Clear Sky Science · en
Spectroelectrochemical insight into copper cobalt catalysts for CO2 and nitrite co-electroreduction to urea
Turning Waste Gases into Fertilizer
Urea fertilizer underpins modern agriculture, but its conventional production is energy-hungry and carbon intensive. This study explores a very different approach: using electricity to turn carbon dioxide (CO2) and nitrogen-containing waste (like nitrite from exhaust or wastewater) directly into urea. For a lay reader, the appeal is clear: a route to essential fertilizer that could cut emissions, use waste streams as feedstocks, and one day even be powered by renewable electricity.
Why Rethink How We Make Urea?
Today, urea is mostly made by first producing ammonia from nitrogen and fossil-derived hydrogen (the Haber–Bosch process), then reacting ammonia with CO2 at high temperatures and pressures. This chain of reactions is centralized, capital-intensive, and emits close to a ton of CO2 per ton of urea produced. As global fertilizer demand grows, so do the environmental costs. An alternative is to bypass fossil fuels altogether by driving the key chemical steps with electricity, ideally from renewable sources, and by using CO2 and nitrogen oxides (NOx) that are currently treated as waste.
A New Role for Copper and Cobalt
The authors design thin-film catalysts made by co-sputtering copper (Cu) and cobalt (Co) onto carbon paper, carefully tuning the Cu:Co ratio. They focus on the simultaneous electrochemical reduction of CO2 and nitrite (NO2⁻) in water at neutral pH, a setting that can favor the subtle formation of carbon–nitrogen (C–N) bonds needed for urea. The standout composition is a 1:1 mixture of Cu and Co, which achieves a urea production rate of about 61 millimoles per hour per gram of catalyst at a moderate applied voltage. Microscopy and X-ray measurements show that the metals are intimately mixed at the atomic scale and remain largely metallic during operation, providing a robust platform where both CO2 and nitrite can react efficiently. 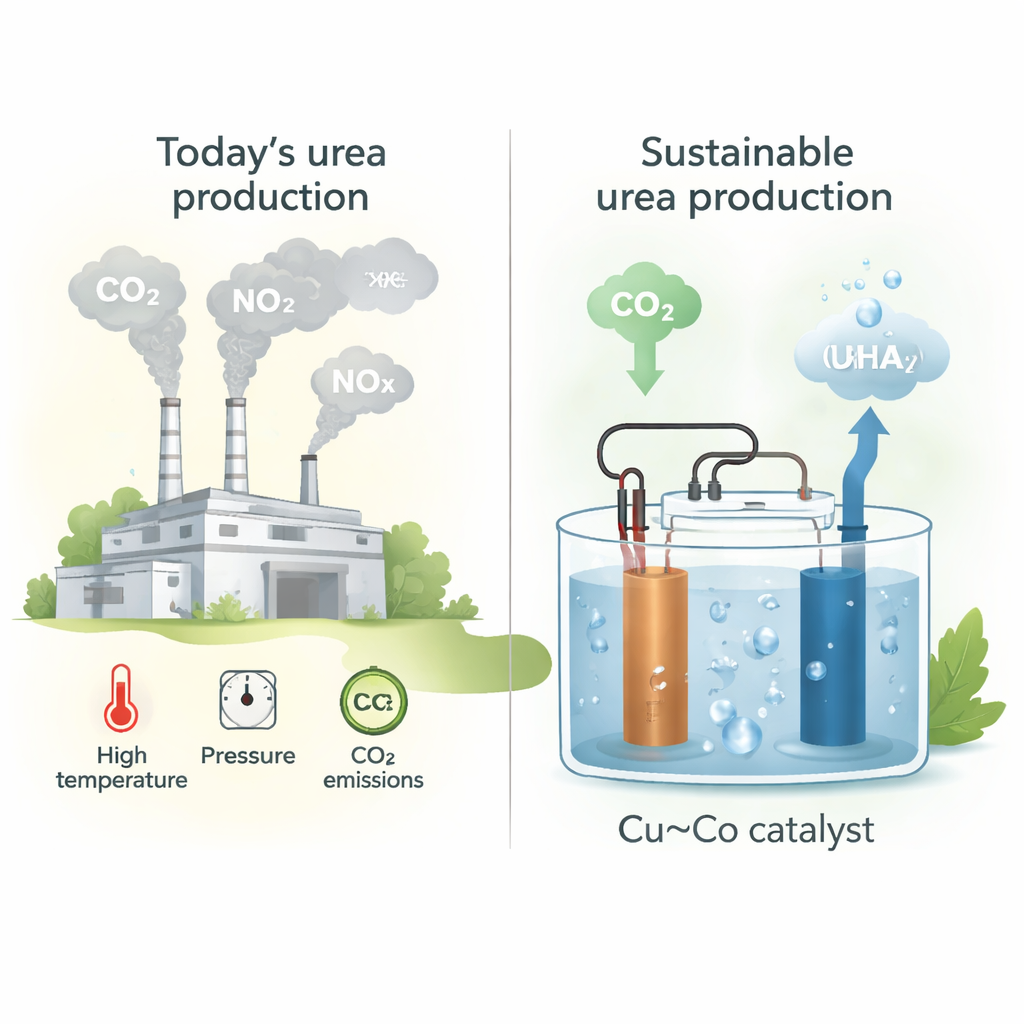
