Clear Sky Science · en
In situ self-assembled cell reservoir hydrogel for maneuvering multistage radioimmunotherapy
Turning Radiation into a Smarter Cancer Weapon
Radiation therapy is one of the most widely used treatments for cancer, but tumors often survive by hiding from the immune system and by growing in low-oxygen (hypoxic) conditions that blunt radiation’s impact. This study describes an injectable “smart” hydrogel that forms directly inside a tumor and acts as a tiny drug factory and oxygen generator. By feeding both radiation and carefully timed immune-stimulating signals to the tumor, the system aims to turn a hard-to-treat cancer into one the body’s own defenses can recognize and destroy.
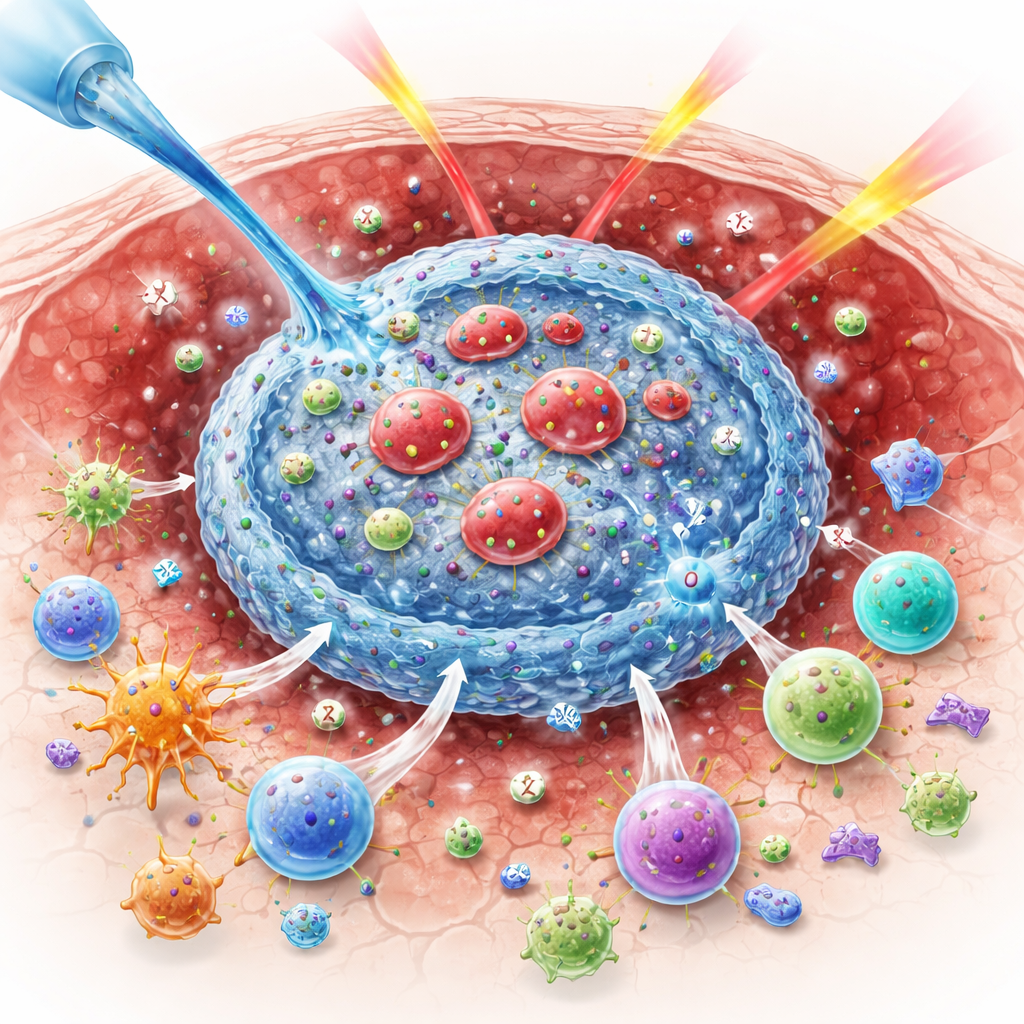
Why Oxygen Matters for Beating Tumors
Radiation kills cancer cells in part by generating highly reactive oxygen molecules that break DNA. When tumors are starved of oxygen, this chemistry is less effective, so more cancer cells survive. At the same time, radiation sends mixed messages to the immune system. It can expose tumor proteins that mark the cancer as dangerous, but it can also boost “brakes” on immune cells, such as the checkpoint protein CTLA-4, and favor regulatory T cells that suppress attack. These opposing effects help explain why combining radiation with immune drugs has not always worked as well in patients as it has in mice.
