Clear Sky Science · en
Selective electrosynthesis of urea from nitrate and carbon dioxide with low overpotential
Turning Waste into Fertilizer
Modern farming depends heavily on urea fertilizer, but making it the conventional way burns a lot of fossil fuel and releases large amounts of carbon dioxide. This study explores a cleaner route: using electricity and specially designed materials to turn carbon dioxide and nitrate—both common pollutants—directly into urea under mild conditions. If scaled up, such a process could help feed people while cutting greenhouse gas emissions and making better use of waste streams.
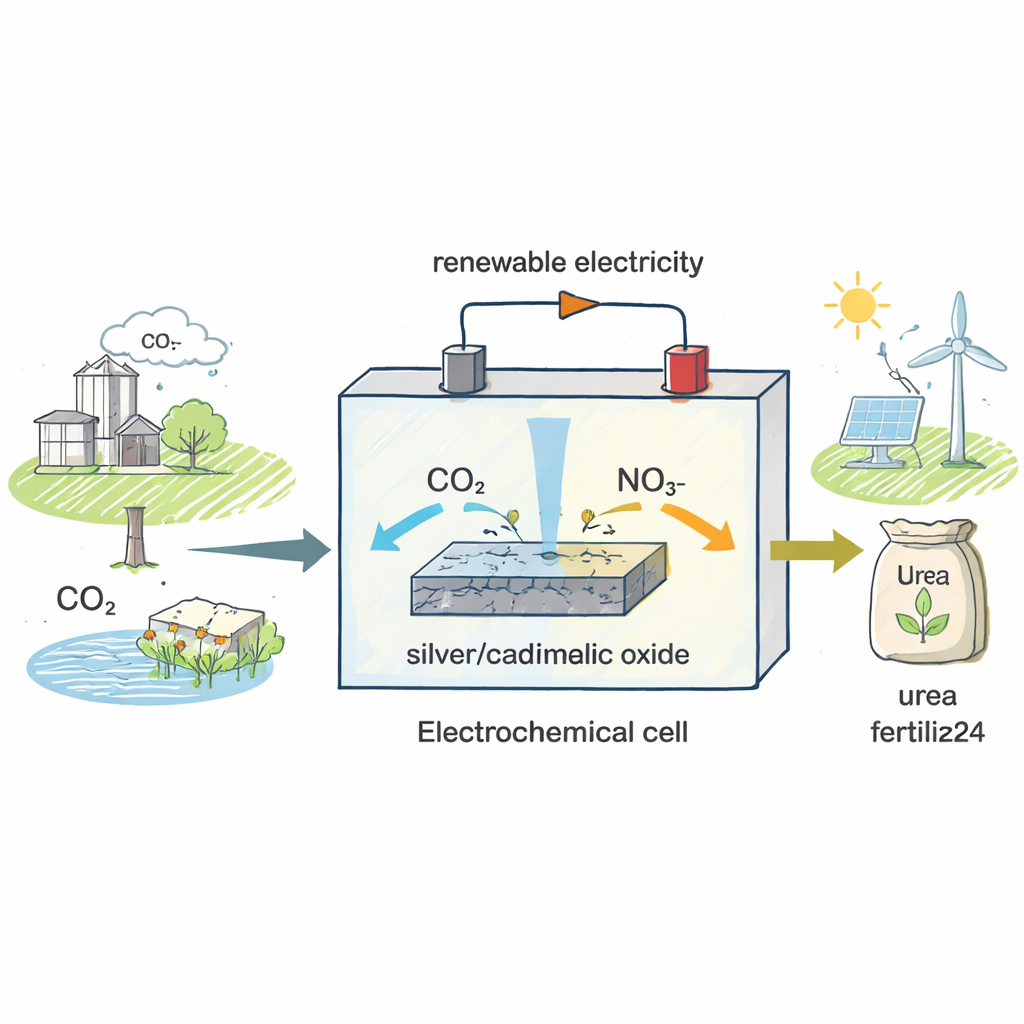
A New Way to Make an Old Fertilizer
Today’s urea factories run at high temperatures and pressures and rely on ammonia first being made from nitrogen gas, a step that alone consumes over 2% of global energy. The authors set out to bypass this energy-hungry route. Instead of starting from nitrogen gas, they use nitrate, a nitrogen compound found in agricultural runoff and industrial wastewaters, and combine it with carbon dioxide, the main greenhouse gas. Both substances are fed into an electrochemical cell—a device where electricity drives chemical reactions—so that, with the right catalyst, they can be stitched together into urea at room temperature and relatively low voltages.
Designing a Helpful Surface
The heart of the device is a solid catalyst made from tiny particles that combine silver (Ag) with cadmium oxide (CdO). Microscopy and X-ray techniques show that these particles form intimate “heterostructures,” where metallic silver and CdO touch at the nanoscale and exchange electrons. This electronic interaction subtly alters how each component binds incoming molecules. Under working conditions, the CdO portion transforms into cadmium carbonate (CdCO₃) that contains missing oxygen atoms, known as oxygen vacancies. These vacant spots act like tiny docking sites that help capture and activate carbon dioxide, while nearby silver regions are especially good at grabbing nitrogen-containing species derived from nitrate.
Making Urea Efficiently and Selectively
When carbon dioxide and nitrate flow across this Ag/CdO catalyst in a lab-scale cell, the material converts them to urea at unusually low “overpotential,” meaning the extra voltage beyond the ideal thermodynamic requirement is small. In a simple H-shaped cell, the optimized composition, called Ag₀.₀₇/CdO, reaches a urea selectivity of about 50% at only −0.10 volts versus a standard reference, and produces little ammonia or carbon monoxide as by-products. In a more advanced flow cell, where gases and liquids are continuously supplied, the same catalyst achieves a high urea production rate of roughly 112 millimoles per gram of catalyst per hour at −0.15 volts. By further optimizing the reactor design and operating conditions, the team pushes the urea production rate to about 427 millimoles per gram per hour at higher currents, while maintaining performance for 1,000 hours of continuous operation—an important sign of stability.
