Clear Sky Science · en
An ultra-long-acting dimeric bictegravir prodrug defined by a short pharmacokinetic tail
Why a twice‑yearly HIV shot matters
For people living with HIV, staying healthy still depends on taking pills every single day. Missing doses can lead to the virus rebounding and becoming resistant to treatment. This study explores a new kind of long‑lasting injection based on the drug bictegravir that could keep protective drug levels in the body for six months after a single shot, while avoiding some of the safety concerns seen with other long‑acting medicines.
Turning a daily pill into a long‑lasting shot
Bictegravir is a powerful HIV medicine usually taken once a day as part of a pill combination. The researchers set out to redesign it so that it could be given just a few times a year by injection. They chemically attached fatty “tails” to bictegravir, creating several new versions called prodrugs. These prodrugs are inactive until the body slowly clips off the added pieces, releasing normal bictegravir. By choosing the right fatty tail and packing the prodrugs into tiny solid particles suspended in water, the team aimed to create a small injection depot in muscle that would dissolve gradually over many months.
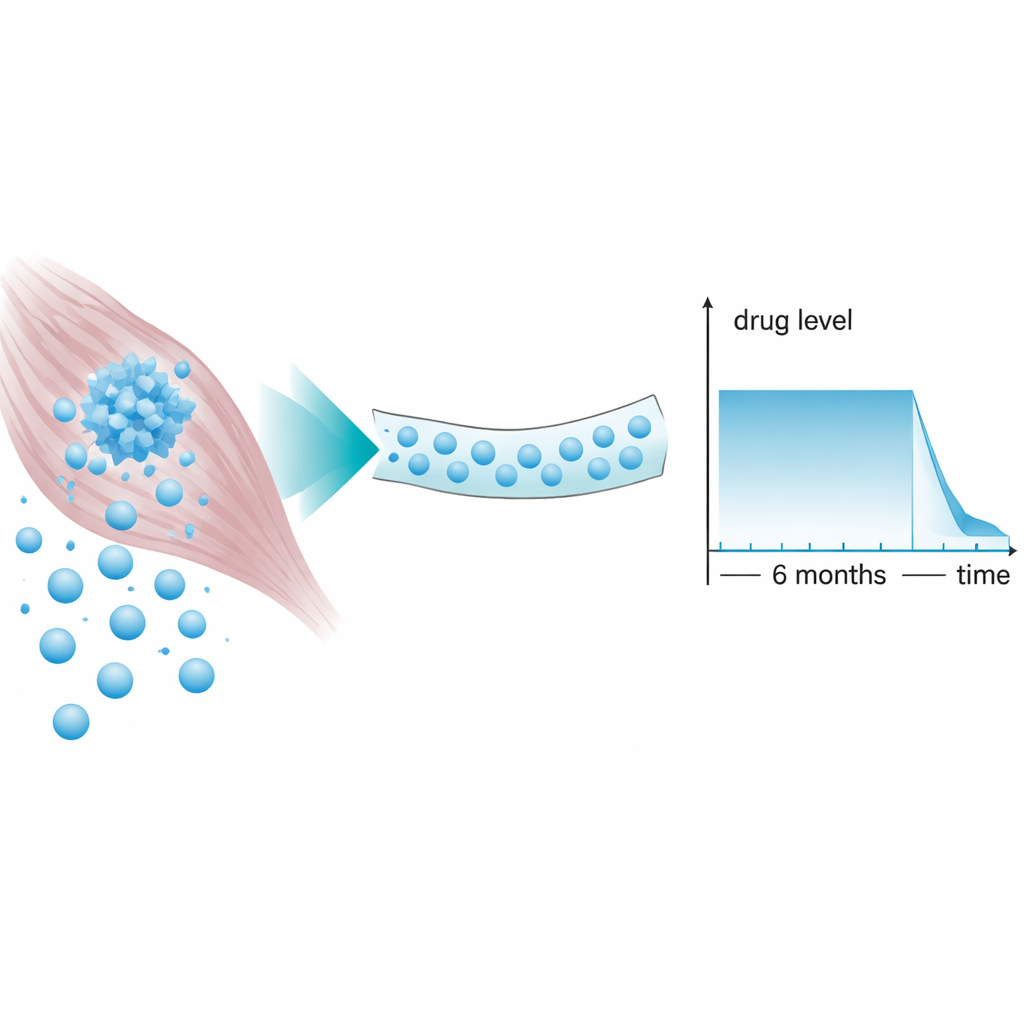
Finding the best slow‑release design
The scientists compared several prodrug designs in mice and rats. One version, a single bictegravir molecule with a fatty chain, released the drug more slowly than regular bictegravir and kept blood levels above the amount needed to control HIV for nearly a year after one injection, though at modest concentrations. The standout candidate, however, was a dimeric form called NMXBIC, in which two bictegravir molecules are linked by a longer fatty spacer. As a nanosuspension, NMXBIC produced a remarkably flat "plateau" of drug levels in the bloodstream for six months at more than 16 times the concentration needed to block the virus, followed by a rapid drop‑off rather than a long, low‑level tail.
Why a short “tail” is so important
Existing long‑acting HIV injections, such as cabotegravir, can leave low amounts of drug in the body for many months after the last dose. During this tail phase, the virus may see just enough drug to evolve resistance but not enough to be fully suppressed. The NMXBIC design tackles this by combining strong, steady exposure during the dosing period with a comparatively sharp decline afterward. In animals, drug levels stayed high for about half a year and then fell quickly, shrinking the window of weak exposure that could foster resistant virus if someone misses a follow‑up injection.
Safety, tissue targeting, and real‑world promise
Beyond blood levels, the team showed that NMXBIC and its monomeric cousin concentrated in key tissues where HIV hides, such as lymph nodes and the gut, and that immune cells readily took up the nanoparticles and slowly released active drug. In rabbits given repeated injections at human‑scale volumes, the muscle and skin reactions were limited to the kind of localized inflammation expected from any depot shot, with no serious tissue damage. In a mouse model with transplanted human immune cells, higher doses of the dimeric prodrug alone were able to suppress HIV replication after strong viral challenges, highlighting its antiviral potential.
What this could mean for people with HIV
Viewed in simple terms, this work turns one daily HIV pill ingredient into an “every six months” shot that keeps drug levels high enough to control the virus while minimizing the risky low‑level tail. Because bictegravir already has a strong track record and a high barrier to resistance, a twice‑yearly NMXBIC injection could one day be paired with another ultra‑long‑acting drug, such as lenacapavir, to create a fully injectable maintenance regimen. If future human trials confirm the animal results, many people with HIV might be able to swap 365 days of pills for two discreet clinic visits a year, making it far easier to stay on effective treatment and reducing chances for the virus to bounce back or spread.
Citation: Nayan, M.U., Sillman, B., Das, S. et al. An ultra-long-acting dimeric bictegravir prodrug defined by a short pharmacokinetic tail. Nat Commun 17, 1793 (2026). https://doi.org/10.1038/s41467-026-68501-5
Keywords: HIV treatment, long-acting injection, bictegravir, drug delivery, antiretroviral therapy