Clear Sky Science · en
Structural mechanisms for inhibition and activation of human small-conductance Ca2+-activated potassium channel SK2
Why tiny potassium channels matter
Every thought you have and every heartbeat you feel depends on electrical signals in your cells. These signals are shaped by microscopic pores, called ion channels, that let charged atoms flow in and out. One such family, the small‑conductance calcium‑activated potassium channels (SK channels), helps control how nerve cells fire and how the heart keeps its rhythm. This paper reveals, in atomic detail, how different drugs and a bee toxin can either shut down or boost one key member of this family, the human SK2 channel—insights that could guide new treatments for disorders ranging from atrial fibrillation to tremor and memory problems.
How SK2 channels act as cellular brakes
SK2 channels sit in the cell membrane and act as tiny brakes on electrical activity. When the inside of a cell briefly fills with calcium ions during an electrical signal, a helper protein called calmodulin senses this rise. Calmodulin is attached to the inner side of SK2 and, when it grabs calcium, it tugs on part of the channel like a pull‑cord, opening a gate that lets potassium ions flow out. This outward flow helps quiet the cell after it fires, shaping how often neurons can send signals and how reliably heart cells beat. Because SK1–3 channels are widespread in the brain and heart, and SK2 is a particularly important subtype, understanding exactly how they open, close, and respond to drugs is crucial for designing precise medicines.
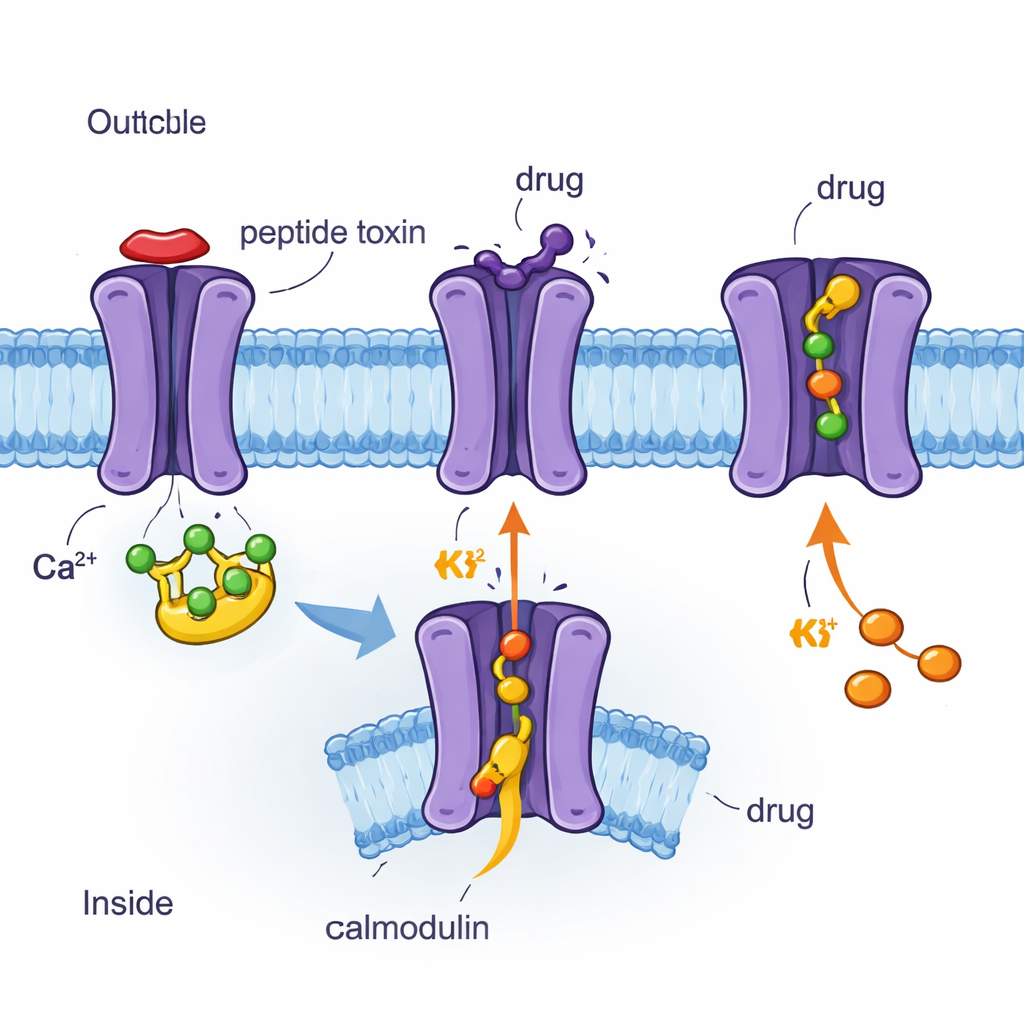
Using cryo‑EM to see channels and drugs atom by atom
