Clear Sky Science · en
Intermittent fasting inhibits Tp53-driven glioma through gut microbiota-mediated methionine-m6A regulation
Why giving the gut a break might slow deadly brain tumors
Glioblastoma is one of the most aggressive brain cancers, and current treatments—surgery, radiation, and chemotherapy—often buy patients only a bit more time. This study asks a simple but powerful question with big implications for everyday life: can the way we eat, specifically intermittent fasting, change our gut bacteria and blood chemistry enough to meaningfully slow certain brain tumors?
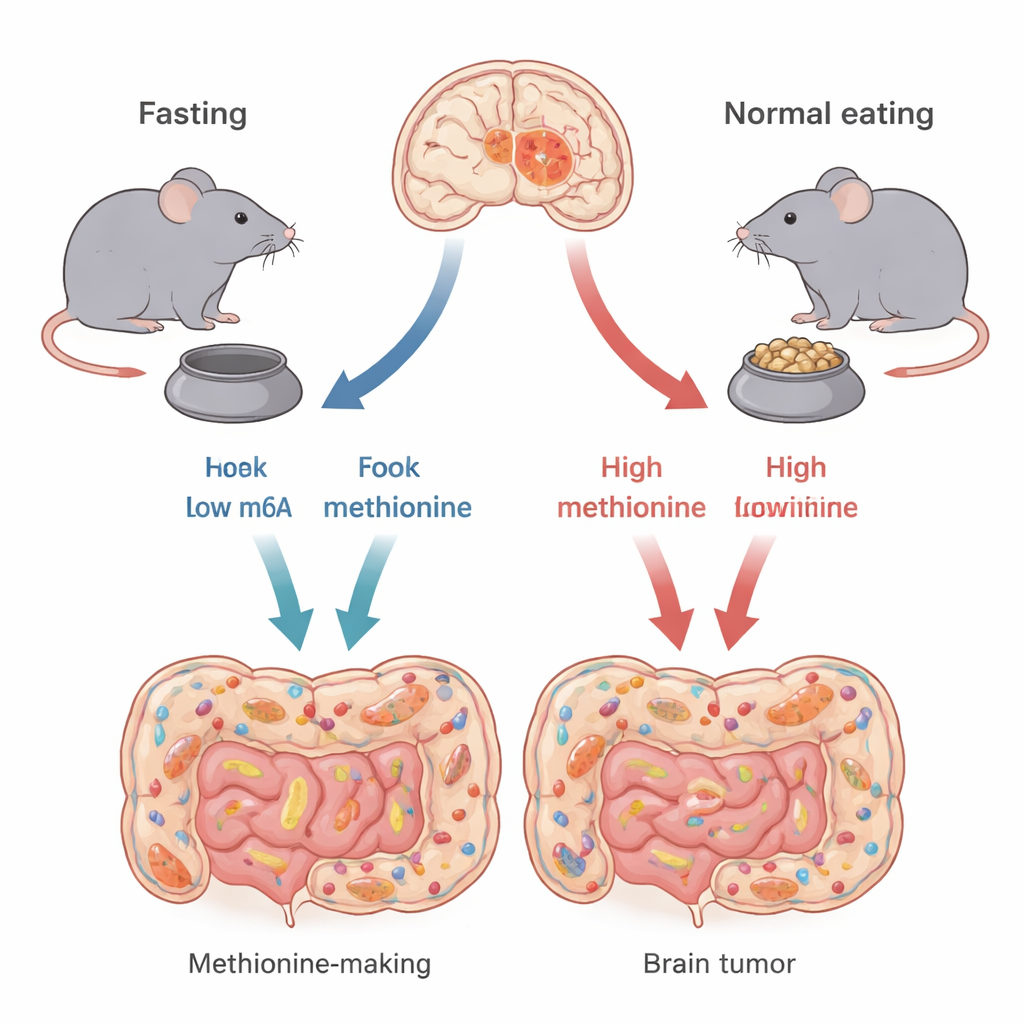
Different kinds of the “same” brain cancer
Not all glioblastomas are alike. The authors focus on two major genetic types, defined by changes in genes called TP53 and CDKN2A. These genes help control how cells grow or die. Using mouse models that closely mimic each human subtype, the researchers show that intermittent fasting—24 hours without food followed by 24 hours with food—does not help all brain tumors equally. Mice with TP53-type tumors lived longer and had smaller tumors when they fasted, while mice with CDKN2A-type tumors showed little benefit. This suggests that whether fasting can help may depend strongly on the tumor’s genetic wiring.
From food pattern to gut bugs to brain chemistry
The team then asked how fasting exerts its selective effect. Because the total amount of food was kept the same between groups, the key difference was the timing of eating. That timing powerfully reshaped the gut microbiota—the vast community of bacteria in the intestine. In fasting TP53 mice, certain bacterial groups that efficiently make the amino acid methionine, such as specific Alistipes and Prevotella strains, declined in abundance. Since methionine is an essential building block that animals must obtain from diet or gut microbes, this microbial shift meant less methionine flowing from the gut into the bloodstream and ultimately into the brain.
