Clear Sky Science · en
Structural basis for selective inhibition of human GABA transporter GAT3
Why calming brain signals matters
Our brains rely on a delicate balance between signals that excite and those that calm. A key calming messenger is GABA, a chemical that helps prevent runaway activity linked to conditions such as epilepsy, stroke damage, and Alzheimer’s disease. This study focuses on a protein called GAT3, which clears GABA from around nerve cells. By revealing, in atomic detail, how a new molecule latches onto GAT3 and shuts it down, the researchers provide a structural roadmap for designing next‑generation drugs that could gently boost the brain’s natural braking system.
The brain’s own brake pedal
GABA is the main inhibitory, or calming, neurotransmitter in the central nervous system. After GABA is released from nerve endings, it must be quickly removed so that brain circuits can reset. That cleanup is carried out by transporter proteins that pump GABA back into cells. Two of these transporters dominate in the brain: GAT1, found mainly on neurons, and GAT3, found on support cells called astrocytes. While a GAT1‑blocking drug (tiagabine) has been used for decades to treat epilepsy, comparable medicines that target GAT3 have lagged behind. Yet GAT3 is especially interesting because it controls GABA levels outside synapses and shapes long‑lasting, background inhibition that can fine‑tune brain activity. 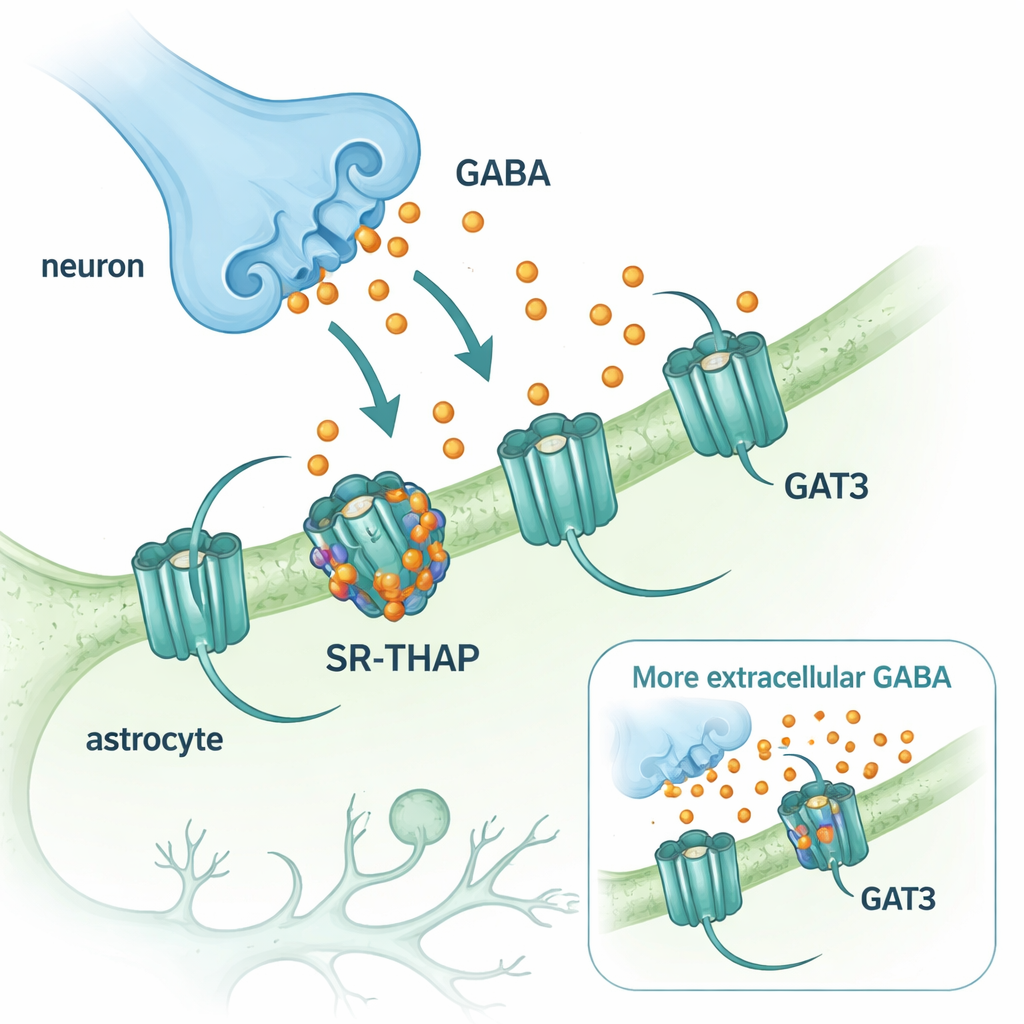
Designing a more precise molecular lock
Attempts to inhibit GAT3 have been hampered by a lack of detailed structural information. Many past compounds were created by trial and error, often by making bulky variations of known GABA‑like molecules. One of the best known, (S)‑SNAP‑5114, turned out to be chemically unstable and difficult to use in living animals. Building on a sturdier relative of this compound, the authors developed SR‑THAP, a new small molecule designed to fit GAT3 more snugly and resist breakdown. Experiments in human cell cultures showed that SR‑THAP strongly blocks GAT3 and does so in a non‑competitive way: instead of simply competing with GABA at the usual site, it changes how the transporter works, reducing its maximum transport capacity.