The authors used cryo‑electron microscopy (cryo‑EM), a technique that images flash‑frozen proteins at near‑atomic resolution, to determine four three‑dimensional structures of the human SK2 channel bound to calmodulin and different molecules. These included the bee venom peptide apamin, the synthetic blocker UCL1684, the clinical inhibitor AP30663, and the clinical activator CAD‑1883. All four structures show SK2 as a four‑part assembly that spans the membrane, with calmodulin attached on the inside. Comparing these snapshots revealed which parts of the channel are rigid, which are flexible, and how calmodulin moves when the channel is in a closed or open state. The researchers also combined these images with electrical measurements in cells, testing how specific amino‑acid changes alter the channel’s response to each compound.
A bee toxin and a lab drug that plug the outer mouth
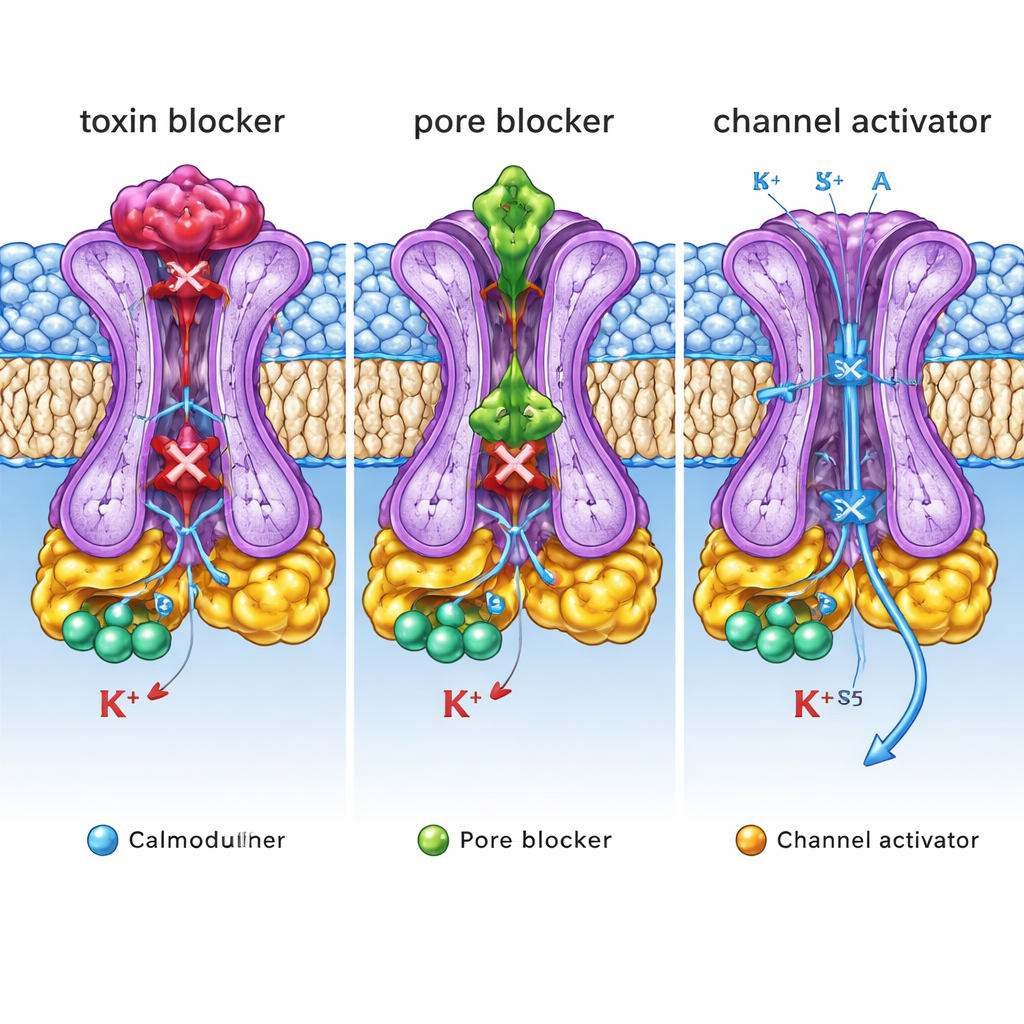
Apamin, a small peptide from bee venom, has long been known as an extremely potent and selective SK2 blocker used in research. The new structures show that apamin binds like a cork on the outer mouth of the SK2 pore. It nestles into a cup‑shaped vestibule formed by a short loop between two helices (the S3–S4 linker) that is well ordered in SK2 but floppy in a related channel, SK4, explaining why SK4 is insensitive to apamin. Key positively charged residues on apamin reach between four aromatic side chains of the channel, making strong electrostatic and stacking interactions that completely seal the potassium pathway. UCL1684, a synthetic bis‑quinolinium compound, occupies essentially the same outer pocket and also blocks the ion exit, but its smaller size lets it sit deeper, mimicking the position of apamin’s critical positive charges. Mutating channel residues that shape this vestibule sharply reduces sensitivity to apamin and, to a lesser extent, UCL1684, confirming that this sculpted outer loop is a major determinant of toxin and drug action.
A central plug and an internal wedge that tune activity
