Clear Sky Science · en
Bionic Janus hydrogel drives infected Achilles tendon regeneration via mechano-immune spatiotemporal steering
Why this matters for injured tendons
Ruptured Achilles tendons are common in both athletes and older adults, and when bacteria infect the injury, healing often goes badly: the tendon can weaken, scar to nearby tissue, or even tear again. This study presents a new "smart" hydrogel bandage, inspired by the tendon’s own natural sheath, that can be painted onto an infected tendon during surgery. It aims to do three things at once: protect the tendon mechanically, kill drug-resistant bacteria, and gently steer the immune system from damaging inflammation toward true regeneration.
A two-sided bandage inspired by nature
Healthy tendons sit inside a slippery sheath that lets them glide smoothly while still being firmly anchored. Surgeons struggle to recreate this after repair: materials that stick well to the tendon often also stick to surrounding tissue, causing painful adhesions. The researchers copied this natural "adhesion plus lubrication" design with a Janus (two-faced) hydrogel called HAPP@H-EXO. One face strongly grips the tendon surface using reversible chemical bonds and hydrogen bonding, giving solid mechanical support. The opposite face is engineered, using a lotus-leaf-like treatment, to be water-repelling and low-friction so that nearby tissues do not stick. This creates a protective sleeve that moves with the tendon but resists scarring to its surroundings. 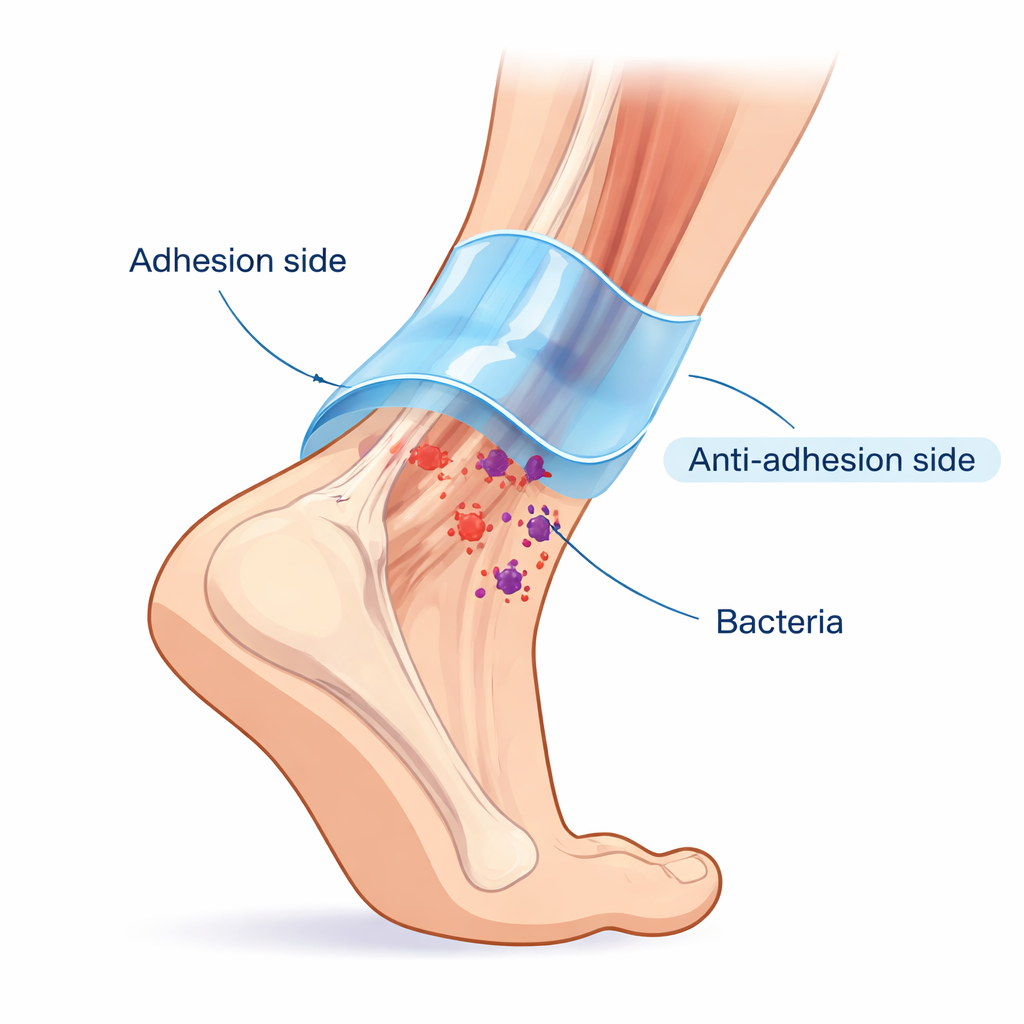
Built to share the load and survive repeated strain
Unlike many soft gels, this material is designed to live in a harsh mechanical environment: every step loads and unloads the Achilles tendon. The team combined a stiff, permanently crosslinked network with a second, dynamic network that can rearrange under stress. Tests showed the hydrogel stretches and compresses without tearing, maintains strength over at least 100 force cycles, and dissipates