin to impair whole-body lipid and glucose metabolism
Why bones matter for blood sugar
Most people think of bones as lifeless scaffolding, but bone cells quietly release hormones that talk to the rest of the body. This study reveals how one such bone protein, called sclerostin, helps link two very common problems in older women: fragile bones (osteoporosis) and type 2 diabetes. By uncovering a specific “handshake” between sclerostin and fat cells, the researchers outline a way to strengthen bones and improve blood sugar and cholesterol without adding strain to the heart.
A bone signal that goes wrong
Sclerostin is a protein made mainly by bone cells. It normally slows down bone formation and, somewhat surprisingly, also helps protect blood vessels. The team focused on a small segment of this protein, called loop3, and asked whether it plays a role in disturbed fat and sugar handling. They found that women with postmenopausal osteoporosis who also had type 2 diabetes, as well as people newly diagnosed with diabetes, had higher levels of sclerostin in their blood than comparable individuals without diabetes. In mice fed a high-fat diet to mimic metabolic stress, sclerostin levels also climbed along with weight gain, higher blood fats, and poorer blood sugar control. These patterns suggested that excess sclerostin might be more than a bystander in metabolic disease.
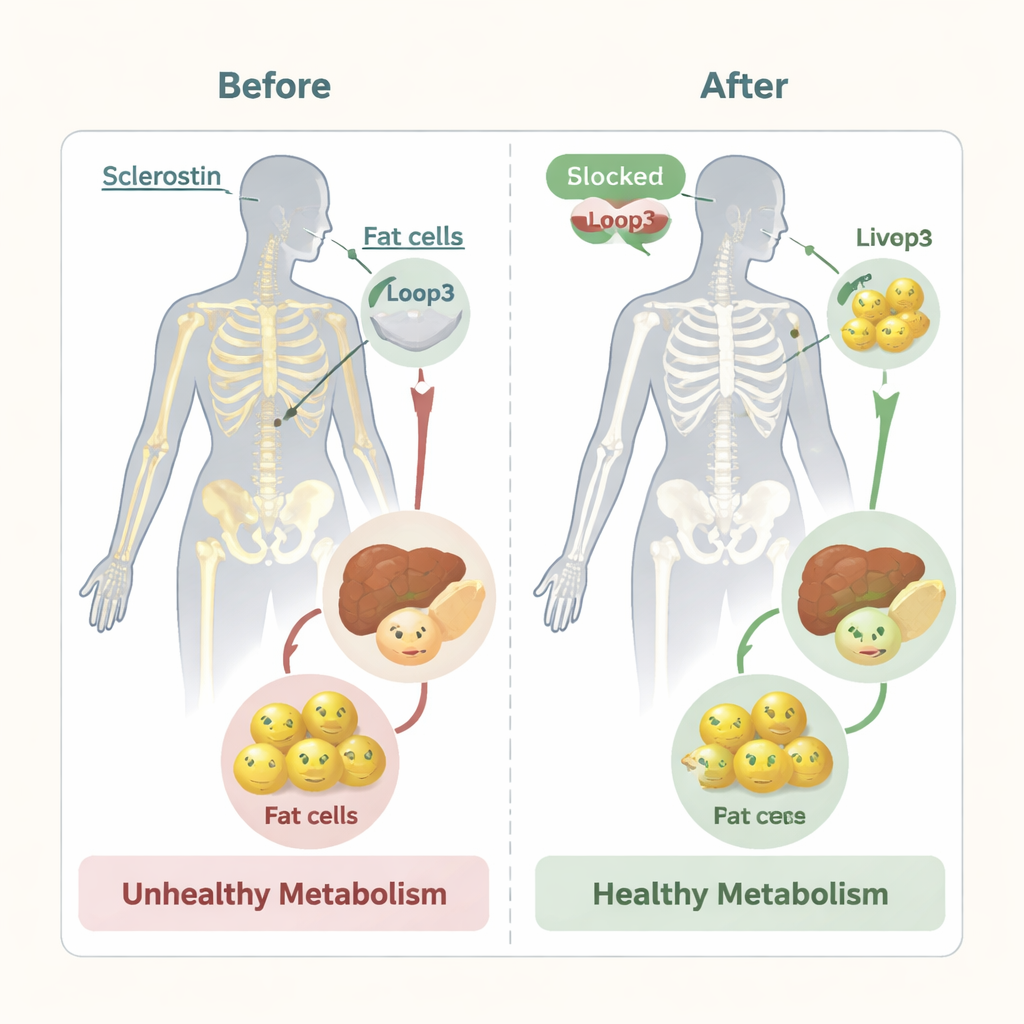
Dissecting a tiny loop with big effects
To understand what loop3 does, the researchers engineered mice in which sclerostin was altered or its loop3 segment was removed. Mice that overproduced normal sclerostin accumulated more fat in their white fat depots, had larger fat cells, higher levels of circulating fatty acids, and worse glucose tolerance and insulin response than normal mice. When loop3 was genetically deleted or subtly mutated, these problems eased: fat pads were smaller, fat cells shrank, blood fats dropped, and sugar handling improved. Remarkably, similar benefits appeared when mice were treated with a specially designed DNA-like drug (an aptamer) that locks onto loop3 and prevents it from working, without changing the amount of sclerostin in the blood.
