Clear Sky Science · en
Zebrafish macrophages convert physical wound signals into rapid vascular permeabilization
How a Tiny Fish Teaches Us About Wound Swelling
When you scrape your knee, the area quickly turns red, puffy, and leaky with fluid. This rapid change in nearby blood vessels is crucial for fighting infection and jump-starting repair, but exactly how the body senses damage and reacts within seconds has been hard to see in real time. By using see-through zebrafish larvae and advanced live imaging, this study uncovers how specialized immune cells wrapped around blood vessels convert the physical shock of a wound into almost instant vessel leakage, offering fresh insight into inflammation, healing, and even human lung injury.
Watching Injury Spread Through the Body
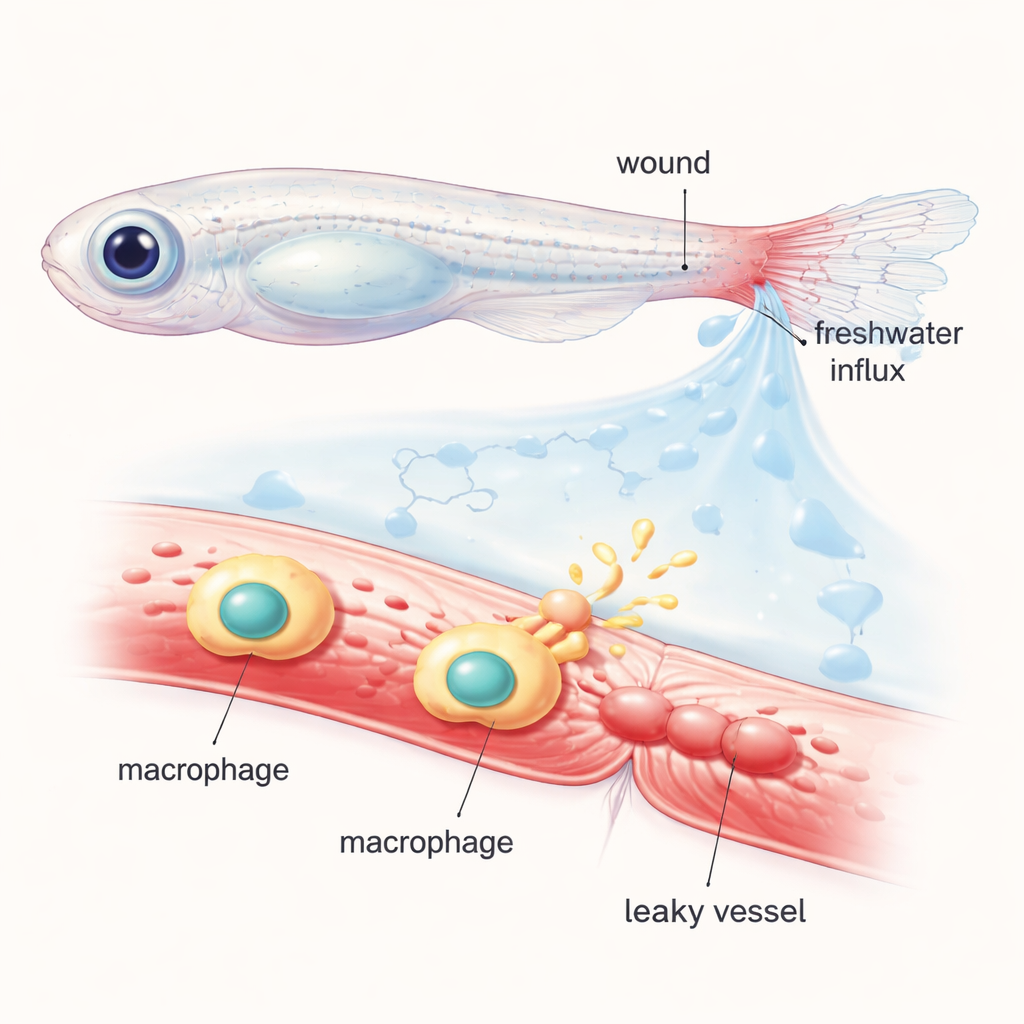
The researchers worked with young zebrafish, whose transparent bodies let scientists watch cells and blood vessels in action under the microscope. They cut the tip of the tail fin and filled the circulation with a glowing dye that stays inside healthy vessels. When the injured fish were placed back into normal freshwater, the sudden influx of water into tissues created an “osmotic shock” around the wound. Within minutes, nearby vessels dilated and began to leak the fluorescent dye into the surrounding tissue, mimicking the swelling and oozing that happen around human wounds. By shifting fish between regular and specially balanced salt or sugar solutions, the team showed that this rapid vessel permeabilization depends strongly on osmotic changes rather than on simple tissue tearing alone.
Tracing the Chemical Messengers Behind the Leak
To find out which molecules turn the physical shock into a leak signal, the scientists examined a family of fatty compounds called eicosanoids, made from arachidonic acid. These lipids are well known for their roles in inflammation and blood flow. Using genetic mutants and drugs, they selectively disabled individual enzymes in the eicosanoid pathway. Blocking a key enzyme called Alox5a (5-lipoxygenase) or its partner Lta4h sharply reduced how much dye escaped from blood vessels after osmotic shock, without stopping wound closure itself. In contrast, interfering with other related enzymes had little effect. This pointed to a specific branch of the lipid pathway as a main driver of the rapid leak response, acting almost like a chemical valve that can be opened or closed on demand.
