Clear Sky Science · en
Customized composition of lithium metal solid-electrolyte interphase by electric field modulation of anion motion direction
Safer, Longer-Lasting Batteries for Everyday Devices
Modern life runs on rechargeable batteries, from smartphones to electric cars. But today’s lithium-ion batteries are brushing up against their energy limits and still rely on flammable liquids that can fuel fires if something goes wrong. This study explores a new, non-flammable battery liquid that not only makes high-energy lithium-metal batteries safer, but also helps them last far longer, even under demanding conditions.
A New Liquid Built to Resist Fire
The researchers start with a special solvent called triethyl phosphate, which is naturally difficult to ignite. On its own, however, this solvent does not work well with the highly reactive lithium metal that can unlock much higher energy than today’s graphite anodes. When the usual battery liquids meet lithium metal, they tend to break down and form a fragile surface layer, leading to poor lifetime and safety issues. To fix this, the team adds a carefully chosen mix of three lithium salts to the non-flammable solvent, creating an electrolyte that can both move charge efficiently and build a strong, protective skin on the lithium metal.
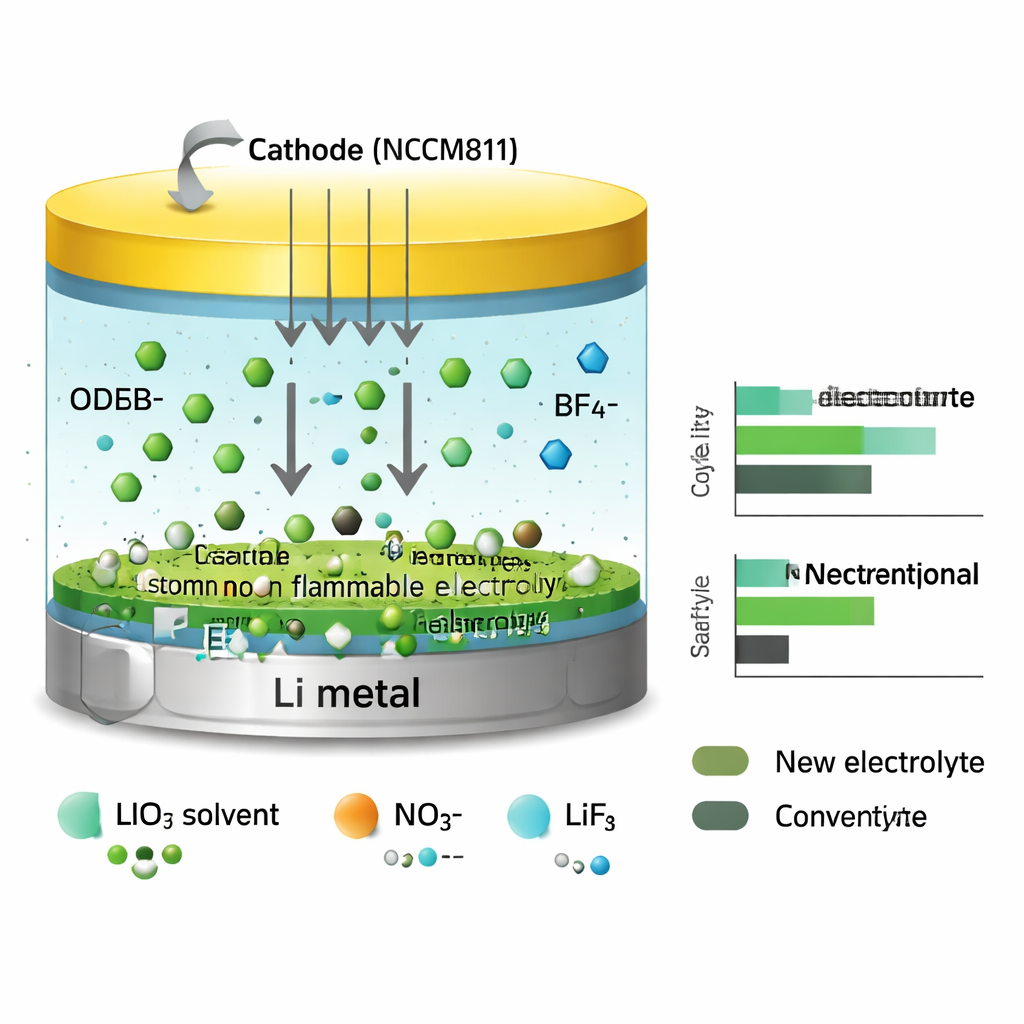
Guiding Ions with an Electric Field
At the heart of the design is how different negatively charged ions (anions) in the liquid interact with lithium ions under an electric field when the battery is working. Using computer simulations, the authors show that two anions (from lithium oxide difluoroborate and lithium nitrate) cling tightly to lithium ions. As lithium ions move toward the metal surface during charging, these anions are dragged along and crowd near the lithium. A third anion (from lithium tetrafluoroborate) binds more weakly, so it stays further away and moves more freely in the liquid. This uneven behavior means the salts do not all decompose in the same place: the strongly bound anions break down right at the lithium surface, while the weaker one reacts mainly farther out.