little energy, meaning it springs back rather than slowly deforming. Computer simulations suggested that, when wrapped around a sutured tendon, the gel spreads out the high stresses from the stitch points toward the tendon edges, reducing the chance of a new tear at the repair site. In animals, this translated into repaired tendons whose strength and stiffness approached those of normal tissue.
Capturing bacteria and resisting drug resistance
Bacterial infection is a major reason tendon repairs fail, especially when caused by multidrug-resistant strains like MRSA. Instead of relying on traditional antibiotics, the hydrogel physically catches and kills bacteria. One component, a phenylboronic acid group, recognizes sugar-rich structures on bacterial cell walls and forms reversible bonds with them, pulling bacteria out of the surrounding fluid. Another component, a positively charged polymer, then destabilizes bacterial membranes, causing them to rupture. In lab tests, the gel rapidly killed MRSA, common Staphylococcus, and E. coli, broke down tough biofilms, and maintained its antibacterial power over days and multiple use cycles without provoking bacterial resistance to the capture or killing mechanisms. 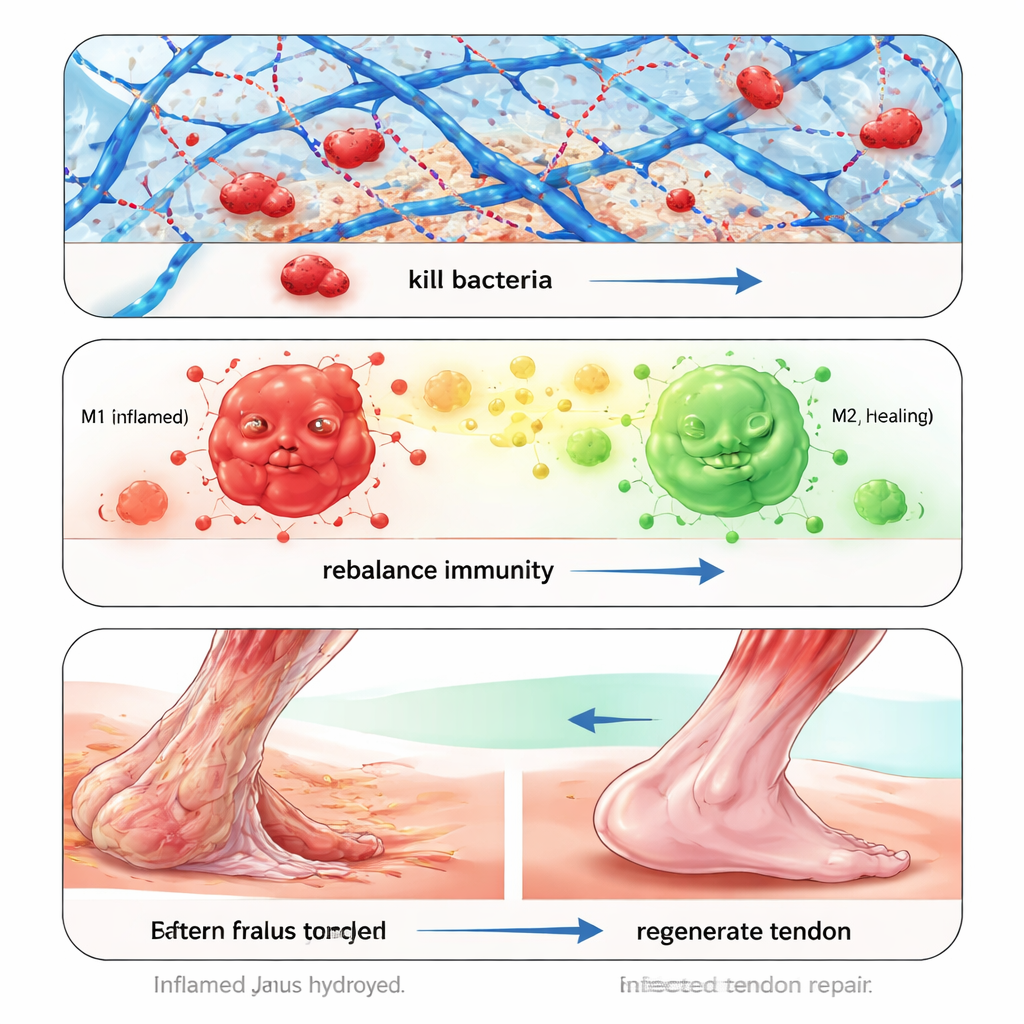
Guiding the immune system and rebuilding tissue
Even if bacteria are removed, infected tendons often remain in a state of oxidative stress and chronic inflammation, which blocks normal repair. To address this, the researchers loaded the hydrogel with tiny membrane packets—exosomes—secreted by tendon stem cells grown under low-oxygen conditions. These "hypoxic" exosomes are rich in anti-inflammatory and pro-regenerative signals. The gel’s chemistry makes it release exosomes faster in the acidic, inflamed early phase and more slowly as conditions normalize. In cell experiments, exosome-loaded gels reduced harmful reactive oxygen species, restored mitochondrial function, encouraged blood vessel formation, and nudged immune cells away from an inflammatory state (M1) toward a healing state (M2). Gene analyses pointed to dampening of the NF-κB pathway, a central driver of inflammation.
