Clear Sky Science · en
An artificial cell capable of signal transduction mediated by ADRB2 for the regulation of glycogenolysis
Teaching Tiny Bubbles to Talk
Imagine a microscopic soap bubble that can sense a hormone-like signal outside and then “decide” to burn stored fuel inside, much like a real human cell. This study builds exactly that sort of artificial cell, showing how man‑made cell mimics can receive a chemical message and turn it into controlled energy‑related chemistry. Such work edges us closer to smart drug carriers, lab‑made tissues, and simple synthetic life forms that can react to their surroundings.
Building a Cell from the Outside In
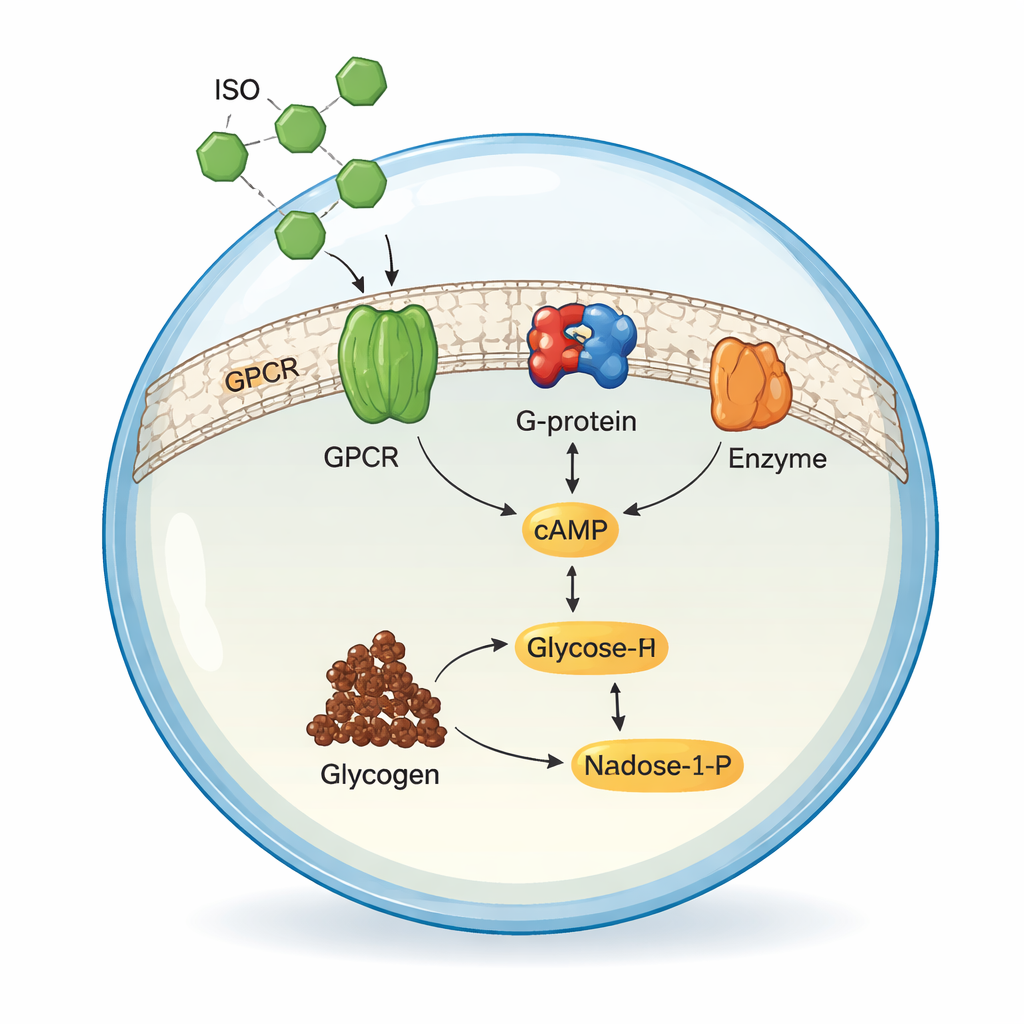
Real cells constantly listen to their environment through receptors in their outer membrane. The authors set out to copy one of these natural pathways using only purified components and simple fat bubbles, called giant vesicles. They focused on a common human receptor, the β2‑adrenergic receptor (ADRB2), which in our bodies helps control heart rate, lung function, and fuel use. When this receptor meets a drug like isoproterenol (ISO), it normally triggers an internal chain of events that produces a messenger molecule called cAMP, which in turn controls how cells break down glycogen, the body’s stored form of glucose. Reproducing this whole sequence in an artificial cell had not previously been achieved.