A Living Gel Built Inside the Tumor
The researchers designed a material that self-assembles only inside the acidic environment of a tumor. It is made from three key parts: a natural sugar-based polymer called alginate, an inorganic powder called barium peroxide, and red blood cells that have been gently engineered to carry two immune drugs. When the mixture is injected into a tumor, the acidic conditions trigger barium peroxide to break down, releasing barium ions and hydrogen peroxide. The barium ions instantly crosslink the alginate into a soft hydrogel, trapping the engineered red blood cells in place and preventing the drugs from washing away. At the same time, enzymes naturally present in the red blood cells convert the hydrogen peroxide into oxygen, helping to relieve tumor hypoxia and prepare the tumor for more effective radiation.
Timed Release of Dual Immune Signals
The real power of the system lies in how it staggers the release of two immune agents: an antibody that blocks CTLA-4 and a cytokine called interleukin-12 (IL-12). The antibody is loaded inside the red blood cells, while IL-12 is anchored on their surface. As oxygen is generated, pores open in the red blood cell membrane, allowing the CTLA-4–blocking antibody to escape quickly. This early burst lifts immune suppression, encourages dendritic cells to carry tumor antigens to nearby lymph nodes, and helps activate tumor-specific T cells. IL-12, by contrast, seeps out slowly as the hydrogel’s structure gradually loosens. This later signal pushes T cells and natural killer cells to produce interferon-gamma, fuels further recruitment of killer immune cells into the tumor, and reinforces a positive feedback loop between immune sentinels and effectors.
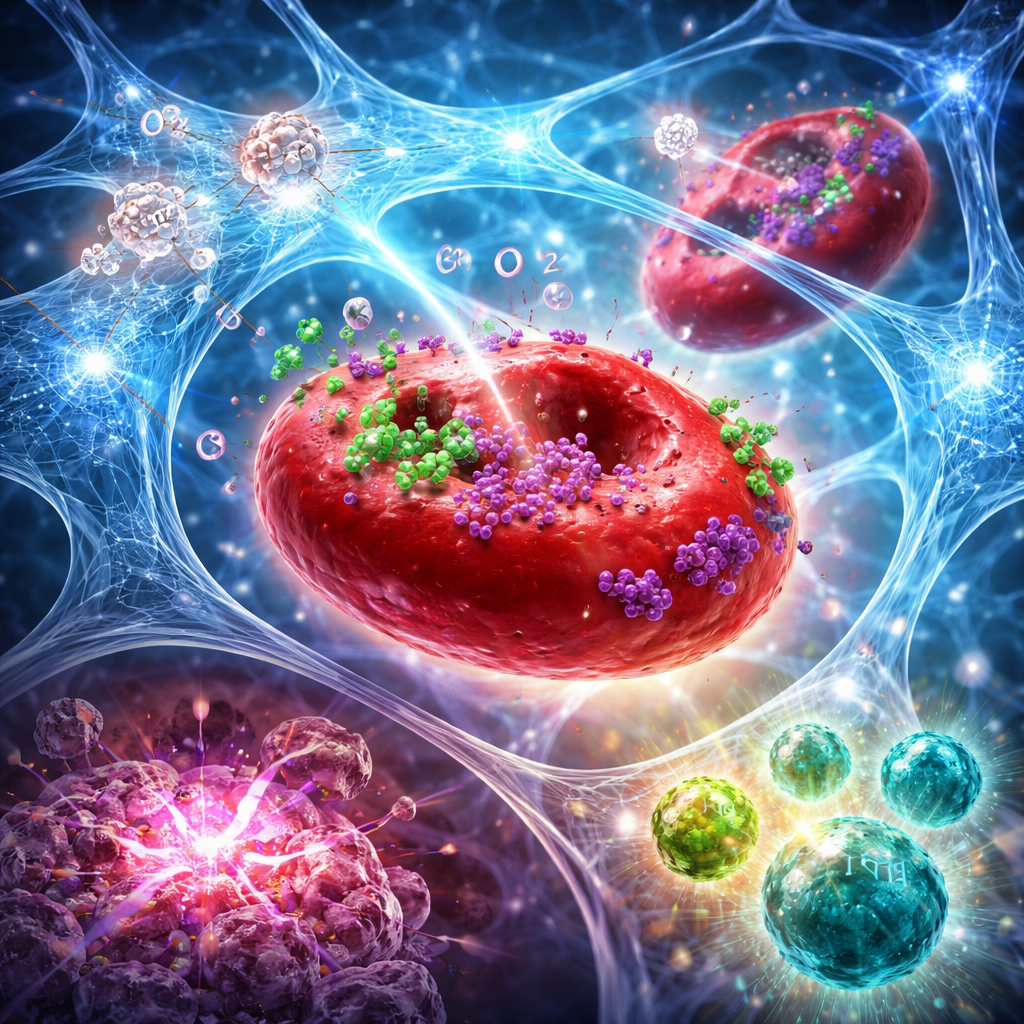
From Local Injection to Systemic Immune Attack
In mouse models of aggressive breast and pancreatic cancers, pairing this hydrogel with focused radiation did more than just shrink the treated tumor. The combined approach increased oxygen levels in tumors for days, amplified radiation-induced DNA damage, and triggered a stronger form of cell death that naturally flags tumors for immune recognition. Flow cytometry and tissue staining showed a pronounced shift