Clear Sky Science · en
Spatiotemporally controlled drug release via a click-release system utilizing mono-alkyl-hydroxylamine and cyclooctyne chemistry
Turning Powerful Medicines On Only Where They Are Needed
Many modern drugs are extremely potent, which is good for fighting cancer or relieving pain—but risky for the rest of the body. This study describes a chemistry-based “on–off switch” that lets scientists park medicines in an inactive, harmless form and then switch them on only at a chosen place and time, such as inside a tumor or in a small area that needs local anesthesia.
A New Kind of Chemical Switch
The researchers build on the concept of “click chemistry,” a family of reactions prized for being fast, precise, and able to run safely inside living organisms. Classic click reactions permanently fasten two molecules together. Here, the team flips that logic: their click reaction is directly tied to breaking a bond and releasing a drug or signal molecule. The key players are a ring-shaped molecule called cyclooctyne and a small partner called mono-alkyl-hydroxylamine. When these two meet, they snap together rapidly and then rearrange themselves so that a linked “payload” is chopped free. By attaching different drugs or fluorescent dyes as the payload, the same basic reaction can be reused for many purposes. 
Fast, Clean Release Inside and Outside Cells
To make sure this switch would be practical, the team measured how quickly the reaction proceeds and how completely it frees the attached payload. They found that carefully chosen mono-alkyl-hydroxylamines react far more quickly than simple hydroxylamine, reaching near-complete conversion within minutes to hours, depending on the structure. Importantly, the chemical steps that actually liberate the payload are so fast and selective that almost all of the attached molecule is released, with hardly any waste byproducts. The authors tested a range of cargos, including fluorescent dyes and even a fluoride ion, and showed that the system can break several common bond types used in drug design, such as carbamates and ethers.
Lighting Up Cells and Activating Cancer Drugs on Demand
Next, the researchers moved into living cells. They first hid bright fluorescent dyes behind the chemical switch and added these “caged” probes to cancer cells. On their own, the probes stayed dark, showing that the cells did not accidentally activate them. When the mono-alkyl-hydroxylamine partner was added, the dyes were released and the cells lit up in blue, green, or near-infrared colors, confirming that the reaction works reliably inside a biological environment. They then applied the same idea to doxorubicin, a widely used chemotherapy drug. In its caged form, the doxorubicin prodrug was much less toxic to cells. But when the click partner was present, the active drug was efficiently released, restoring its ability to kill cancer cells almost as strongly as free doxorubicin.
Building Smart Triggers for Place and Time
One limitation of many existing click-based systems is that the reactive parts are always “on,” making it hard to control exactly where and when they fire. To solve this, the team temporarily blocked the reactivity of mono-alkyl-hydroxylamine with small protective caps that are removed only by specific triggers. They created versions that respond to high levels of glutathione (a small molecule abundant in many tumors), to enzymes, and to flashes of violet light. In cells and in tumor-bearing mice, the glutathione-responsive version stayed silent in normal conditions but turned on in the tumor’s chemically reducing environment, releasing fluorescent dyes or activating the doxorubicin prodrug right where the cancer was growing. Mice treated with the combination of prodrug plus trigger showed stronger tumor shrinkage than those given standard doxorubicin, while experiencing less drug exposure in sensitive organs like the heart.