Recreating the First Signal Relay
The team first rebuilt the early stages of the signaling pathway in solution, outside any membrane. They produced three human proteins in insect cells: ADRB2, its partner G‑protein subunit (Gsα), and an enzyme called adenylate cyclase V (ADCY5), which makes cAMP from ATP. When they mixed these parts with ISO under carefully tuned conditions, adrenergic receptor activation led to ADCY5 turning ATP into cAMP. By measuring cAMP with high‑performance liquid chromatography, the researchers optimized pH, temperature, and magnesium levels and found that their reconstituted system worked as efficiently as, or better than, many previous preparations, confirming that the core signaling chemistry was in place.
Installing Real Receptors in Artificial Membranes
Next, the authors embedded the three proteins into the membranes of giant unilamellar vesicles—cell‑sized lipid bubbles that serve as the chassis for artificial cells. They fluorescently tagged ADRB2 and ADCY5 to verify that the proteins sat in the membrane, moved freely, and were present in large numbers, with roughly 1.8 million receptors per vesicle. Enzymatic treatment showed that more than 94% of these receptors faced the correct way, with their binding sites exposed to the outside. A fluorescent GTP probe confirmed that ISO binding at the membrane really did activate G‑proteins inside the vesicles. This meant the artificial cells could, in principle, transmit an external ISO signal across their membrane just as natural cells do.
Turning Signals into Fuel Breakdown
To see whether these synthetic cells could do something useful with the signal, the team added a sensor for cAMP and then a full glycogen‑breaking pathway inside the vesicles. Using a FRET‑based cAMP probe, they showed that adding ISO outside the vesicles caused cAMP levels to rise inside in a dose‑dependent fashion then level off as the receptors saturated, mirroring classic behavior of natural G‑protein‑coupled receptors. Drugs that block or silence ADRB2—alprenolol and carazolol—shut down this response exactly as expected. When the authors also encapsulated five enzymes that normally turn glycogen into glucose‑1‑phosphate (G‑1‑P) and then into NADPH, ISO stimulation led to measurable production of both G‑1‑P and NADPH inside the artificial cells, tracked by mass spectrometry, chromatography, and fluorescence.