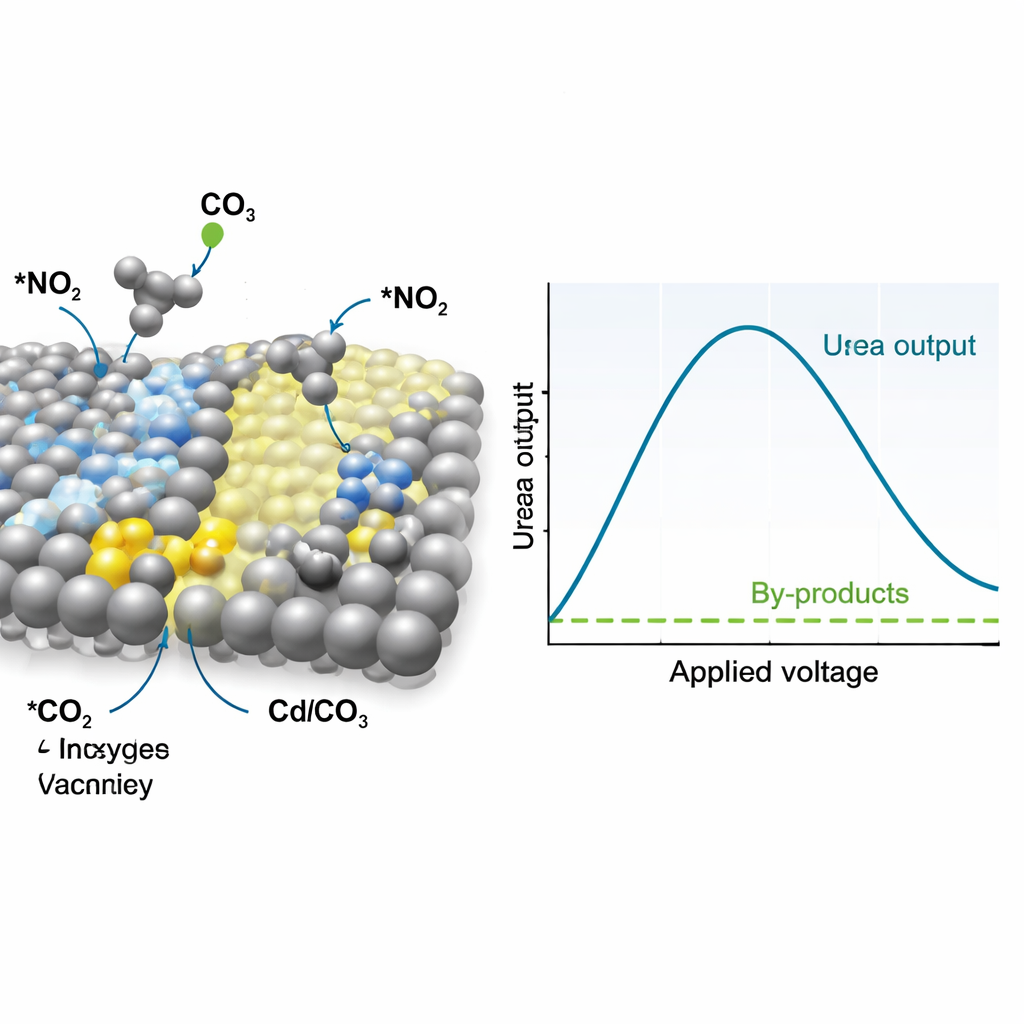
How the Catalyst Steers the Reaction
To understand why this material works so well, the researchers followed the catalyst and reaction intermediates in real time using Raman and infrared spectroscopy, along with advanced X-ray probes. These measurements indicate that silver sites preferentially convert nitrate into a reactive intermediate often described as *NO₂, while the reconstructed CdCO₃ with oxygen vacancies binds carbon dioxide and turns it into *CO, another short-lived intermediate. Signals associated with carbon–nitrogen bonds appear at the operating voltages, showing that *NO₂ and *CO couple early in the process to form a urea-like species rather than drifting off to form ammonia or carbon monoxide separately. Computer simulations support this picture, revealing that the interface between silver and vacancy-rich CdCO₃ lowers the energy barrier for this key C–N coupling step and makes it more favorable than competing reactions.
What This Could Mean for Fertilizers and Climate
Overall, the work demonstrates that carefully engineered catalyst surfaces can guide simple waste molecules like carbon dioxide and nitrate along an energy-efficient pathway to urea, under mild conditions and with long lifetimes. For a non-specialist, the key takeaway is that the team has built a kind of “electronic filter” that prefers to join carbon and nitrogen into urea rather than letting them escape as other products. While real-world deployment will require addressing issues such as material cost, reactor engineering, and sourcing cadmium safely, the study offers a blueprint for future fertilizer production that could be powered by renewable electricity and aligned with a more sustainable carbon and nitrogen cycle.
Citation: Liu, S., Wang, T., Liu, J. et al. Selective electrosynthesis of urea from nitrate and carbon dioxide with low overpotential. Nat Commun 17, 1787 (2026). https://doi.org/10.1038/s41467-026-68497-y
Keywords: urea electrosynthesis, carbon dioxide utilization, nitrate reduction, electrocatalyst design, green fertilizer