Clear Sky Science · en
Cryo-electron tomography reveals coupled flavivirus replication, budding and maturation
How Brain-Infecting Viruses Hijack Cell Membranes
Flaviviruses, a large group of viruses that includes the agents behind dengue fever and tick-borne encephalitis, can invade the brain and cause severe disease. Yet exactly how these tiny pathogens copy themselves and assemble new viral particles inside our cells has remained largely hidden. This study uses cutting-edge 3D electron imaging at cryogenic temperatures to watch tick-borne flaviviruses at work inside human cells and mouse brain tissue, revealing an unexpectedly tight coupling between genome copying, virus assembly, and virus maturation.
Viral Factories Hidden in Cell Membranes
Flaviviruses do not simply float in the cell as they replicate. Instead, they remodel a cellular membrane system called the endoplasmic reticulum (ER) to form specialized pockets known as replication organelles. These look like tiny inward buds on the ER surface and serve as protected chambers where viral RNA genomes are copied. Using cryo-electron tomography, a technique that produces 3D snapshots of cells flash-frozen in a near-native state, the researchers visualized these buds in human lung cells infected with a tick-borne flavivirus related to tick-borne encephalitis virus. They found that the ER around infected regions swells and becomes packed with clusters of these replication organelles, some of which are empty while others are filled with dense, thread-like RNA.
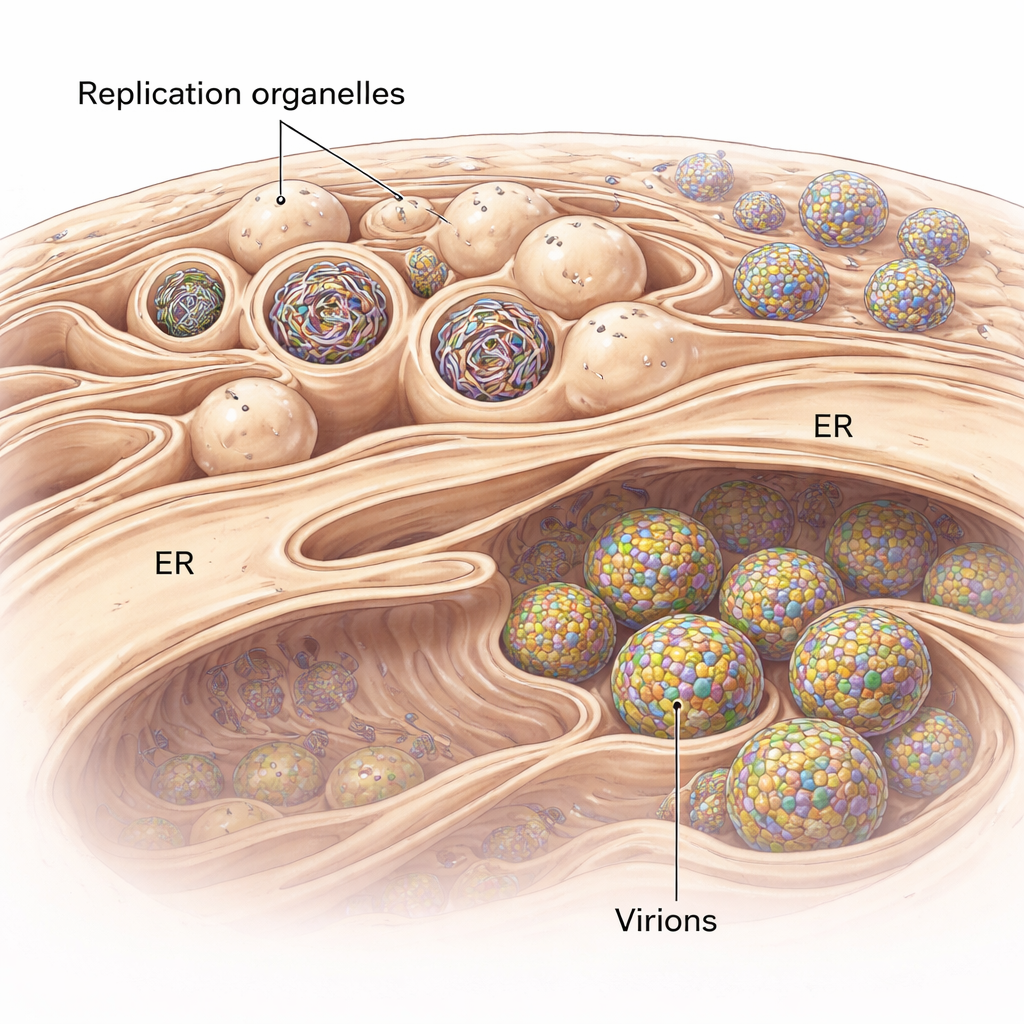
Building the Perfect Viral Bubble
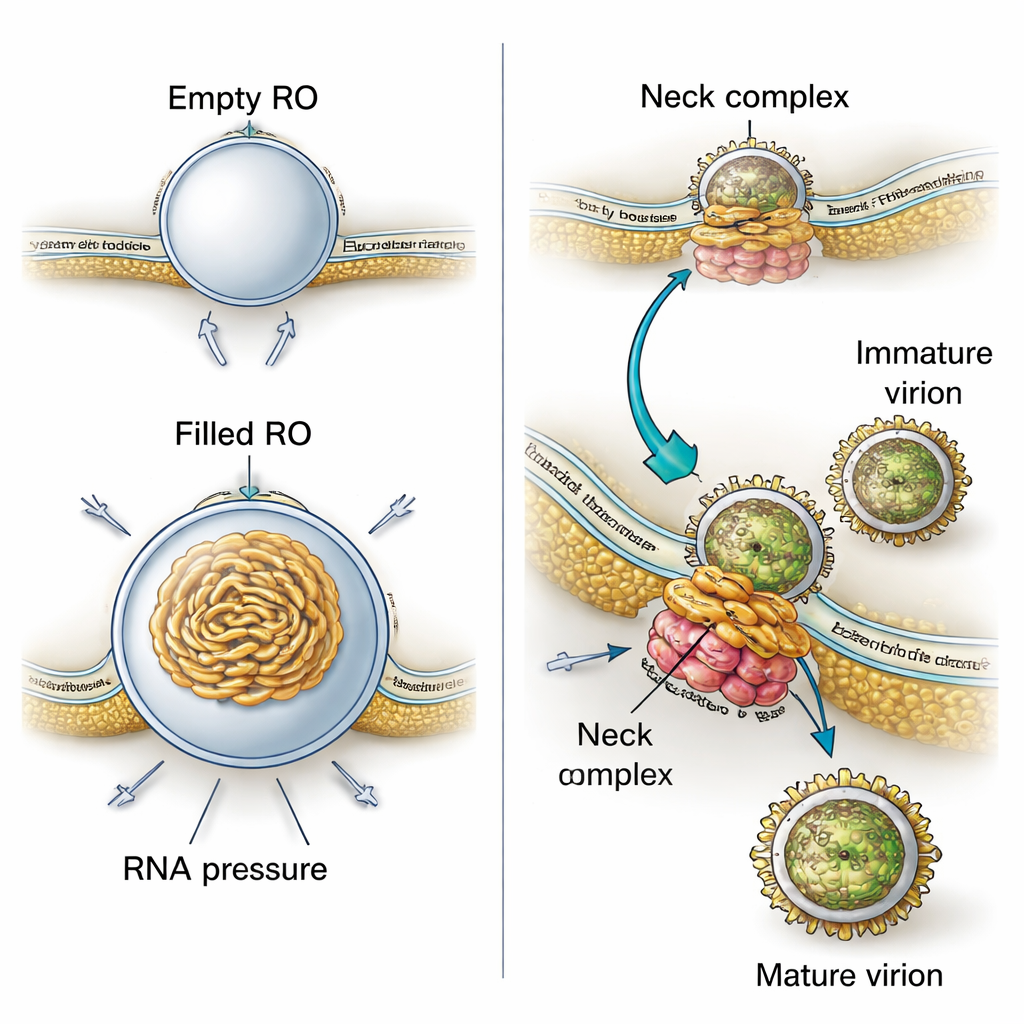
Why do these membrane pockets have such a precise, bud-like shape? By combining their 3D images with physical modeling, the authors show that two forces work together. First, the membrane of the replication organelles is consistently thicker than normal ER, suggesting that viral proteins or altered lipids stiffen and curve the membrane into a stable bud even before any RNA enters. Second, when viral RNA is present, it coils up inside the pocket and pushes outward, increasing the size of the bud. Calculations based on the measured sizes indicate that each filled organelle likely contains pressure from roughly one copy of the viral genome in double-stranded form. This means the virus uses both a pre-shaped membrane and the physical push of its own genetic material to build and stabilize its miniature factories.