How a single nutrient helps tune cancer-driving signals
Methionine is not just a nutrient; it is also raw material for S-adenosylmethionine (SAM), the cell’s universal “methyl donor.” SAM feeds a chemical tagging process on RNA called m6A. These tags help control how strongly certain genes are expressed. In the TP53 tumor model, fasting lowered methionine and SAM levels in blood and in brain tumor regions. Using several layers of “omics” technologies—standard RNA sequencing, single-cell RNA profiling, and spatial maps of both gene activity and metabolites—the researchers found fewer m6A tags on key cancer‑linked RNAs under fasting. One important target was TGFB2, a gene in the TGF‑β signaling pathway, which is known to drive tumor growth and invasion. With fasting, the enzyme METTL3 (which writes m6A marks) and TGFB2 were both dialed down, and TGF‑β signaling weakened.
Proving cause and effect with targeted interventions
To move beyond correlation, the scientists ran “rescue” experiments. They blocked METTL3 with a drug, or transferred gut bacteria from fasting mice into normally fed mice, and saw tumor growth slow even without fasting—supporting the idea that both the microbiota and the m6A machinery are key players. Conversely, when fasting mice were given extra methionine or treated with a drug that boosts m6A marks, their tumors grew more like those of normally fed mice, and the survival advantage disappeared. Importantly, disrupting gut bacteria with broad antibiotics erased many of the fasting-related changes, underscoring that the gut community is a central hub in this chain of events.
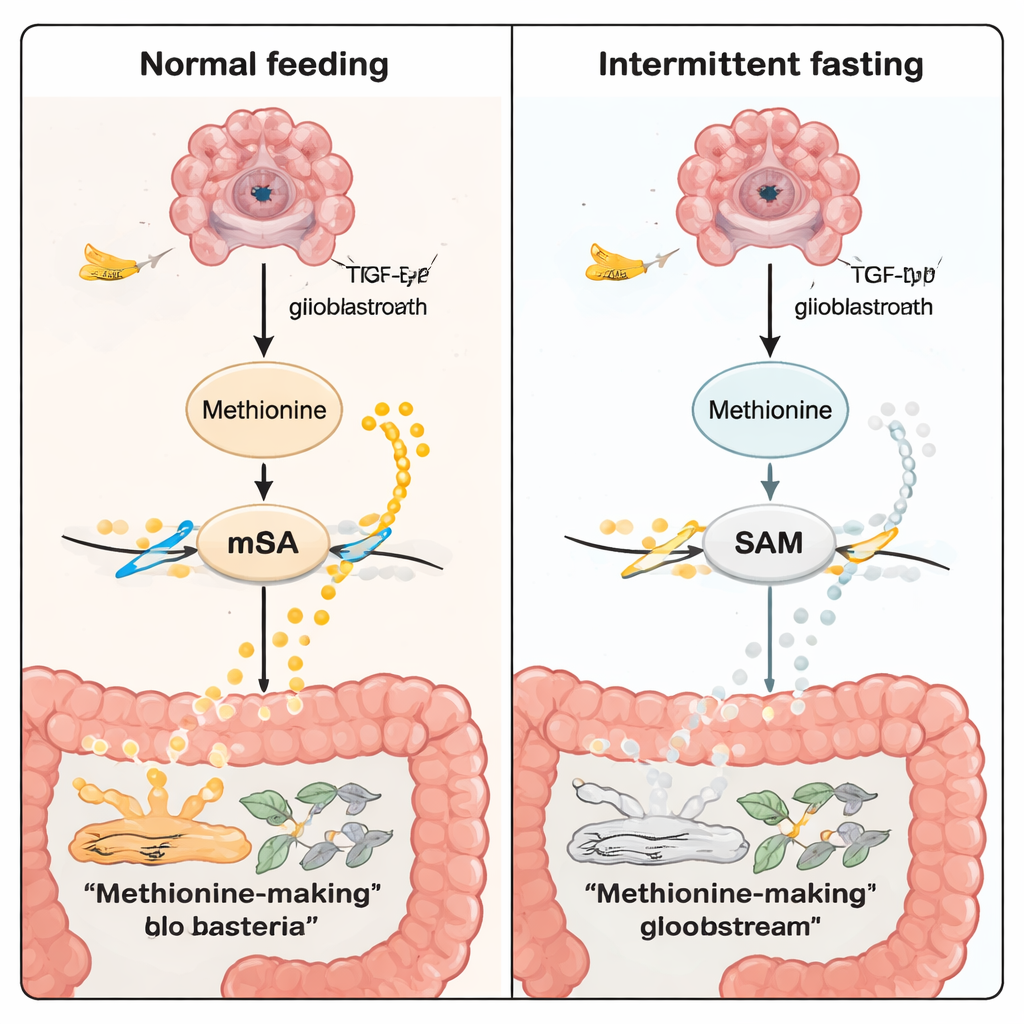
What this means for people and future therapies
For a layperson, the core message is that not all cancers, or even all glioblastomas, will respond the same way to a dietary strategy like intermittent fasting. In TP53-type gliomas, fasting appears to “talk” to the brain through the gut: it reshapes intestinal bacteria, which in turn lowers methionine production, reduces RNA chemical tagging, and turns down powerful growth signals inside tumor cells. While this work is in mice and needs careful clinical translation, it points toward a future where simple lifestyle patterns, gut‑microbiota–based treatments, and drugs targeting methionine or m6A pathways could be tailored to the genetic subtype of a patient’s tumor.
Citation: Lin, Y., Li, S., Xu, X. et al. Intermittent fasting inhibits Tp53-driven glioma through gut microbiota-mediated methionine-m6A regulation. Nat Commun 17, 1804 (2026). https://doi.org/10.1038/s41467-026-68512-2
Keywords: intermittent fasting, glioblastoma, gut microbiome, methionine metabolism, RNA m6A modification