Remote-Control Local Anesthesia With Light
To showcase timing control, the authors designed a second application: light-guided local anesthesia. They attached the pain-numbing drug tetracaine to their cyclooctyne scaffold, rendering it inactive, and mixed this prodrug with a light-sensitive version of the hydroxylamine in a temperature-responsive gel. Injected into a rat paw, the mixture did nothing until the skin was illuminated with 405-nanometer light. Illumination triggered the click-release reaction, freeing tetracaine and producing numbness comparable to that from direct tetracaine injection. By changing how long and how strongly they shone the light, the researchers could tune how long the nerve block lasted, and they were even able to trigger repeated waves of anesthesia from a single injection. 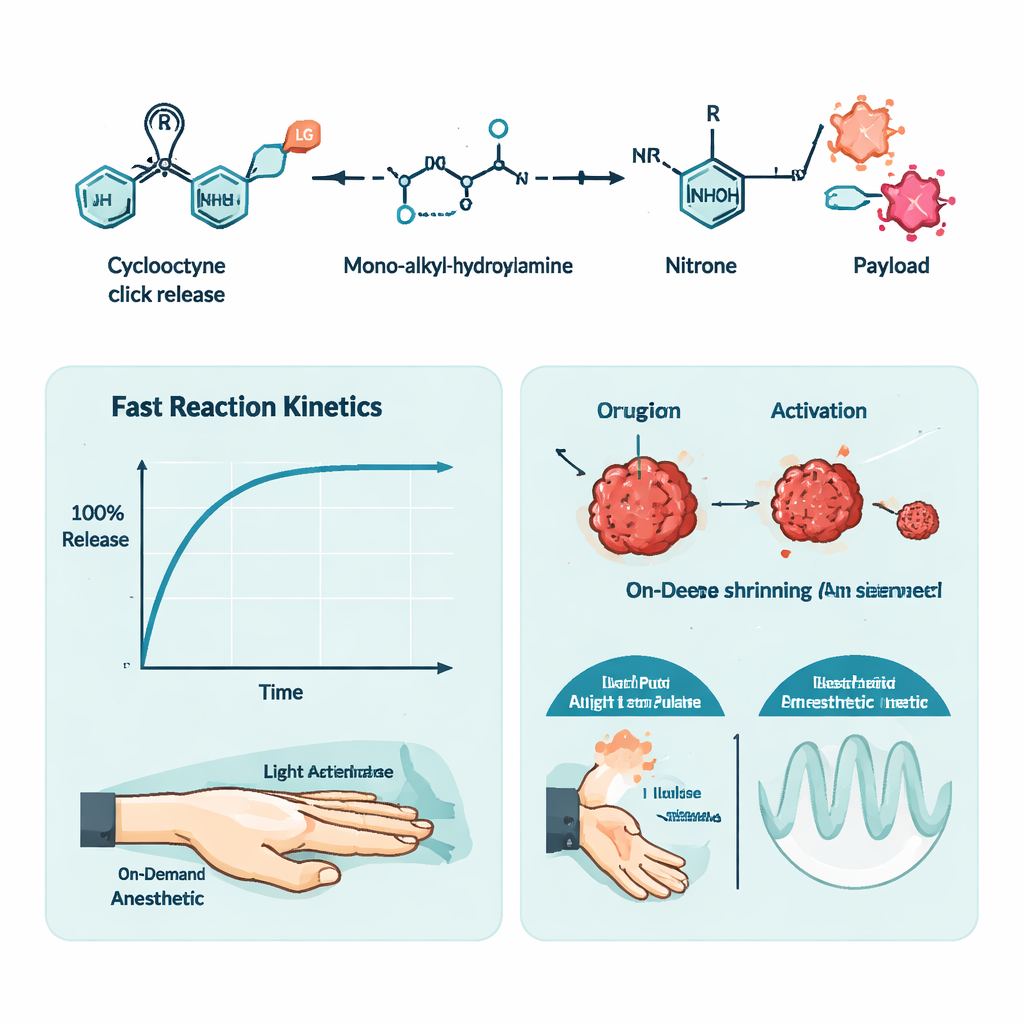
What This Could Mean for Future Treatments
For non-specialists, the key message is that this work offers a flexible chemical remote control for medicines: drugs can be delivered in a safe, quiet form and then activated only at the right spot and moment, using signals that come from the body itself or from an external light source. While more testing is needed before clinical use, the approach could eventually make cancer treatments more targeted and less toxic, enable customizable pain control after surgery, and support a range of other therapies that benefit from precise spatiotemporal control.
Citation: Xu, X., Tong, X., Shi, Y. et al. Spatiotemporally controlled drug release via a click-release system utilizing mono-alkyl-hydroxylamine and cyclooctyne chemistry. Nat Commun 17, 1794 (2026). https://doi.org/10.1038/s41467-026-68502-4
Keywords: click chemistry, targeted drug delivery, prodrugs, tumor microenvironment, photo-controlled anesthesia