How the Catalyst Splits and Shares the Work
Making urea this way is challenging because simpler reactions—like making hydrogen gas or reducing CO2 or nitrite alone—compete strongly. The key, the researchers find, is a division of labor between copper and cobalt sites that work together. Electrochemical tests show that copper on its own mainly turns CO2 into carbon-based fuels and chemicals, while cobalt on its own mainly turns nitrite into ammonia, with no urea in either case. Only when Cu and Co atoms are mixed closely do they see significant urea. Spectroscopic “snapshots” taken during the reaction detect telltale surface-bound fragments: carbon species derived from CO2 (*CO and *COOH) that prefer copper, and nitrogen species derived from nitrite (*NH2 and related groups) that prefer cobalt. At the borders between Cu and Co, these fragments can meet and couple to form the C–N backbone of urea.
Finding the Right Conditions for Bond-Building
The study shows that composition alone is not enough; the surrounding solution and membrane must also be tuned. Urea appears only in a neutral bicarbonate electrolyte, which keeps enough dissolved CO2 available and supplies just the right amount of protons (hydrogen ions) to drive “proton-coupled electron transfer” steps without overwhelming the surface with hydrogen evolution. Under strongly acidic or strongly alkaline conditions, urea either does not form or quickly decomposes into simpler products. Experiments with different nitrogen sources reveal that nitrite is especially effective for urea formation, outperforming nitrate and hydroxylamine. Tests over many hours indicate that the Cu–Co films are relatively stable, with only modest metal loss and sustained urea production.