From infected rupture to functional recovery
In rat and rabbit models of Achilles rupture deliberately infected with MRSA, applying the Janus hydrogel during surgery nearly eliminated live bacteria at the injury site within a week. Over the following weeks, treated tendons showed less oxidative damage, fewer inflammatory markers, and more pro-healing signals than untreated controls. Microscopy revealed better-aligned, thicker collagen fibers, more blood vessels, and expression of key tendon-maturity proteins. Importantly, the outer non-stick face of the gel prevented the tendon from adhering to nearby tissues, confirmed by imaging and gross inspection. Animals treated with the exosome-loaded version regained more normal gait patterns and higher tendon load-bearing capacity, indicating not just structural repair but functional restoration.
What this could mean for patients
This work demonstrates a single, biomimetic material that tackles mechanical support, infection control, and immune balance in an integrated way. By combining a two-sided physical design with bacteria-trapping chemistry and timed release of regenerative exosomes, the Janus hydrogel helped infected Achilles tendons in animals heal stronger, with fewer scars and better movement. While human trials are still needed, the approach suggests a future in which complex soft-tissue injuries, especially those complicated by drug-resistant infections, could be treated with "smart" surgical dressings that actively orchestrate the healing process.
Citation: Li, J., Wang, Z., Yang, W. et al. Bionic Janus hydrogel drives infected Achilles tendon regeneration via mechano-immune spatiotemporal steering. Nat Commun 17, 1805 (2026). https://doi.org/10.1038/s41467-026-68514-0
Keywords: Achilles tendon repair, hydrogel bandage, drug-resistant infection, tissue regeneration, immune modulation