Clear Sky Science · en
Mechanical forces from intercellular peptide self-assembly drive spheroid formation
Building Tiny Tissues in the Lab
Scientists are racing to create miniature versions of human tissues that behave like the real thing. These living models can make cancer drugs safer to test and bring new treatments for diabetes and other diseases closer to reality. This study introduces a clever way to coax cells into forming realistic three-dimensional clusters, complete with their own primitive “blood vessel–like” networks that improve nutrient delivery and function.
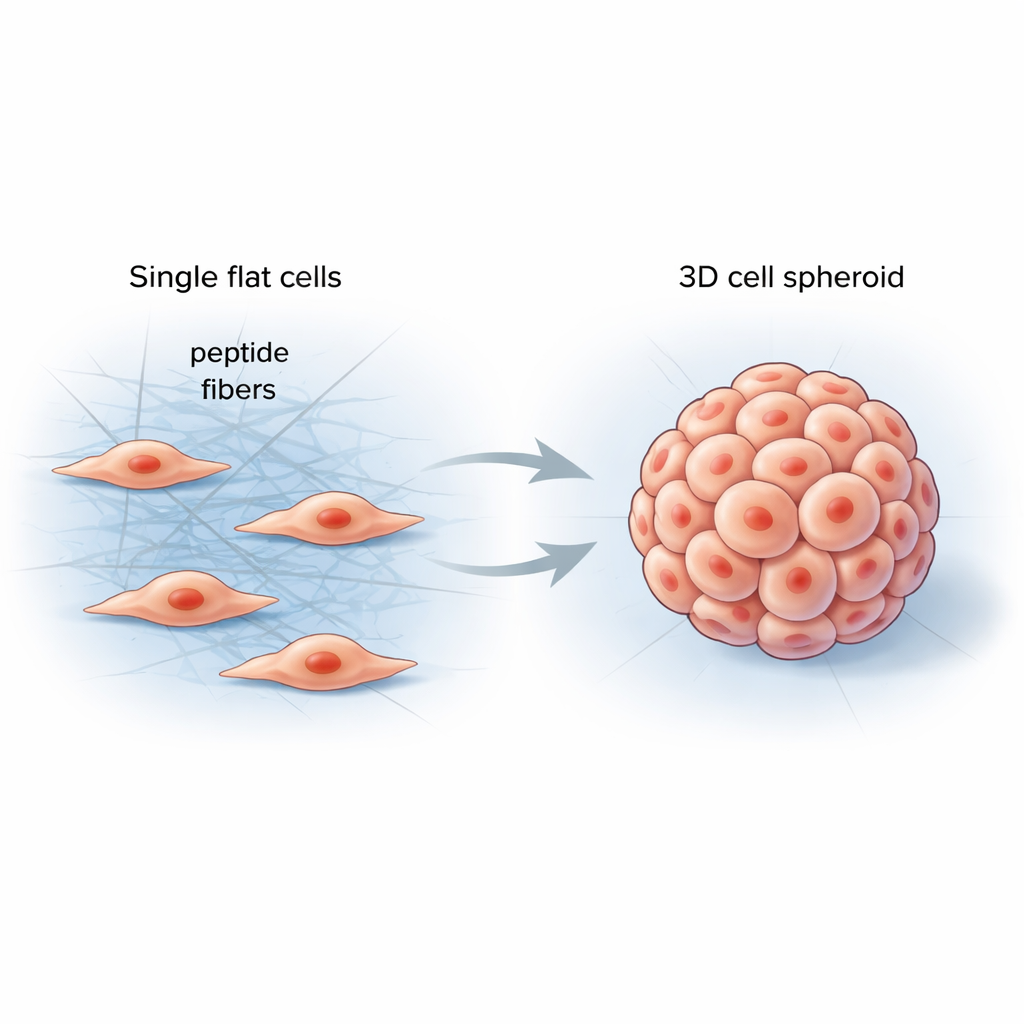
Helping Cells Feel Their Surroundings
Inside our bodies, cells live in a soft yet structured environment called the extracellular matrix—a mesh of proteins that supports tissues and sends constant mechanical signals to cells. Conventional lab materials struggle to mimic this complexity, often producing flat cell layers that behave very differently from real organs or tumors. The researchers set out to design a simple, programmable material that would form directly between cells, stiffen their surroundings in a controlled way, and nudge them into more natural three-dimensional shapes.
Smart Peptides That Assemble Between Cells
The team created short protein fragments, called peptides, that can link together into long, thin fibers when they encounter specific enzymes on cell surfaces. When these peptides were added to cancer cells, they remained inactive at first, like a clear solution. Once the cells’ own enzymes trimmed part of the molecule, the peptides rapidly self-assembled into a fibrous network wrapped around and between neighboring cells. This new fiber mesh changed the local mechanical forces, encouraging cells to pull on one another, remodel their internal scaffolding, and gather into round 3D spheroids instead of spreading out in a sheet.
From Cell Clusters to Vessel-Like Networks
To move closer to real tissue, the scientists next introduced human endothelial cells, the type that normally lines blood vessels, along with a second peptide carrying a small “address tag” that attracts these cells. Together, the original and modified peptides formed a shared nanofiber scaffold. Endothelial cells used this scaffold to sprout into tube-like extensions that wrapped around and penetrated the spheroids, creating capillary-like structures. When the team flowed fluorescent sugar-like molecules around these structures, they saw much deeper penetration into the spheroid core compared with spheroids lacking these inner tubes, showing that transport of nutrients and signals was markedly improved.
