Clear Sky Science · en
The UFL1-AKT positive feedback loop promotes breast cancer progression by enhancing lipid synthesis
Why cancer cells care so much about fat
Breast cancer cells, like all fast-growing cells, need a steady supply of building blocks to make new membranes and signaling molecules. Fats, or lipids, are a key part of that supply. This study uncovers how a little-known protein system shifts cancer cells into "fat‑production overdrive," helping tumors grow and evade treatment. Understanding this hidden fuel line could open new routes to slow or starve breast cancers.
A hidden protein tag with big consequences
Our cells constantly adjust the behavior of their proteins using tiny chemical tags. One such tag, called UFM1, is added by an enzyme named UFL1. UFM1 tagging (UFMylation) has been linked to DNA repair and stress responses, but its role in cancer was unclear. The authors show that UFL1 is much more active in human breast tumors than in normal breast tissue, across all major subtypes. Patients whose tumors carry higher UFL1 levels tend to have worse survival. When researchers reduced UFL1 in breast cancer cells or in mouse tumors, cancer growth slowed, cell division dropped, and cell death rose, suggesting that UFL1 behaves like a cancer‑promoting factor.
How a signaling hub is flipped into fat‑making mode
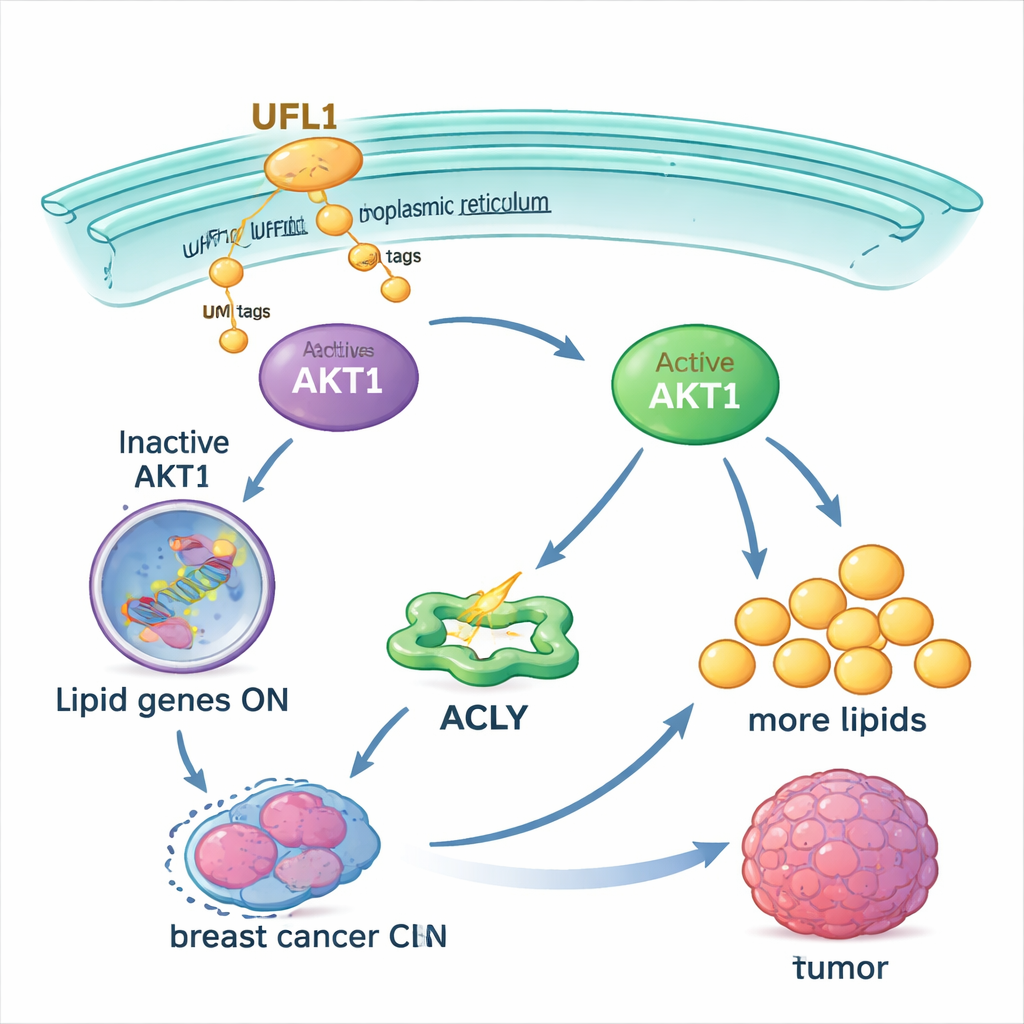
To understand how UFL1 helps tumors, the team looked for its molecular partners and found a key signal‑control protein called AKT1. AKT1 is a central switch that tells cells to grow and to make new lipids. The study shows that UFL1 physically binds to AKT1 and decorates it with UFM1 tags at several specific spots. This tagging changes AKT1’s shape, loosening an internal "brake" that normally keeps it quiet. It also helps AKT1 move to a membrane compartment inside the cell, the endoplasmic reticulum, where other enzymes can fully activate it by adding phosphate groups. Without UFM1 tags at those sites, AKT1 is much harder to activate.
Turning on the cancer fat factory
Once activated, AKT1 pushes cells to ramp up their fat production. It boosts the activity of a key metabolic enzyme, ACLY, and increases levels of master regulators like SREBP1 that turn on many fat‑making genes. In breast cancer cells grown in the lab, UFL1 made cells accumulate more fat droplets, free fatty acids, and cholesterol. Removing UFL1 sharply reduced these lipid stores, both in dishes and in tumors grown in mice. Supplying extra fatty acids from outside the cell could largely rescue the growth of UFL1‑deficient cancer cells, indicating that the main effect of UFL1 is to secure enough lipids to sustain tumor expansion.