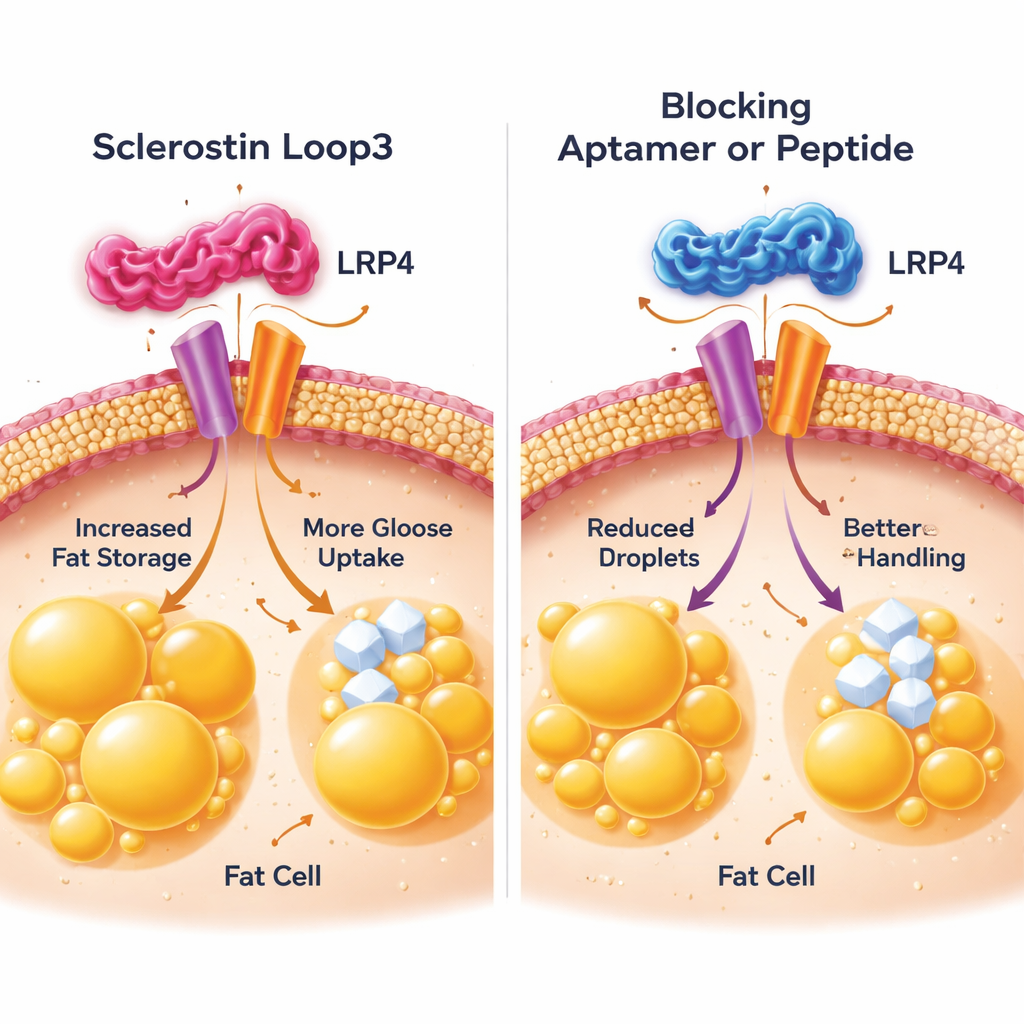
How fat cells ‘hear’ the bone signal
The group then zoomed in on fat cells to see how loop3 delivers its harmful message. They focused on a receptor protein on the fat cell surface called LRP4, previously known for helping sclerostin act on bone. Using cell culture experiments, they showed that loop3 physically binds to a specific portion of LRP4 on fat cells. This contact triggered changes that boosted the cells’ capacity to build and break down fats and to pull more glucose out of the blood, leading to oversized, overactive fat cells. When the scientists subtly altered key building blocks in LRP4 so loop3 could no longer dock, or when they added a small blocking peptide that mimicked the LRP4 docking site, the effect of sclerostin on fat buildup and sugar uptake was greatly reduced. In both cells and mice, blocking this loop3–LRP4 interaction restored more normal fat cell size and improved glucose tolerance.
Stronger bones, healthier metabolism
An important concern with existing sclerostin-blocking drugs for osteoporosis is that, while they build bone and improve metabolism, regulators have warned about increased risk of serious heart problems. Earlier work from this group showed that loop3 is crucial for sclerostin’s brake on bone formation, but not for its heart-protective actions. Here they further show that targeting loop3—either by genetic tweaks or by loop3-focused drugs—improves whole-body fat and sugar metabolism in several mouse models, while also increasing bone mass and strength. Unlike current medicines, this strategy did not trigger a compensatory rise in another bone-related inhibitor, DKK1, which can blunt long-term benefits.
What this could mean for patients
In plain terms, the study argues that a very small piece of one bone hormone helps drive both poor bone health and metabolic disease by talking directly to fat cells. By interrupting the conversation between sclerostin’s loop3 segment and the LRP4 receptor on fat cells, it may be possible to design treatments that strengthen bones and normalize blood fats and sugars, yet leave the heart-protective functions of sclerostin intact. For postmenopausal women who often face both osteoporosis and type 2 diabetes, such loop3-targeted therapies could someday offer a safer, longer-lasting way to manage two conditions at once.
Citation: Jiang, H., Tao, X., Yu, S. et al. Adipocytic sclerostin loop3-LRP4 interaction required by sclerostin to impair whole-body lipid and glucose metabolism. Nat Commun 17, 1812 (2026). https://doi.org/10.1038/s41467-026-68526-w
Keywords: sclerostin, loop3, LRP4, osteoporosis, type 2 diabetes