Clear Sky Science · en
Engineering energy-efficient Saccharomyces cerevisiae for methanol and CO2 assimilation
Turning Waste Gases into Useful Products
Methanol and carbon dioxide are often treated as waste or climate-warming pollutants, but they are also rich sources of carbon and energy. This paper shows how scientists redesigned ordinary baker’s yeast so it can live on methanol and even pull in CO₂ at the same time. That kind of “gas-eating” microbe could one day help make fuels, chemicals, and materials while cutting greenhouse emissions.
Why Methanol Matters for a Warming Planet
To slow climate change, we need alternatives to fossil fuels that do not compete with food crops. Methanol made from renewable sources—such as captured CO₂, plant waste, and green hydrogen—stands out because it is easy to transport, store, and feed to microbes. Many bacteria naturally grow on methanol, but they can be hard to engineer or scale up. By contrast, the yeast Saccharomyces cerevisiae is already a workhorse of the brewing and biotech industries. Unfortunately, previous attempts to make yeast grow well on methanol ran into a basic problem: the cells did not have enough energy to power all the reactions needed to turn this simple alcohol into biomass and useful products.
Building a Yeast That Runs on Methanol
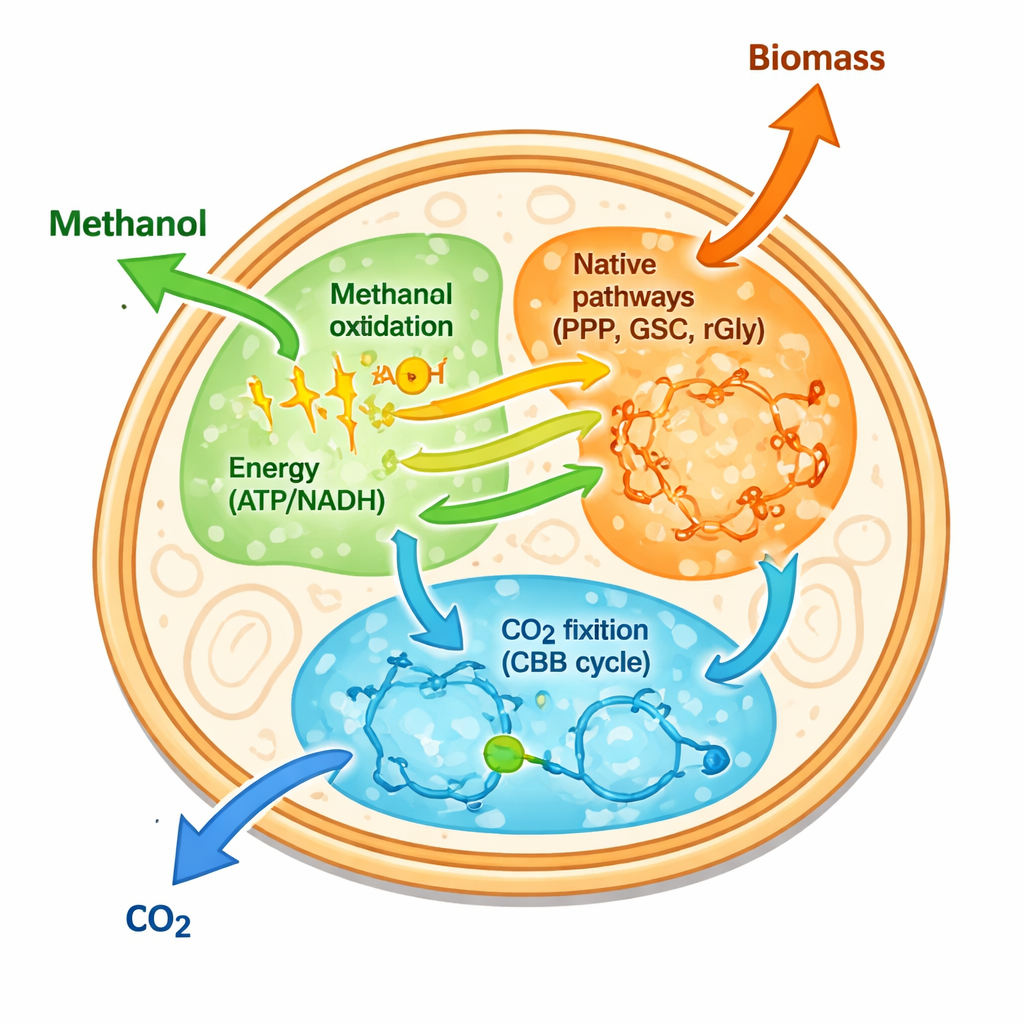
The authors tackled this by first focusing on energy, rather than on installing complete new carbon-fixing routes. They added a “methanol–formaldehyde–formate” oxidation module to yeast. This module is a chain of enzymes borrowed from other microbes that oxidizes methanol stepwise to carbon dioxide. As it does so, it generates the cellular energy currencies ATP and NADH. The team then used adaptive laboratory evolution: for months, they repeatedly grew the engineered yeast in methanol-only medium, selecting the survivors that grew a little better each time. This process yielded an evolved strain called SC-AOX25 that could more than double its cell density on methanol and grow faster than any previous methanol-using yeast strain reported.
