Clear Sky Science · en
Simultaneous sulfide oxidation and sulfate reduction for intracellular redox homeostasis under highly acidic conditions
Why cleaning toxic gases needs clever microbes
Many industries release hydrogen sulfide, a poisonous, rotten-egg–smelling gas that can corrode equipment, foul the air, and harm workers. To clean it up, engineers increasingly rely on microbes that transform sulfide into safer forms of sulfur. This paper reveals an unexpected microbial strategy: a newly identified Mycobacterium that runs two normally opposing chemical pathways at the same time to both detoxify sulfide rapidly and protect itself from internal damage, even in extremely acidic conditions.
A tug-of-war in the sulfur cycle
In nature, the sulfur cycle is driven by two opposite reactions. Some microbes oxidize sulfide (the most reduced, toxic form) to less harmful products such as elemental sulfur or sulfate, gaining energy in the process. Others go the opposite way, reducing sulfate back to sulfide so they can build essential cell components like sulfur-containing amino acids. Because these reactions undo each other, biologists long assumed that a single cell would avoid running both at once; doing so would seem wasteful, like heating and cooling a house with the windows open. 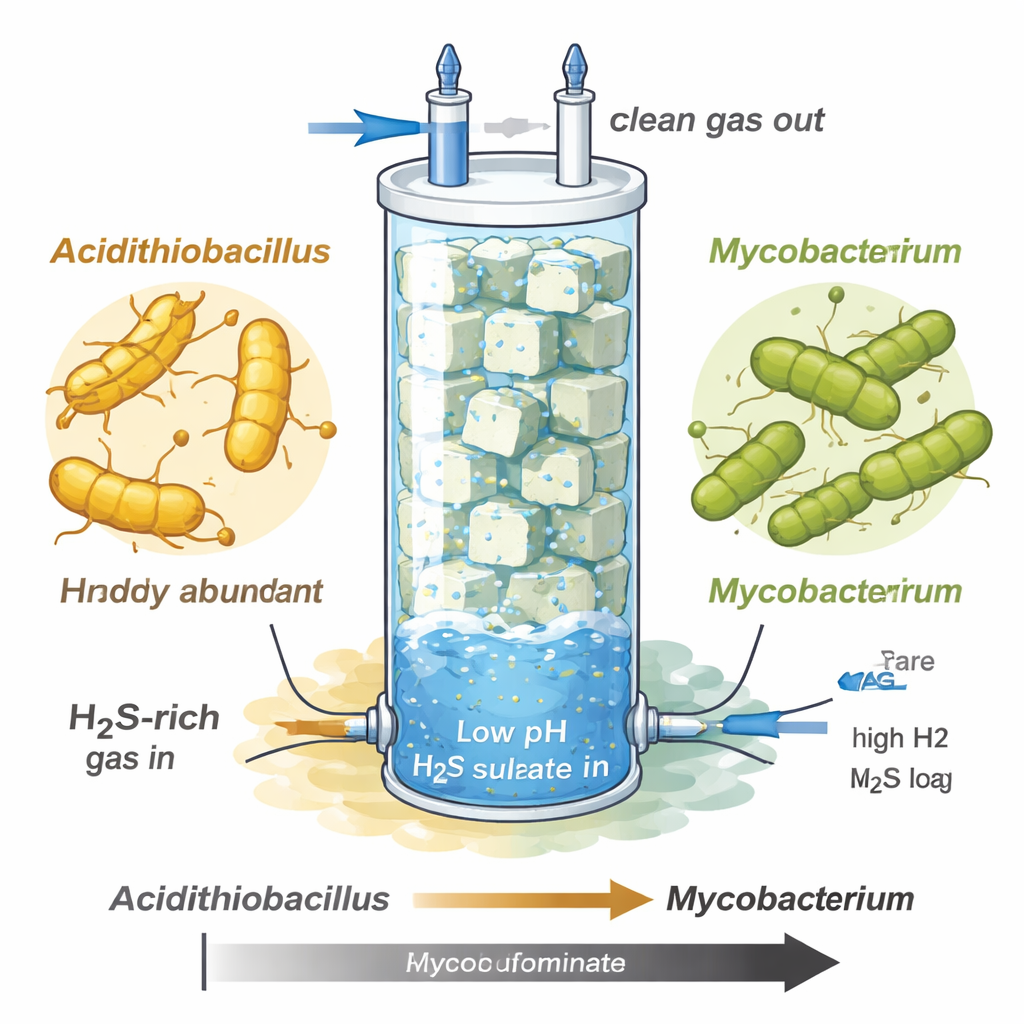
Finding a survivor in an extreme reactor
The researchers studied a biotrickling filter—a tall glass column packed with foam cubes—designed to scrub hydrogen sulfide from gas streams similar to raw biogas or natural gas. As they cranked up the sulfide load and kept the system very acidic (pH around 1–1.5), the microbial community shifted dramatically. At first, a well-known sulfide oxidizer called Acidithiobacillus thrived and turned much of the sulfide into sulfate, but it later crashed under the extreme sulfide stress. In its place, a previously uncharacterized Mycobacterium species, labeled MAG-M116, took over almost the entire community, despite growing more slowly in the early, milder phase.
A microbe that breaks the usual rules
Genetic and protein-level analyses showed that MAG-M116 has an unusual metabolic setup. It carries a key enzyme, sulfide:quinone oxidoreductase, that oxidizes sulfide only as far as solid elemental sulfur, which accumulates as tiny particles. At the same time, the microbe has a full set of genes for assimilatory sulfate reduction, the pathway that pulls sulfate into the cell and reduces it to build amino acids like cysteine and methionine. Under high sulfide conditions, both pathways were switched on together. Sulfide oxidation pumped electrons into the cell’s energy machinery, while sulfate reduction soaked up part of that electron flow, linking the two directions of the sulfur cycle inside a single cell.