Peeking Under the Hood with Theory
To understand why this tandem catalyst works so well, the team turns to computer simulations based on density functional theory. These calculations support the idea that copper favors binding CO2-derived species, while cobalt better activates nitrite and stabilizes nitrogen-containing fragments. The simulations point to a particular step as the energetic bottleneck: coupling an *NH2 fragment (from nitrite) with a *COOH fragment (from CO2) to form an intermediate sometimes written as *NH2CO. Once this piece is in place, the remaining steps to urea are comparatively easy. The Cu–Co interface lowers the energy cost of this crucial coupling compared with pure copper or pure cobalt, explaining the experimentally observed boost in urea yield for the bimetallic system. 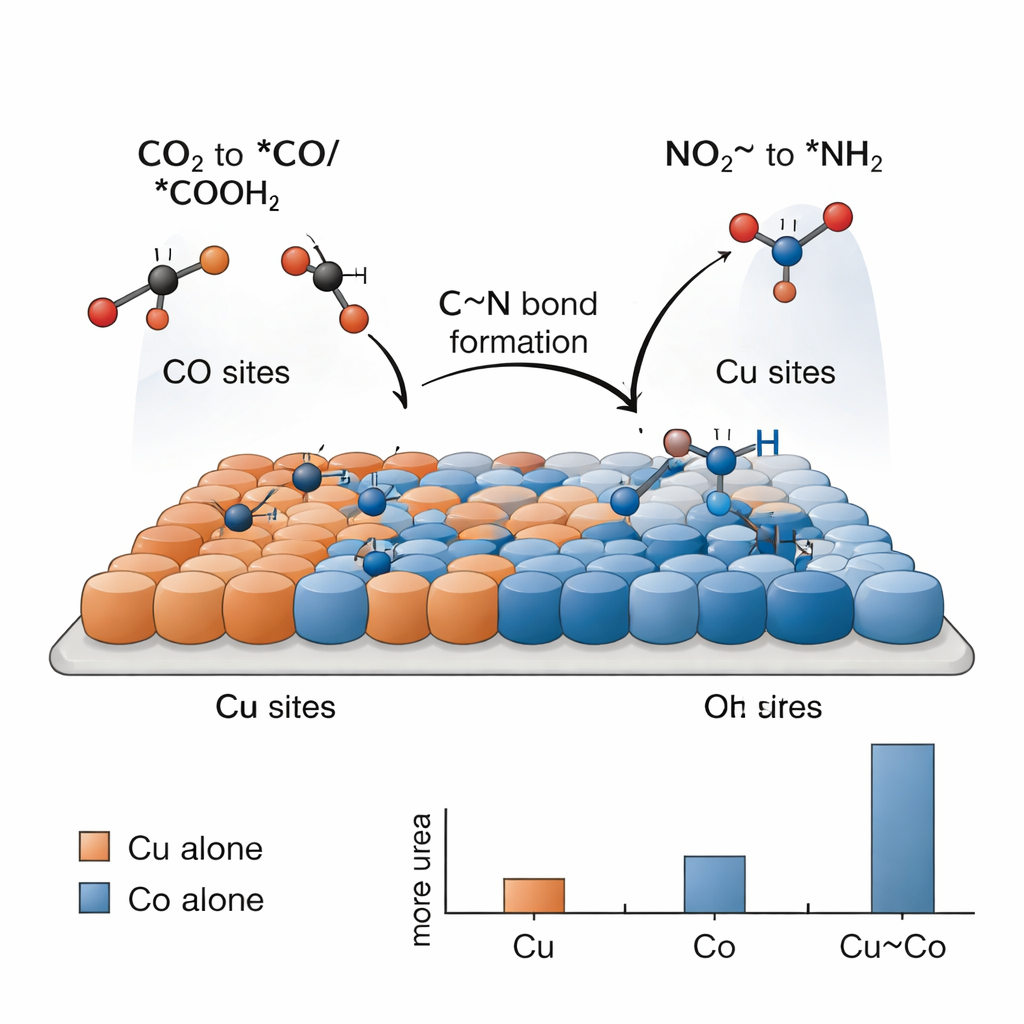
From Lab Insight to Greener Fertilizer
In practical terms, this work does not yet replace today’s gigantic urea plants, but it clarifies how a future electrochemical route might be designed. By showing that copper and cobalt can operate in tandem—one feeding activated carbon species, the other activated nitrogen species, both under carefully controlled proton conditions—the study maps out a realistic pathway toward cleaner, potentially decentralized urea production that uses CO2 and nitrogen wastes as resources rather than pollutants.
Citation: Ramadhany, P., Trần-Phú, T., Yuwono, J.A. et al. Spectroelectrochemical insight into copper cobalt catalysts for CO2 and nitrite co-electroreduction to urea. Nat Commun 17, 1776 (2026). https://doi.org/10.1038/s41467-026-68481-6
Keywords: electrochemical urea synthesis, copper cobalt catalyst, CO2 utilization, nitrite reduction, C–N coupling