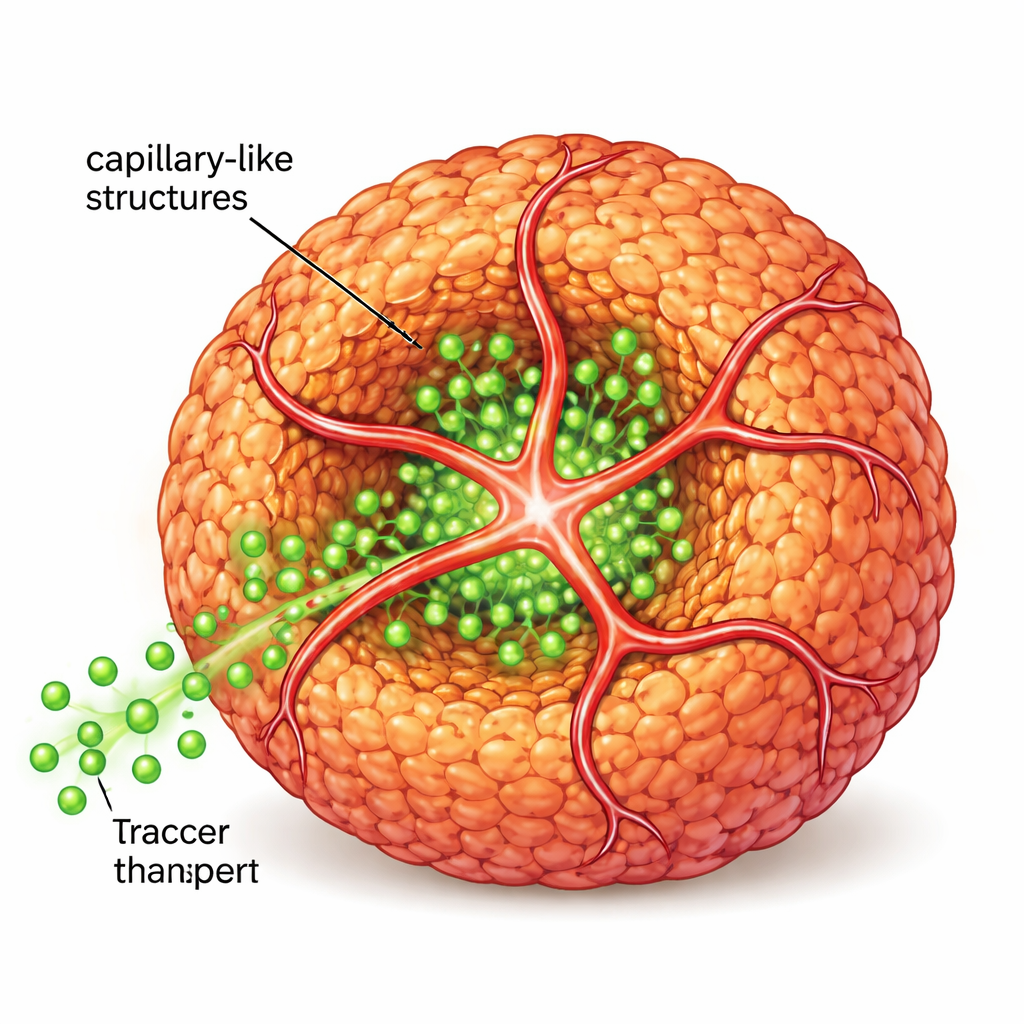
Closer to Real Tumors and Working Islets
Gene-activity profiling revealed that tumor spheroids with capillary-like structures more closely resembled patient glioblastoma samples than did traditional flat cultures or non-vascularized spheroids. In mice, spheroids containing these primitive vessel networks grew into larger, more vascularized tumors, indicating that the in vitro model captures key features of real tumor behavior. The same strategy was applied to pancreatic beta cells, which normally live in highly vascularized islets and secrete insulin in response to glucose. When beta-cell spheroids were equipped with capillary-like structures using the peptide system and endothelial cells, they showed much stronger, more finely tuned insulin release, suggesting a powerful tool for diabetes research and regenerative medicine.
Why This Matters for Future Therapies
By letting simple peptides self-assemble between living cells, this work offers a general recipe for building miniature, vascularized tissues that feel and function more like those inside the body. The resulting spheroids better mirror patient tumors and working islets, making them valuable for testing drugs, studying disease mechanisms, and designing new cell-based therapies. For non-specialists, the key takeaway is that giving lab-grown cells the “right feel” and tiny vessel-like networks can dramatically improve how faithfully they mimic human tissues, potentially speeding the path from basic research to effective treatments.
Citation: Lu, H., Li, Y., Yang, X. et al. Mechanical forces from intercellular peptide self-assembly drive spheroid formation. Nat Commun 17, 1801 (2026). https://doi.org/10.1038/s41467-026-68513-1
Keywords: tissue engineering, cancer models, angiogenesis, organoids, peptide self-assembly