Turning a “wasteful” cycle into a safety valve
Running both pathways at once could, in theory, be futile—burning energy without net gain. But the team shows that for MAG-M116 it acts as a safety valve for excess electrons. Oxidizing large amounts of sulfide releases a surge of electrons into the cell’s respiratory chain, which can leak and generate reactive oxygen species (ROS)—damaging molecules that attack DNA, proteins, and membranes. By actively reducing sulfate to amino acids, the cell diverts 10–14% of those electrons into constructive work, lowering the buildup of an over-reduced, leak-prone state. Experiments comparing conditions with and without sulfate showed that active sulfate reduction cut ROS production by up to about 60% and prevented the buildup of reduced cofactors that would otherwise fuel oxidative damage. 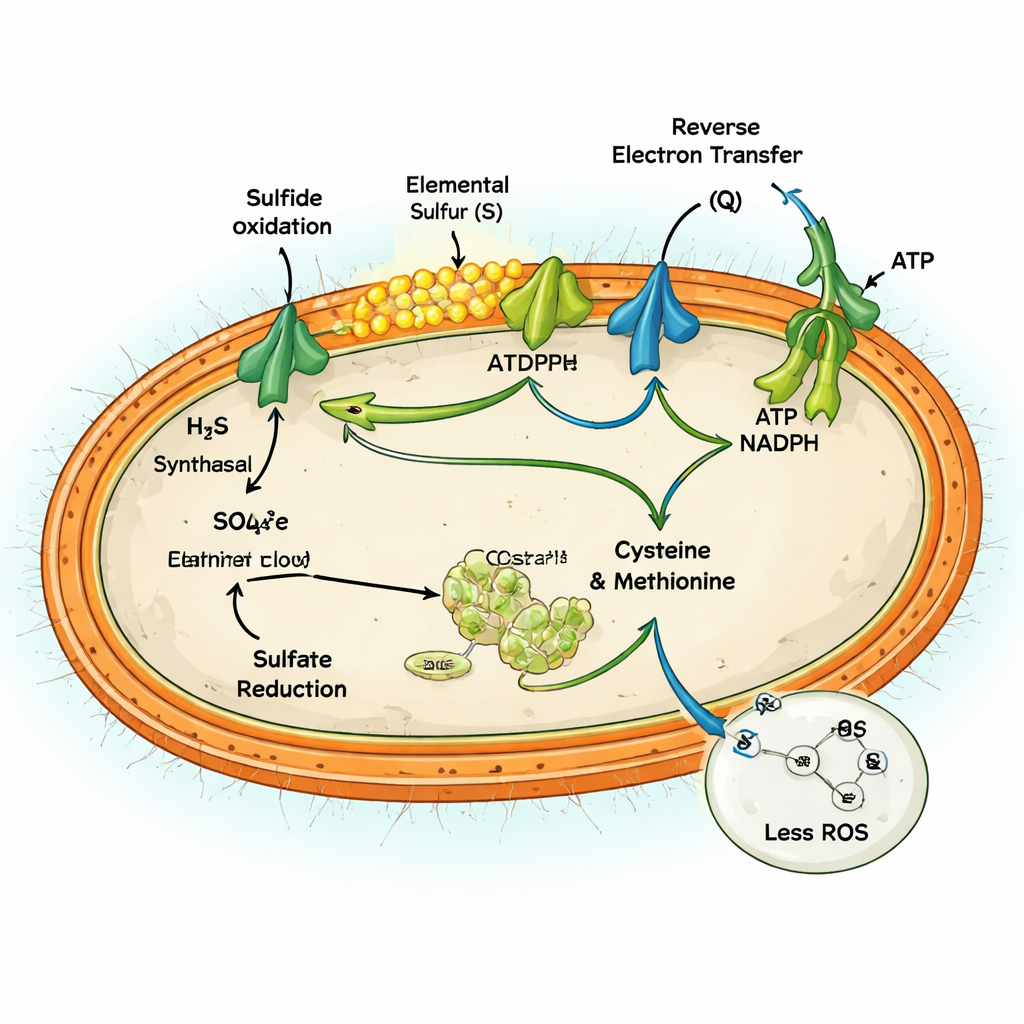
From toxic gas to useful products
In the later stages of reactor operation, when MAG-M116 dominated, nearly all incoming hydrogen sulfide was converted into solid elemental sulfur, rather than sulfate. At the same time, the researchers detected rising levels of cysteine and methionine in the liquid phase, suggesting the microbe was releasing surplus sulfur-rich amino acids into its surroundings. This combination—stable elemental sulfur plus valuable organic sulfur compounds—turns what was once a hazardous pollutant into a mix of recoverable products with uses in agriculture, materials, and energy systems.
What this means for health and technology
To a non-specialist, the key message is that this Mycobacterium has solved a double problem: it can clean up high levels of a toxic gas while keeping its own internal chemistry in balance. It does so by running “push–pull” sulfur reactions that would normally be kept separate, using the supposedly wasteful cycle as a built-in shock absorber for excess electrons and harmful oxygen radicals. This discovery not only suggests new ways to design robust, low-pH bioreactors for sulfide and sulfate removal, but also hints that related microbes, including disease-causing Mycobacterium species, may use similar tricks to survive oxidative attacks inside the human body.
Citation: Jia, T., Peng, Y., Niu, L. et al. Simultaneous sulfide oxidation and sulfate reduction for intracellular redox homeostasis under highly acidic conditions. Nat Commun 17, 1797 (2026). https://doi.org/10.1038/s41467-026-68508-y
Keywords: sulfur cycle, hydrogen sulfide, Mycobacterium, redox homeostasis, biodesulfurization