The Surprising Role of Vessel-Hugging Immune Cells
Blood contains different types of immune cells, and in zebrafish larvae, neutrophils and macrophages are the main players. The team used a clever genetic trick to selectively destroy either neutrophils or macrophages before wounding. Removing neutrophils made little difference to vessel leakage. But when macrophages were wiped out, the leak response dropped by about half, mirroring the effect of blocking the Alox5a–Lta4h lipid pathway. Importantly, these manipulations did not stop the wound from closing quickly, showing that leukocyte recruitment and vessel leak can be controlled somewhat independently. This revealed perivascular macrophages—macrophages sitting snugly along the outside of vessels—as key translators between physical injury and vascular behavior.
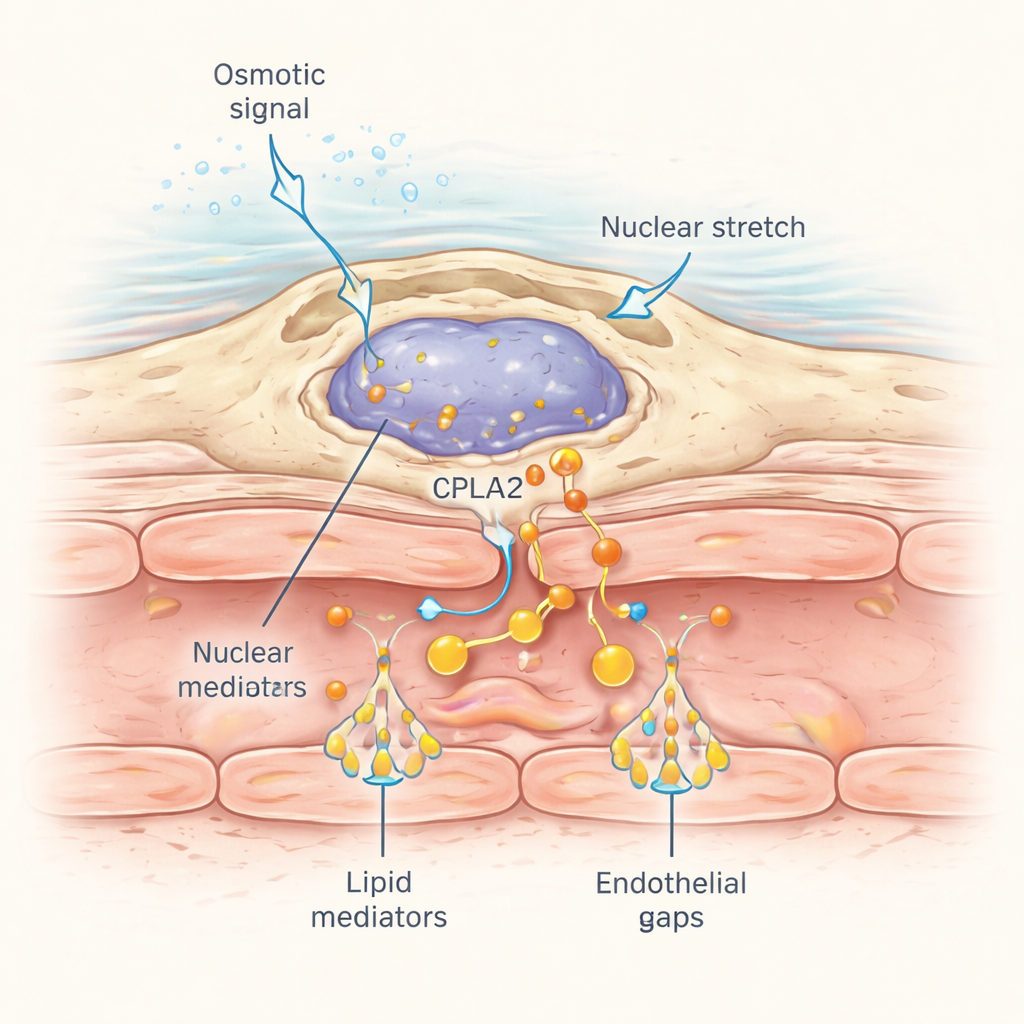
From Nuclear Stretch to Chemical Signal
How do these perivascular macrophages detect a wound that may be some distance away? The answer lies in the way their nuclei respond to mechanical stress. When osmotic shock from the wound spreads through nearby tissue, water entry causes subtle cell swelling and nuclear deformation. The researchers tagged a mechanosensitive enzyme called cPla2 with a fluorescent marker and expressed it specifically in macrophages. After wounding in normal freshwater, cPla2 rapidly moved onto the inner surface of the nuclear membrane in macrophages along a wave that swept through the tissue at about 50 micrometers per second, matching the spread of dissolved salts. This translocation depended on hypotonic conditions and was much weaker in isotonic solutions. Macrophages close to blood vessels showed especially sharp, reversible pulses of nuclear cPla2 binding, which in turn releases arachidonic acid for conversion into the leak-inducing lipids.
Why This Matters Beyond Fish
Put simply, this study shows that certain macrophages act as physical sentinels: they feel the stretch of tissue and nuclear membranes caused by osmotic changes at a wound, flip on cPla2 at their nuclear surface, and quickly produce lipid messengers that tell nearby blood vessels to “open the gates” and let fluid and immune factors flood in. Although zebrafish live in freshwater and face unique osmotic challenges, similar mechanosensitive pathways likely operate in human tissues exposed to changing pressures and flows, such as the lungs during mechanical ventilation. Understanding how nuclear stretch and lipid signals control vessel permeability in real time could eventually inform treatments that fine-tune inflammation—either dampening harmful leakage in conditions like acute lung injury, or boosting helpful responses to speed tissue repair.
Citation: Gelashvili, Z., Shen, Z., Ma, Y. et al. Zebrafish macrophages convert physical wound signals into rapid vascular permeabilization. Nat Commun 17, 1807 (2026). https://doi.org/10.1038/s41467-026-68520-2
Keywords: wound healing, blood vessels, macrophages, inflammation, zebrafish