Clear Sky Science · en
Targeting the UFL1-AKT cascade suppresses triple-negative breast cancer progression
Why this research matters
Triple-negative breast cancer (TNBC) is one of the deadliest forms of breast cancer because it lacks the hormone and growth-factor switches that many modern drugs target. Patients often receive only traditional chemotherapy, which can stop working as tumors become resistant. This study uncovers a previously hidden molecular “engine” that helps TNBC cells grow and shrug off chemotherapy, and it proposes a new type of drug that directly jams this engine.
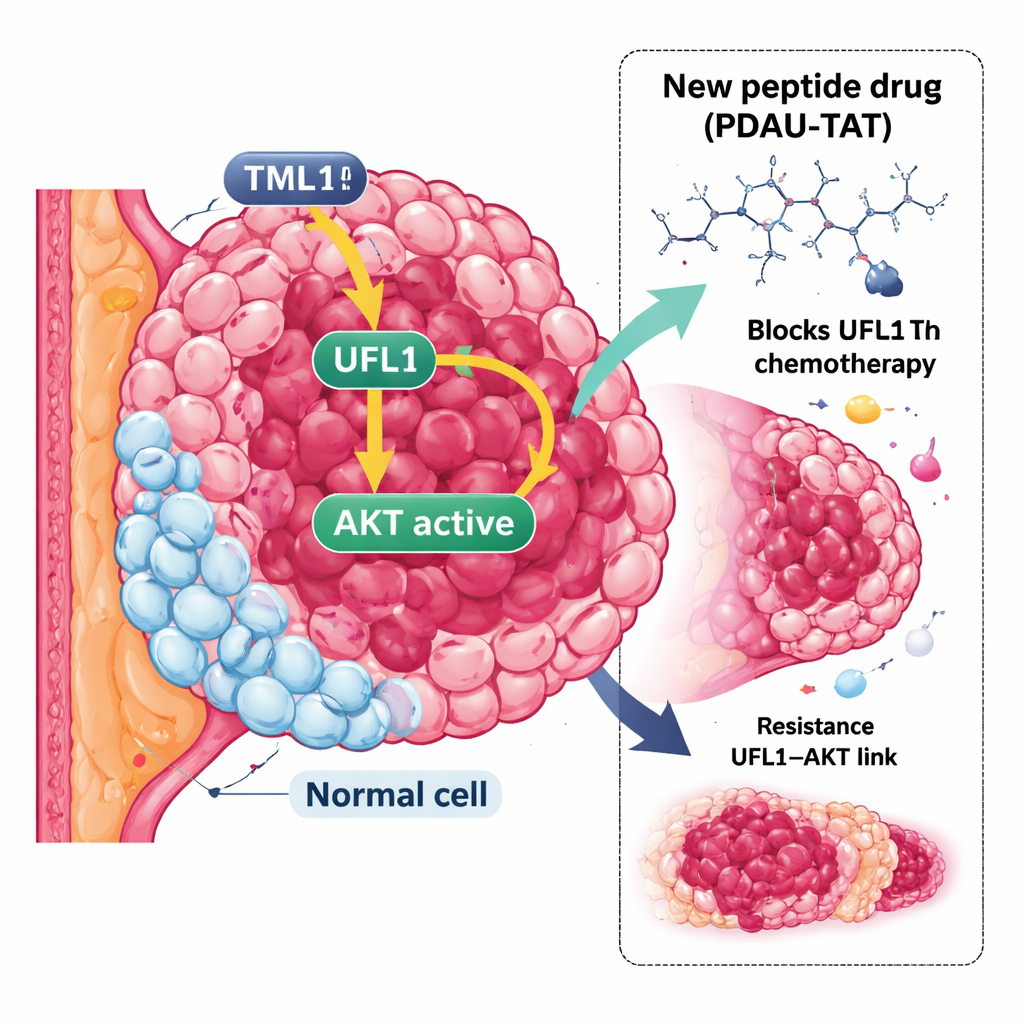
A stubborn form of breast cancer
TNBC is defined by the absence of three common markers: the estrogen receptor, the progesterone receptor and the HER2 growth receptor. Without these markers, many successful targeted therapies simply do not apply. Standard drugs such as cisplatin and doxorubicin are used instead, but TNBC tumors often resist or quickly adapt to these treatments and are more likely to spread. This creates an urgent need for fresh strategies that go beyond the usual suspects and look for other weaknesses in the tumor’s control systems.
A little-known protein with a big impact
The researchers focused on UFL1, a protein that attaches a small tag, called UFM1, onto other proteins in a process known as UFMylation. This tagging system has only recently been discovered, and its role in cancer has been unclear and sometimes even contradictory. By analyzing patient data and tumor samples, the team showed that UFL1 is markedly elevated in TNBC compared with normal breast tissue. When they reduced UFL1 levels in TNBC cell lines and in mouse tumor models, cancer cells multiplied more slowly and became far more sensitive to chemotherapy, suggesting that UFL1 plays a tumor-promoting role in this context.
Connecting UFL1 to a master growth switch
Digging deeper, the scientists discovered that UFL1 directly binds to AKT, a central signaling protein that drives cell growth, survival and metabolism and is often hyperactive in cancers. In TNBC, AKT is frequently overactive even when the usual upstream mutations are missing. The team found that UFL1 chemically tags AKT at three specific positions, which in turn makes it easier for AKT to be switched on by its activating partners, while shielding it from enzymes that would normally turn it off. Cells lacking UFL1 or expressing an engineered form of AKT that cannot receive these tags showed much lower AKT activity, slower growth and greater vulnerability to chemotherapy in both cell cultures and animal models.