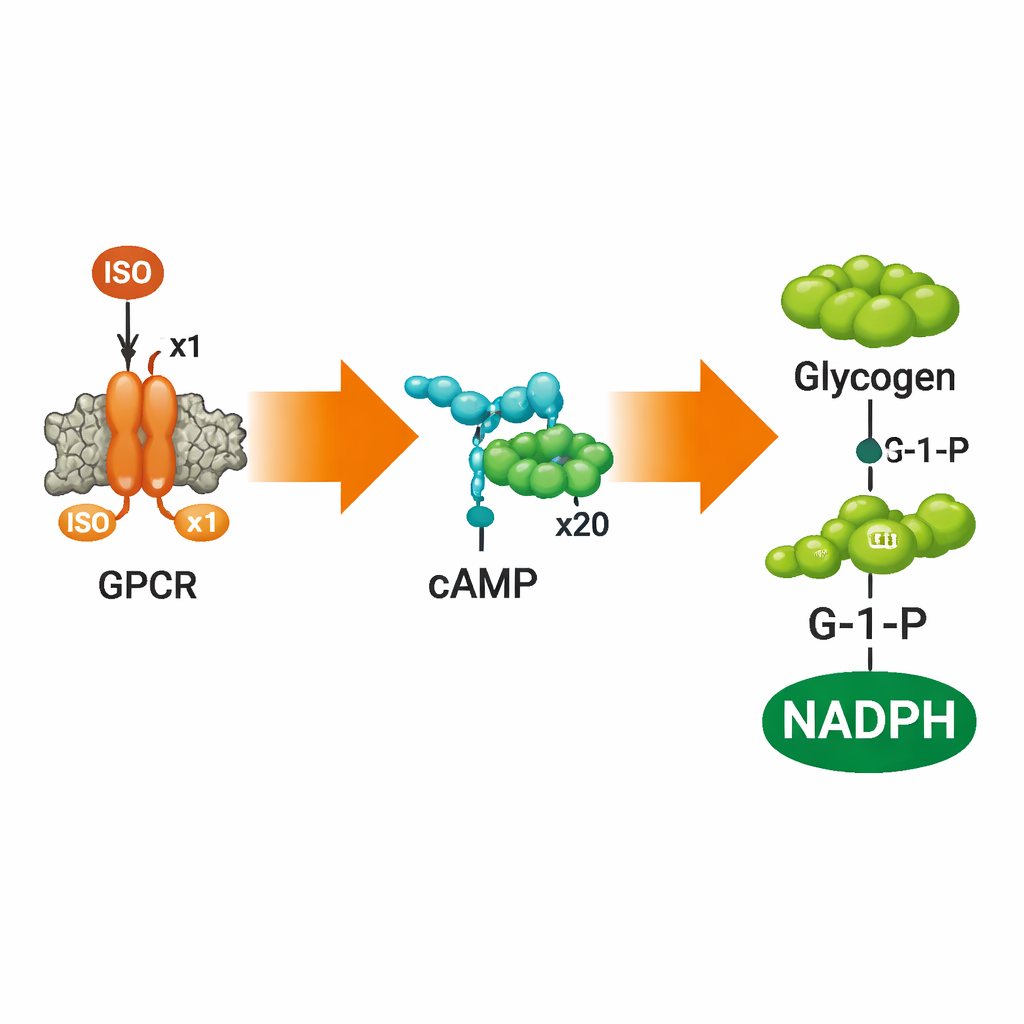
Amplifying a Whisper into a Shout
A key finding is how strongly the internal response was amplified. A modest amount of ISO outside the vesicles produced roughly 22 times more cAMP molecules than there were ISO molecules, and this amplification grew further down the pathway. By the time the signal had driven glycogen breakdown and conversion to 6‑phosphogluconolactone with accompanying NADPH, the overall amplification exceeded one hundredfold. This stepwise boost is a hallmark of natural hormone signaling and shows that the artificial system does more than just detect ISO—it processes and amplifies the message into a robust metabolic output.
Why This Matters for Future Synthetic Life
To a non‑specialist, the technical details boil down to a simple idea: the researchers have taught a minimal, man‑made cell to listen and react like a living cell. An outside drug molecule docks on a realistic human receptor, a signal is passed across a synthetic membrane, and an internal enzyme network responds by mobilizing stored chemical energy. Demonstrating this full chain—from receptor activation to controlled metabolism—in a stripped‑down artificial system is a major step toward autonomous synthetic cells that can sense, decide, and act in useful ways, such as adjusting their own energy supply or delivering therapies only when they detect the right chemical cues.
Citation: Liu, Y., Zhao, W., Zhao, Y. et al. An artificial cell capable of signal transduction mediated by ADRB2 for the regulation of glycogenolysis. Nat Commun 17, 1795 (2026). https://doi.org/10.1038/s41467-026-68503-3
Keywords: artificial cells, signal transduction, GPCR, glycogenolysis, synthetic biology