Assembly Line: From Genome Copying to New Viruses
The study also uncovers how closely virus production is tied to these replication sites. In their 3D views, the researchers frequently observed new virus particles forming right next to replication organelles, budding into nearby ER-like spaces. Immature particles had a spiky surface, while more mature ones appeared smooth. Measuring distances in tomograms showed that immature particles sit closer to the replication organelles than mature ones, implying that as particles move only a short way through intertwined membrane compartments, they undergo the structural changes needed to become infectious. A large protein assembly at the narrow “neck” where each replication bud connects to the cytoplasm appears to tether the replication organelle to a neighboring membrane where budding occurs. This complex likely acts as a molecular zipper that coordinates RNA production with the packaging of that RNA into new virus particles.
Fine-Tuning Maturation with a Single Amino Acid
To probe how virus maturation is controlled, the team compared two versions of a chimeric virus that differ by just one amino acid in a site recognized by a host enzyme called furin. This enzyme cuts a viral surface protein, helping transform immature spiky particles into smooth mature ones. In test-tube cleavage assays and infected cells, the variant with an arginine at this position was cut more quickly than the variant with a glutamine. When visualized in cells by cryo-electron tomography, the faster-cleaving virus produced a higher proportion of mature particles near the replication organelles. Despite these microscopic differences, both variants showed similar growth and disease patterns in a mouse model, suggesting that the virus can tolerate a range of maturation efficiencies while still remaining infectious.
Watching Infection Directly in the Brain
Most structural studies of viruses rely on cell lines, but this work extends into living tissue. The researchers isolated a brain structure called the choroid plexus from infected mice and prepared it for cryo-electron tomography using high-pressure freezing and focused ion-beam milling. Even in this complex environment, they observed the same hallmark features: clusters of replication organelles, thickened membranes, and nearby vesicles packed with mature virus particles. This demonstrates that the membrane remodeling and tightly coupled replication–assembly–maturation sequence seen in cultured cells also operates in the brain, where these viruses cause disease.
Why These Hidden Workflows Matter
For non-specialists, the central message is that these brain-infecting viruses run a highly organized assembly line inside our cells. They sculpt cell membranes into tiny pressurized bubbles where genomes are copied, link those bubbles directly to sites where new particles bud, and even begin the final maturation steps within arm’s reach of the original replication site. Identifying the thicker replication membranes, the neck-spanning protein complex, and the closely intertwined maturation compartments not only explains how flaviviruses replicate so efficiently, but also points to new targets for antiviral drugs designed to disrupt the virus’s hidden factories and assembly tracks.
Citation: Dahmane, S., Schexnaydre, E., Zhang, J. et al. Cryo-electron tomography reveals coupled flavivirus replication, budding and maturation. Nat Commun 17, 828 (2026). https://doi.org/10.1038/s41467-026-68483-4
Keywords: flavivirus, cryo-electron tomography, replication organelles, virus maturation, tick-borne encephalitis