Clear Sky Science · en
Structure of a Brochothrix thermosphacta bacteriophage reveals cell wall adsorption mechanism in Gram-positive infecting siphophages
Viruses That Keep Our Food Fresh
Bacteria-killing viruses, called bacteriophages, are quietly shaping our health and food supply. Some cause deadly infections, but others can be harnessed to combat foodborne germs and spoilage. This study reveals, in striking atomic detail, how one such virus, named NF5, latches onto and pierces the tough outer shell of a meat-spoilage bacterium. Understanding this microscopic battle could help us design safer ways to preserve food and fight antibiotic‑resistant bacteria.
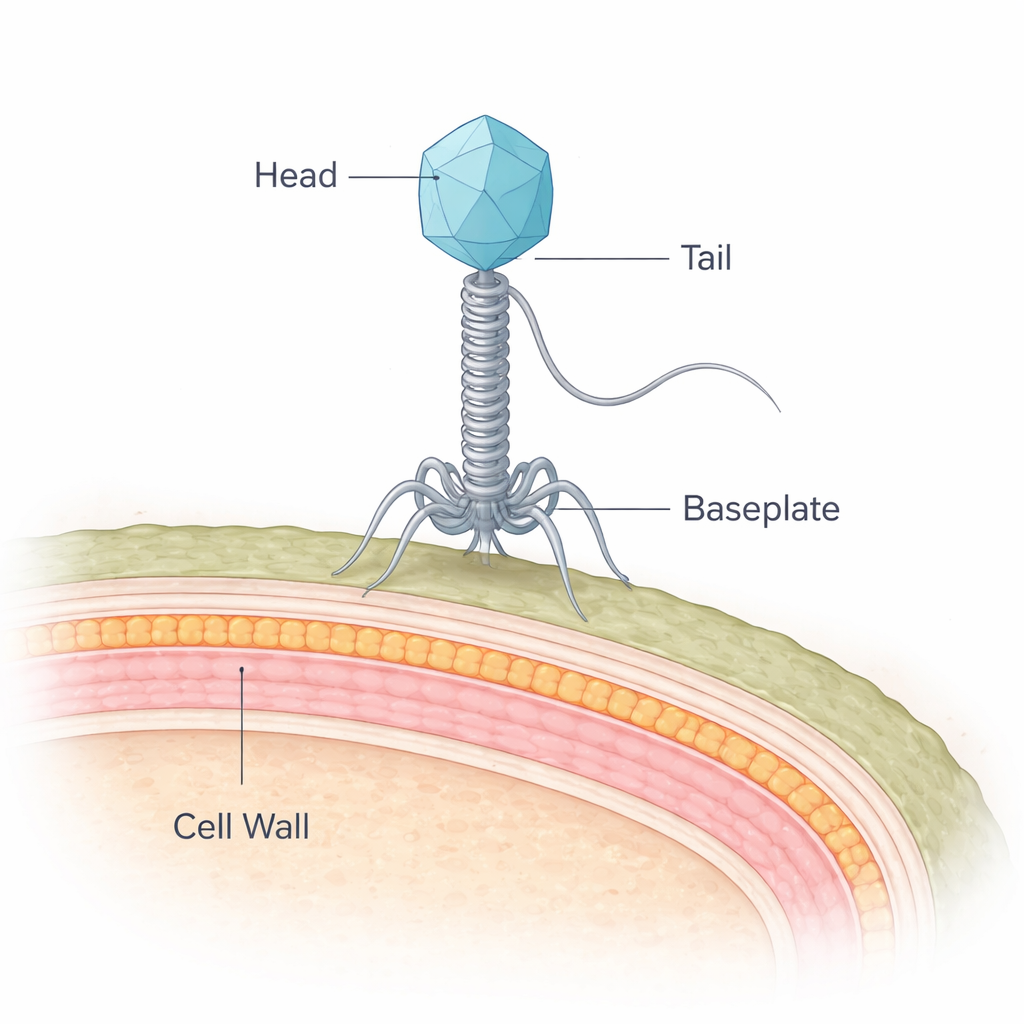
A Meat Spoiler Meets Its Natural Enemy
The bacterium at the center of this work, Brochothrix thermosphacta, is a common culprit behind the off-odors and slime that develop on refrigerated meat. NF5 is a virus that infects this bacterium and belongs to a group called siphophages, which carry their DNA in a protein shell and deliver it through a long, flexible tail. While scientists have detailed the structures of many viruses that infect so‑called Gram-negative bacteria, those targeting Gram-positive bacteria like B. thermosphacta have remained much less understood. Gram-positive bacteria pose a special puzzle because they are shielded by a thick, multilayered cell wall rather than a thin wall and outer membrane.
Building a Molecular Syringe, Atom by Atom
Using state-of-the-art cryo-electron microscopy, the researchers froze millions of NF5 particles and reconstructed their three-dimensional structure at near‑atomic resolution. They identified 11 different viral proteins that together assemble the viral head, neck, tail tube, and an elaborate baseplate at the tail tip, totaling 643 protein chains. The head forms a sturdy icosahedral shell around the viral DNA, while a 135‑nanometer tail extends downward like a flexible syringe. Rings of repeated proteins form a hollow tail tube, whose inner surface is strongly negatively charged—an arrangement that likely helps the virus rapidly shoot its DNA into the host.