Building a Smart Protective Skin
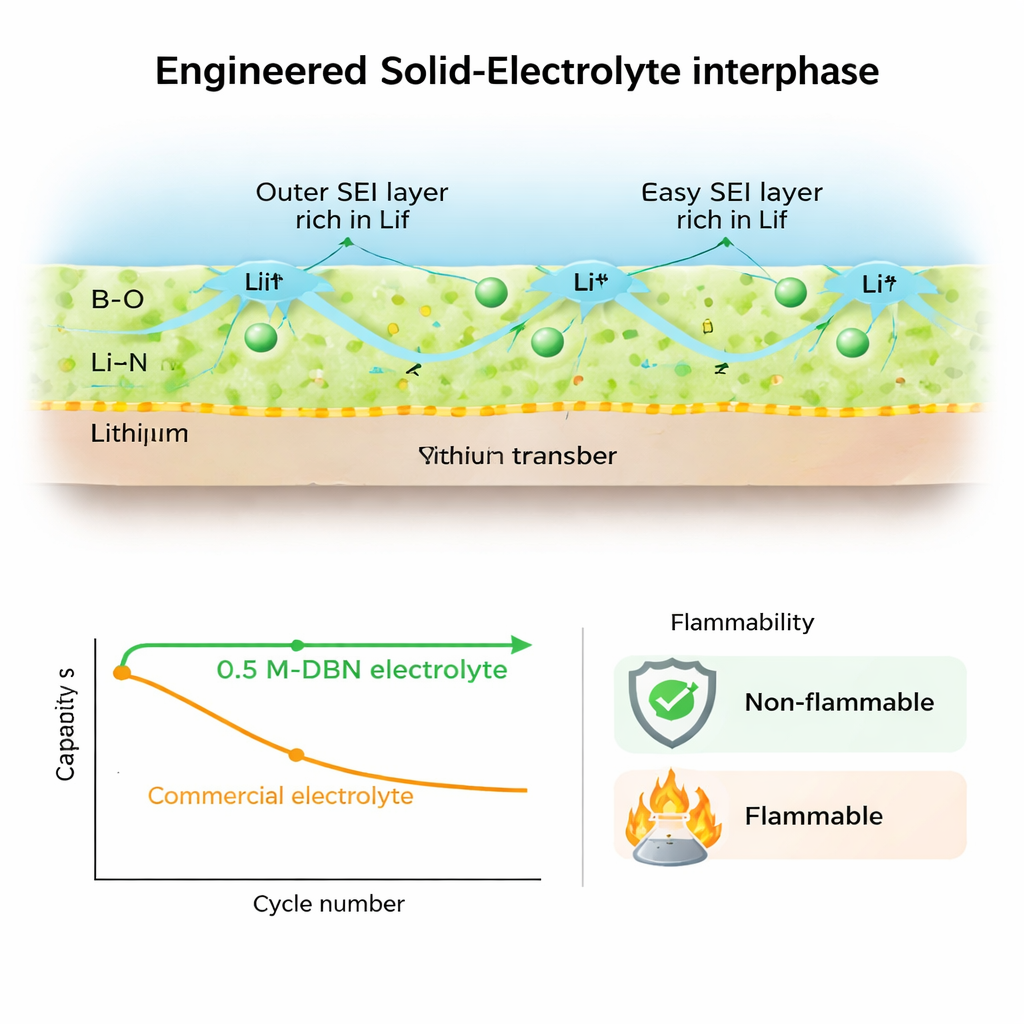
This controlled decomposition builds a “smart” solid layer, called the solid-electrolyte interphase (SEI), with a deliberate structure. Close to the lithium metal, the layer is rich in compounds containing boron and nitrogen, which form a flexible, glass-like matrix and a highly conductive lithium nitride phase. These inner components help lithium ions move quickly and evenly, reducing the chance of sharp, needle-like growths called dendrites that can pierce the separator. In the outer region, the breakdown of the fluorine-containing salt produces a shell rich in lithium fluoride, a hard, stable compound that stiffens the surface and further discourages dendrite growth. Experiments using advanced microscopes and surface probes confirm this inner–outer layering and show that the new SEI is both mechanically strong and highly conductive.
Stronger Performance at Both Battery Electrodes
The benefits of the tailored electrolyte appear on both sides of the battery. On the lithium-metal side, test cells show much smoother, denser lithium deposits and far fewer dendrites than those using a standard, flammable carbonate liquid. The cells cycle for over 1000 hours in simple lithium metal tests and maintain high efficiency when lithium is repeatedly plated and stripped. On the positive side, the team pairs the electrolyte with a high-energy cathode material called NCM811, common in advanced electric-vehicle cells. At high voltages where many electrolytes fail, the new liquid forms a thin, mostly inorganic protective film on the cathode surface. This film reduces unwanted side reactions, keeps metal atoms in the cathode from dissolving into the liquid, and helps preserve the cathode’s crystal structure during repeated charging and discharging.
High Energy, Long Life, and Improved Safety
Put together, these effects yield a high-performance lithium-metal battery that is also safer. Full cells using the new electrolyte can run at a high cut-off voltage of 4.5 V for 600 cycles while retaining about 90% of their capacity at room temperature, and over 80% at 60 °C—figures that are far better than those of cells with conventional liquids. A practical pouch cell with a realistic, high-loading cathode delivers a specific energy around 430 Wh per kilogram of total cell mass and still keeps most of its capacity after dozens of cycles. Heat and flame tests show that the non-flammable electrolyte greatly reduces the energy released during overheating and resists ignition compared to commercial formulations. In simple terms, the study demonstrates that by carefully steering how different ions move and break down in an electric field, it is possible to build a safer, non-flammable liquid that protects both electrodes, enabling high-energy lithium-metal batteries that last longer and pose less fire risk.
Citation: Xu, S., Zheng, L., Guo, X. et al. Customized composition of lithium metal solid-electrolyte interphase by electric field modulation of anion motion direction. Nat Commun 17, 1790 (2026). https://doi.org/10.1038/s41467-026-68498-x
Keywords: lithium metal batteries, non-flammable electrolyte, solid electrolyte interphase, high-voltage cathodes, battery safety