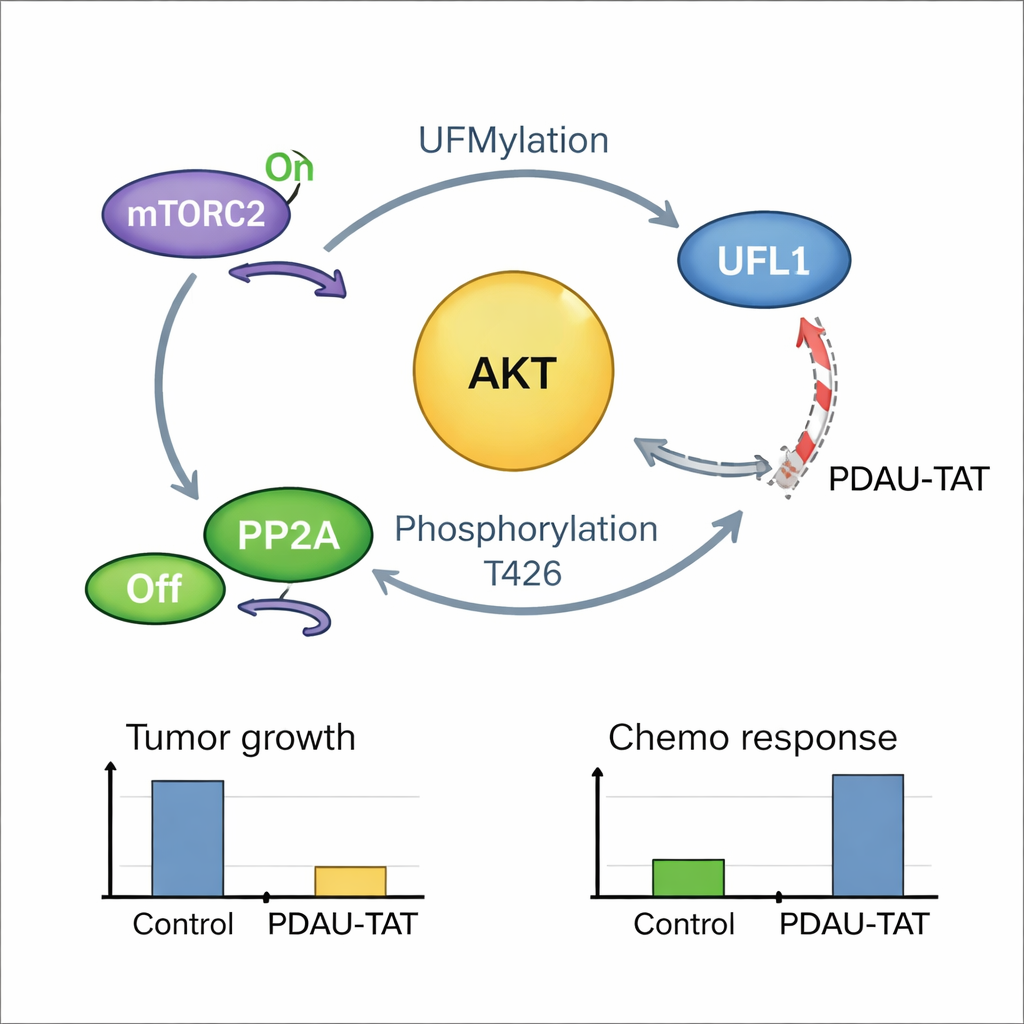
A self-reinforcing cancer loop and a way to break it
The story turned out to be even more tightly linked: once AKT is activated, it feeds back and modifies UFL1 at a single crucial site, boosting UFL1’s tagging activity on AKT and other targets. This creates a positive feedback loop in which UFL1 activates AKT, and active AKT, in turn, supercharges UFL1. Patient tumor samples showed that high levels of modified UFL1 strongly correlate with high levels of active AKT, supporting the existence of this loop in human disease. To exploit this vulnerability, the team designed a short, cell-penetrating peptide, called PDAU-TAT, that mimics a small region of UFL1 and wedges itself between UFL1 and AKT. This peptide disrupts their interaction, cuts AKT activation, slows tumor growth and makes TNBC cells and patient-derived tumors more responsive to chemotherapy, all without obvious damage to major organs in mice.
What this could mean for patients
In simple terms, the researchers have uncovered a hidden wiring diagram inside TNBC cells: UFL1 and AKT are locked in a self-amplifying loop that keeps growth signals stuck in the “on” position and helps tumors resist treatment. By building a small peptide that slips into this loop and disconnects UFL1 from AKT, they show a plausible new way to weaken TNBC tumors while sparing normal cells, which tend to have lower UFL1 levels. Although this work is still at the preclinical stage, it points toward a new class of targeted therapies aimed not at the usual receptors but at the protein–protein connections that power some of the most aggressive breast cancers.
Citation: Yang, X., Wen, Y., Ma, X. et al. Targeting the UFL1-AKT cascade suppresses triple-negative breast cancer progression. Nat Commun 17, 613 (2026). https://doi.org/10.1038/s41467-026-68493-2
Keywords: triple-negative breast cancer, AKT signaling, UFMylation, UFL1, peptide cancer therapy