The Smart Drill at the Tail Tip
The most intricate part of NF5 is the baseplate, a multi-layered structure that acts as a sensor, drill, and anchor all at once. Near the center sits a “tape measure” protein filling the tail tube and a tail-associated lysin that plugs the tube shut until infection begins. Surrounding this core are proteins that recognize and grip the bacterial surface. Some act like springy arms that can bend dramatically, helping the baseplate tilt and then snap into a position perpendicular to the cell wall. Others resemble fibers found in related viruses and are thought to bind specific molecules in the thick Gram-positive wall and even nibble away at parts of the cell wall’s sugar‑rich mesh. Intriguingly, one NF5 side-fiber protein appears to combine functions that are split across multiple proteins in other Gram-positive viruses, hinting at an efficient, evolutionarily streamlined design.
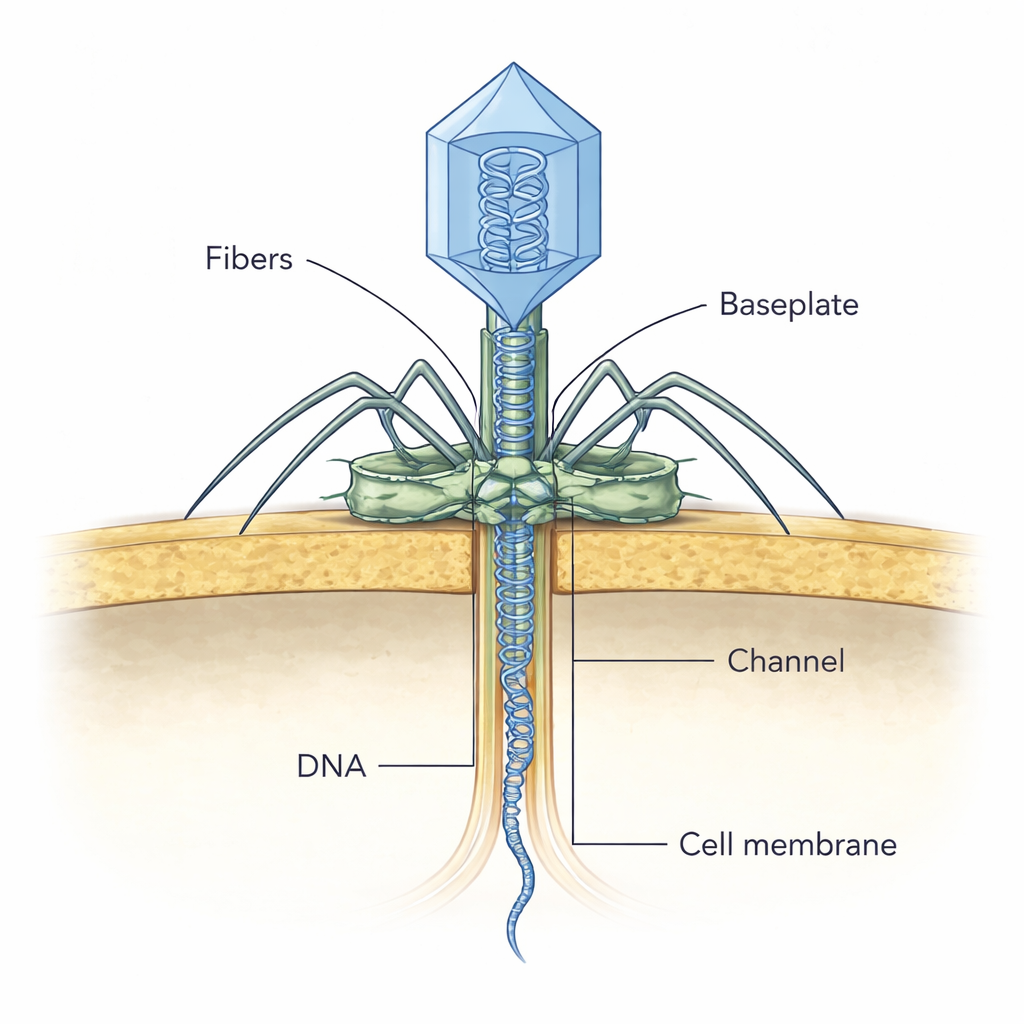
Catching a Virus in the Act
To see how these parts work together during infection, the team used cryo-electron tomography on thin slices of infected bacteria. They captured NF5 particles at different apparent stages of attack. In early snapshots, the virus attaches to the cell wall at an angle, likely using its outer fibers to locate suitable receptors. Later, the baseplate aligns itself perpendicular to the cell surface, and the attached viral head still appears full of DNA. In subsequent stages, the head’s interior fades as DNA is released, and a thin, channel-like density appears crossing the bacterial wall toward the cell membrane. The authors propose that once the tail tip enzyme contacts and digests the cell wall, it rearranges, opening the plug and allowing the tape‑measure protein to slide out and form this temporary tunnel through which the viral DNA travels safely into the cell.
Why This Matters Beyond One Virus
By comparing NF5 to viruses that infect other bacteria, the researchers show that key tail and baseplate proteins have evolved differently in Gram-positive versus Gram-negative phages to cope with their hosts’ very different surface architectures. These structural tweaks—extra domains, longer arms, or combined functions in single proteins—appear to be fine-tuned adaptations to thicker, more complex cell walls. The work delivers a detailed blueprint of how a virus can recognize, attach to, and breach a tough bacterial barrier, offering a foundation for engineering phages or phage-inspired tools to better control food spoilage and potentially combat harmful Gram-positive pathogens.
Citation: Peng, Y., Pang, H., Zheng, J. et al. Structure of a Brochothrix thermosphacta bacteriophage reveals cell wall adsorption mechanism in Gram-positive infecting siphophages. Nat Commun 17, 1772 (2026). https://doi.org/10.1038/s41467-026-68477-2
Keywords: bacteriophage structure, Gram-positive bacteria, cryo-electron microscopy, food spoilage control, phage infection mechanism