in the tumor microenvironment: more cytotoxic CD8 T cells and natural killer cells, fewer regulatory T cells, and a higher ratio of “effector” to “suppressor” cells. Specialized dendritic cells in lymph nodes also increased, indicating better antigen presentation. Many mice experienced complete tumor regression, and some were protected against tumor re-growth when cancer cells were reintroduced weeks later, suggesting the formation of durable immune memory.
Safety, Practicality, and Future Directions
Because the gel forms locally and holds onto its cargo, only small amounts of IL-12 and anti-CTLA-4 leak into the bloodstream, reducing the risk of severe side effects such as cytokine storms or organ damage that have limited these agents in the past. The building blocks—alginate, red blood cells, and barium peroxide—are relatively inexpensive and scalable, raising hopes that this strategy could be adapted for different cancers. While challenges remain in manufacturing, quality control, and delivering the gel to deep or widely spread tumors, the work showcases a promising way to synchronize radiation with precisely timed immune activation, turning a physical cancer treatment into a powerful trigger for whole-body antitumor immunity.
What This Could Mean for Patients
In everyday terms, this research suggests that instead of giving radiation and immune drugs separately and hoping they cooperate, we can package them into a smart material that sits inside the tumor, makes its own oxygen, and releases immune signals in a carefully choreographed sequence. In animals, this approach not only made radiation more lethal to cancer cells but also trained the immune system to recognize and remember the tumor, helping to prevent it from coming back. If similar results can be achieved in humans, such hydrogels could one day turn hard-to-treat tumors into targets that the body’s own defenses can reliably find and destroy.
Citation: Chen, Y., Chen, Q., Ma, Y. et al. In situ self-assembled cell reservoir hydrogel for maneuvering multistage radioimmunotherapy. Nat Commun 17, 1784 (2026). https://doi.org/10.1038/s41467-026-68490-5
Keywords: radioimmunotherapy, hydrogel, cancer immunotherapy, tumor microenvironment, radiotherapy