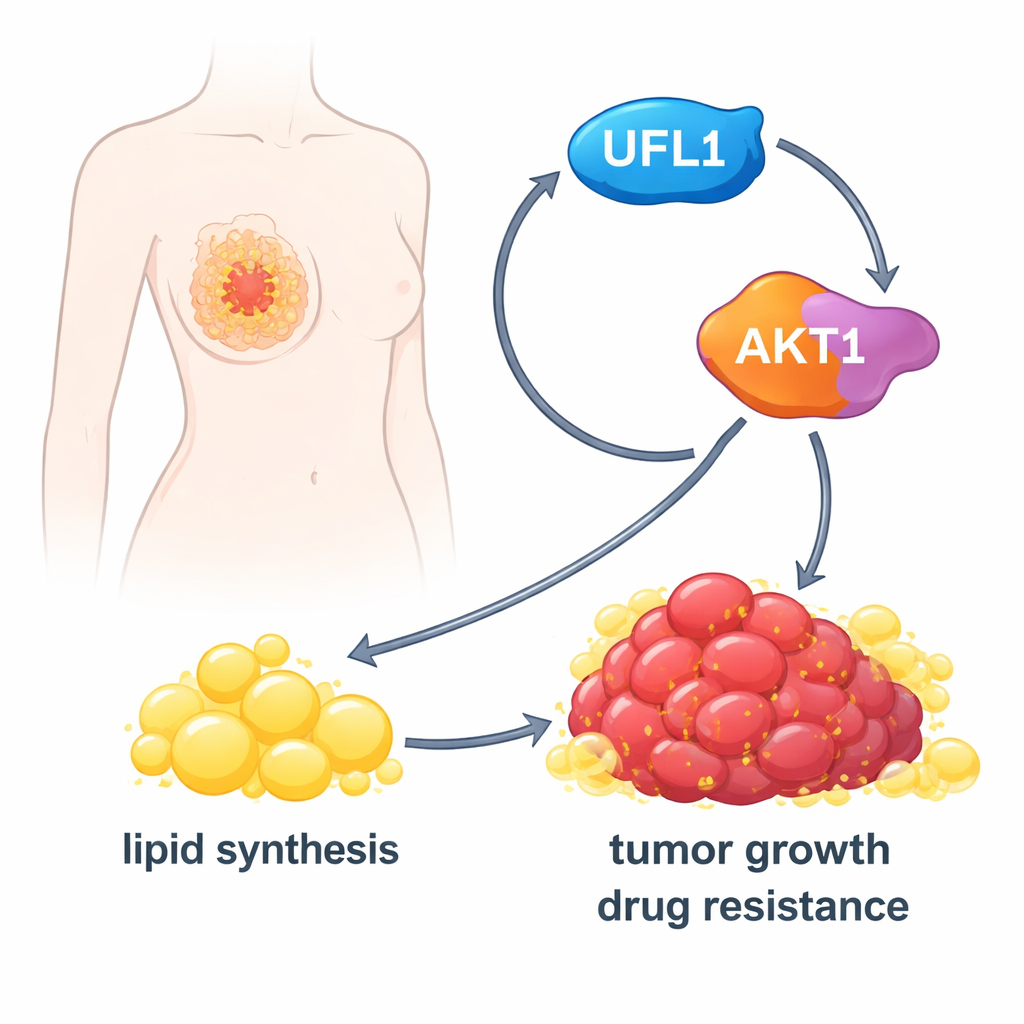
A self‑amplifying loop that drives tumor growth
Surprisingly, the relationship between UFL1 and AKT1 runs both ways. The researchers discovered that once AKT1 is active, it in turn adds a phosphate tag to UFL1 at a particular site. This modification further boosts UFL1’s ability to tag AKT1 with UFM1, creating a positive feedback loop: UFL1 activates AKT1, and AKT1 activates UFL1. Mutating either the UFM1‑tagging sites on AKT1 or the phosphate site on UFL1 breaks this loop. In mice, tumors carrying these mutant versions grew poorly, contained fewer lipids, and showed more dying cells. In samples from patients with triple‑negative breast cancer, higher levels of activated UFL1 and activated AKT1 tended to appear together, underscoring that this loop operates in real tumors.
Weakening the loop to help treatments work
Because many drug trials already target AKT, the authors tested whether blocking UFMylation could make these treatments more effective. In breast cancer cells, a small‑molecule UFMylation inhibitor and an AKT inhibitor each lowered AKT1 activity and lipid buildup, but using them together worked much better than either alone. The combination also slowed tumor growth and cut fat content in mouse tumors without noticeable weight loss in the animals. Blocking UFMylation further made cancer cells more sensitive to standard chemotherapies such as cisplatin and etoposide, which often fail when AKT is highly active.
What this means for patients
To a lay reader, the study shows that certain breast cancers wire themselves with a self‑reinforcing loop that feeds both their growth and their fat supply. UFL1 and AKT1 act together like two pedals pressed down at once: one tags, the other signals, and together they drive lipid production and tumor expansion. By finding drugs that interfere with this loop—either by blocking UFMylation, AKT1, or their interaction—future therapies may be able to slow tumor growth and make existing treatments more effective, especially in aggressive breast cancer types.
Citation: Meng, F., Du, Y., Liang, J. et al. The UFL1-AKT positive feedback loop promotes breast cancer progression by enhancing lipid synthesis. Nat Commun 17, 614 (2026). https://doi.org/10.1038/s41467-026-68492-3
Keywords: breast cancer, lipid metabolism, AKT signaling, UFMylation, targeted therapy