In contrast, the clinical candidate AP30663 binds inside the central cavity of SK2, just below the selectivity filter that chooses potassium ions. There it acts as a physical plug, wedged between specific side chains lining the pore. Changes to these lining residues weaken the ability of AP30663 to inhibit SK2, and sequence comparisons explain why its close analogs can distinguish SK1–3 from SK4. CAD‑1883, a positive modulator developed for movement disorders, uses yet another strategy. It slips into a pocket at the interface between calmodulin’s N‑lobe and a short linker (S4–S5) that couples calmodulin’s movement to the channel gate. By reinforcing contacts in this pocket, CAD‑1883 effectively glues calmodulin and SK2 together in a configuration that pulls the inner gate open and widens the pore. Structural comparisons show that this CAD‑1883‑bound state resembles a fully open channel, although the selectivity filter may adopt an inactivated form after prolonged opening.
What these structural blueprints mean for medicine
Taken together, these structures reveal three distinct “control knobs” on a single SK2 channel: an outer lid where toxins and some blockers sit, an inner pore cavity where other inhibitors plug the flow, and a side pocket where activators stabilize opening. For a non‑specialist, the key message is that the same ion channel can be tuned up or down by drugs that grab very different structural handles. By mapping these handles with atomic precision and tying them to functional effects, this work provides a detailed blueprint for designing next‑generation molecules that selectively dampen or enhance SK2 activity. Such tailor‑made drugs could one day help normalize abnormal brain rhythms, ease tremors, or correct irregular heartbeats with fewer side effects.
Citation: Ma, B., Wu, D., Cao, E. et al. Structural mechanisms for inhibition and activation of human small-conductance Ca2+-activated potassium channel SK2. Nat Commun 17, 1770 (2026). https://doi.org/10.1038/s41467-026-68475-4
Keywords: SK2 potassium channels, calcium-activated channels, cryo-EM structures, ion channel modulators, atrial fibrillation and tremor