Seeing GAT3 at near‑atomic resolution
To understand exactly how SR‑THAP works, the team turned to cryo‑electron microscopy, a technique that images flash‑frozen proteins with electrons and reconstructs their three‑dimensional shape. They solved three versions of human GAT3: bound to SR‑THAP, bound to GABA, and in a substrate‑free state. All three reveal the typical architecture seen in related neurotransmitter transporters: 12 tightly packed helices that span the membrane and form a central pathway for GABA and its companion ions, sodium and chloride. In the GABA‑bound structure, the transporter sits in an “inward‑occluded” state—GABA and a sodium ion are clamped in the middle, sealed off from both outside and inside. A key aromatic amino acid forms a specialized interaction with GABA’s amino group, helping explain how GAT3 recognizes its substrate.
How SR‑THAP wedges the transporter open
In striking contrast, the SR‑THAP‑bound structure captures GAT3 in an “inward‑open” state, with the internal side of the protein gaping toward the cell’s interior. SR‑THAP is nestled deep in this inner cavity, wedged between several helices that normally move during the transport cycle. It occupies three small pockets with different chemical characters, engaging a network of hydrogen bonds and greasy contacts. This positioning effectively jams the transporter so that GABA and ions can no longer complete their journey. The authors show that a few specific amino acids lining this cavity differ between GAT3 and its close relatives; swapping one of these residues alters both transport activity and SR‑THAP potency, helping explain why the inhibitor is far more selective for GAT3 than for GAT1 or related transporters. 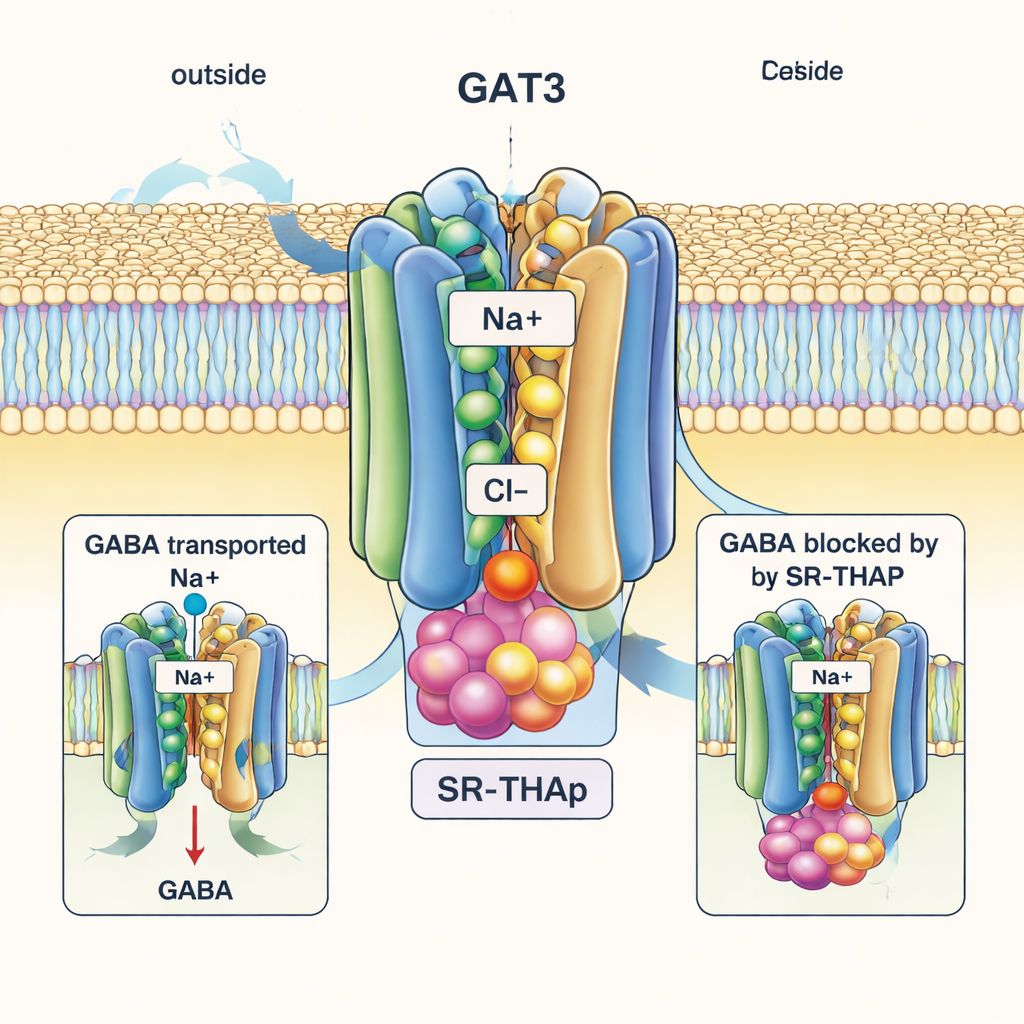
Opening doors to new epilepsy and brain‑health drugs
Together, these structures reveal how GAT3 cycles between different shapes to move GABA, and how SR‑THAP exploits a less‑conserved inner pocket to freeze that cycle. For non‑specialists, the key takeaway is that the study delivers a detailed molecular blueprint for switching off GAT3 in a highly selective way. Such compounds could raise calming GABA levels in a more finely tuned manner than existing drugs, potentially leading to new treatments or research tools for epilepsy, stroke recovery, Alzheimer’s disease, and other conditions where inhibitory signaling is disturbed.
Citation: Mortensen, J.S., Bavo, F., Jensen, M.H. et al. Structural basis for selective inhibition of human GABA transporter GAT3. Nat Commun 17, 1774 (2026). https://doi.org/10.1038/s41467-026-68479-0
Keywords: GABA transporter, GAT3, epilepsy, cryo-EM, neurotransmitter inhibition