How the Engineered Yeast Uses Carbon and Energy
With SC-AOX25 in hand, the researchers traced how methanol-derived carbon moves through the cell. Using carbon-13 labeling, they found that methanol is not only burned for energy; parts of it are also woven into amino acids and central metabolites. Three native routes in yeast turned out to be key: the pentose phosphate pathway, the glyoxylate–serine cycle, and the reductive glycine pathway. Together, these routes let the cell incorporate carbon from formaldehyde, formate, and CO₂ generated by the oxidation module. At the same time, specific mutated enzymes—named Adh2m, Aoxm, and Rgi2m, along with a native enzyme Fdh1—boosted ATP and NADH production. Knocking out these factors sharply reduced methanol use and growth, showing they form an “energy module” that underpins the new lifestyle.
Pulling CO₂ Back In with a Classic Plant Pathway
The team then asked whether this energetic yeast could also help fix extra CO₂. They introduced the Calvin–Benson–Bassham cycle, the same CO₂-fixing pathway used by plants and some bacteria, by adding plant and bacterial enzymes for the key steps. In the new strain, named SC-AOX25-CBB, labeling experiments showed that CO₂—both from the medium and from methanol oxidation—was recaptured into sugar phosphates. This extra carbon-fixing loop slightly increased growth and methanol consumption, proving that the engineered yeast can serve as a flexible platform where different one-carbon pathways are plugged together.
Dealing with the Dark Side of Methanol
Methanol’s intermediates, especially formaldehyde, are highly toxic because they can glue DNA and proteins together, forming so-called DNA–protein crosslinks. Using electron microscopy and proteomics, the authors showed that such crosslinks build up as the yeast grows on methanol and involve hundreds of essential proteins, including many tied to energy production and cell division. SC-AOX25 copes better with this stress than its ancestors, aided by both improved detoxification and large repeated DNA segments that amplify genes in ATP production and protein synthesis. These features hint at new strategies for hardening industrial strains against the chemical damage that comes with running on aggressive feedstocks.
What This Means for Future Green Biotech
In plain terms, the researchers have taught baker’s yeast to live off methanol more efficiently by giving it a strong internal power plant and then letting evolution tune the system. The resulting strain not only burns methanol for energy but also uses its own existing pathways to recycle the carbon and, with added enzymes, even refixes CO₂. This work moves us closer to microbes that can turn waste gases into everyday products, offering a potential tool for cleaner manufacturing in a carbon-constrained world.
Citation: Zhong, W., Liu, N., Chen, B. et al. Engineering energy-efficient Saccharomyces cerevisiae for methanol and CO2 assimilation. Nat Commun 17, 1806 (2026). https://doi.org/10.1038/s41467-026-68516-y
Keywords: methanol bioconversion, engineered yeast, carbon dioxide fixation, synthetic methylotrophy, green biomanufacturing