Clear Sky Science · en
Impaired stem cell migration and divisions in Duchenne muscular dystrophy revealed by live imaging
Why this muscle story matters
Duchenne muscular dystrophy (DMD) is a fatal childhood disease that gradually destroys muscle. For decades, most research has focused on the large muscle fibers that break down in DMD. This study shows that a second, quieter player is also in trouble: the muscle’s own stem cells, which normally repair damage. By watching these cells live inside mouse muscles and on individual fibers, the authors reveal how stem cells in DMD lose their way, offering new ideas for treatments that could help muscles regenerate more effectively.
Muscle’s built-in repair crew
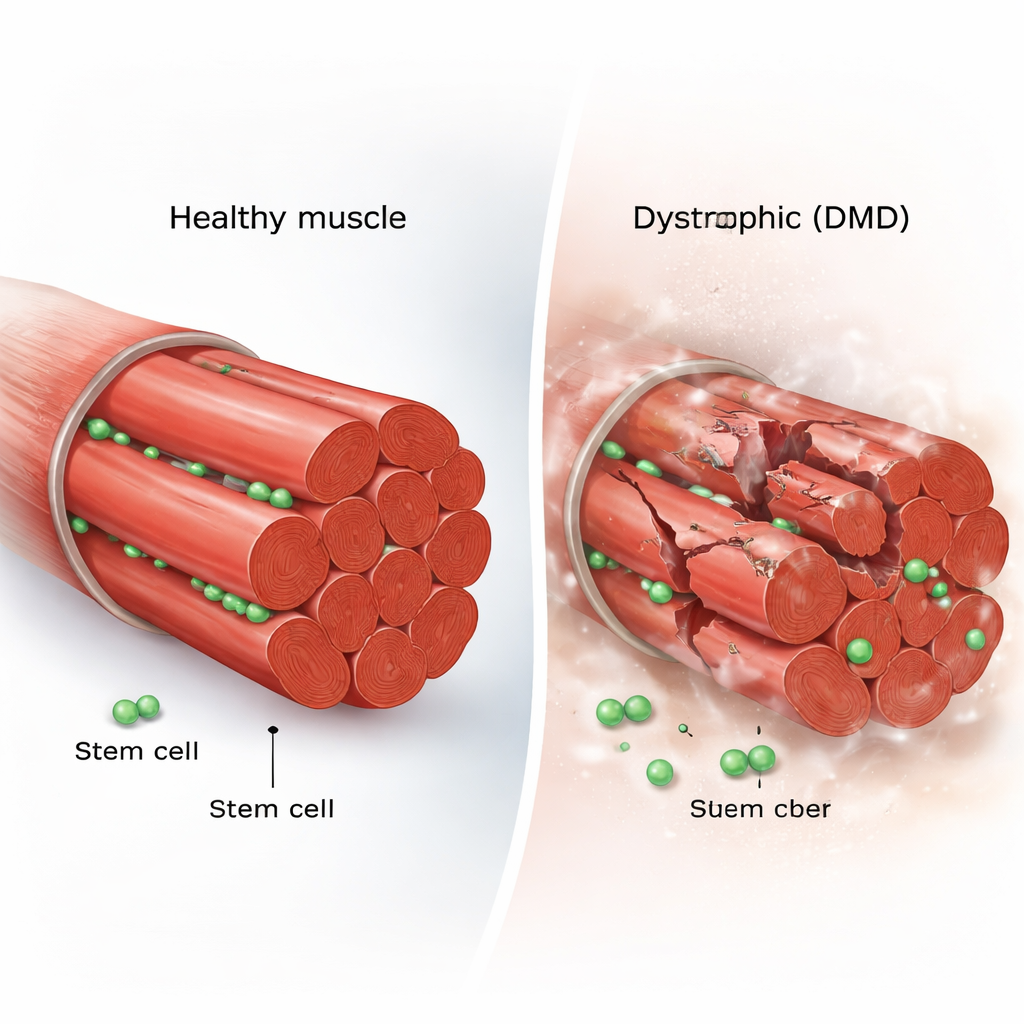
Every skeletal muscle fiber is flanked by a handful of resident stem cells, known as muscle satellite cells. In healthy muscle, these cells sleep most of the time. When injury strikes, they wake up, divide, and either make new stem cells to refill the reserve or produce specialized cells that fuse into damaged fibers. This careful balance between self-renewal and differentiation keeps muscle tissue strong over a lifetime. In DMD, however, the muscle fibers repeatedly tear because they lack dystrophin, a key structural protein, raising the question of whether the repair crew itself becomes exhausted or misdirected.
Filming stem cells in action
To answer this, the researchers used the mdx mouse, a standard model of DMD, and engineered its muscle stem cells to glow under the microscope. They then performed intravital imaging, a technique that lets scientists film living cells inside an anesthetized animal over many hours. They also developed a custom “microwell” system to hold single muscle fibers in culture while tracking every stem cell division and movement. This dual approach allowed them to follow individual cells from quiet rest through repeated divisions, migrations, and ultimately fusion into new muscle, capturing behavior that static snapshots could easily miss.
When repair cells hurry and stumble
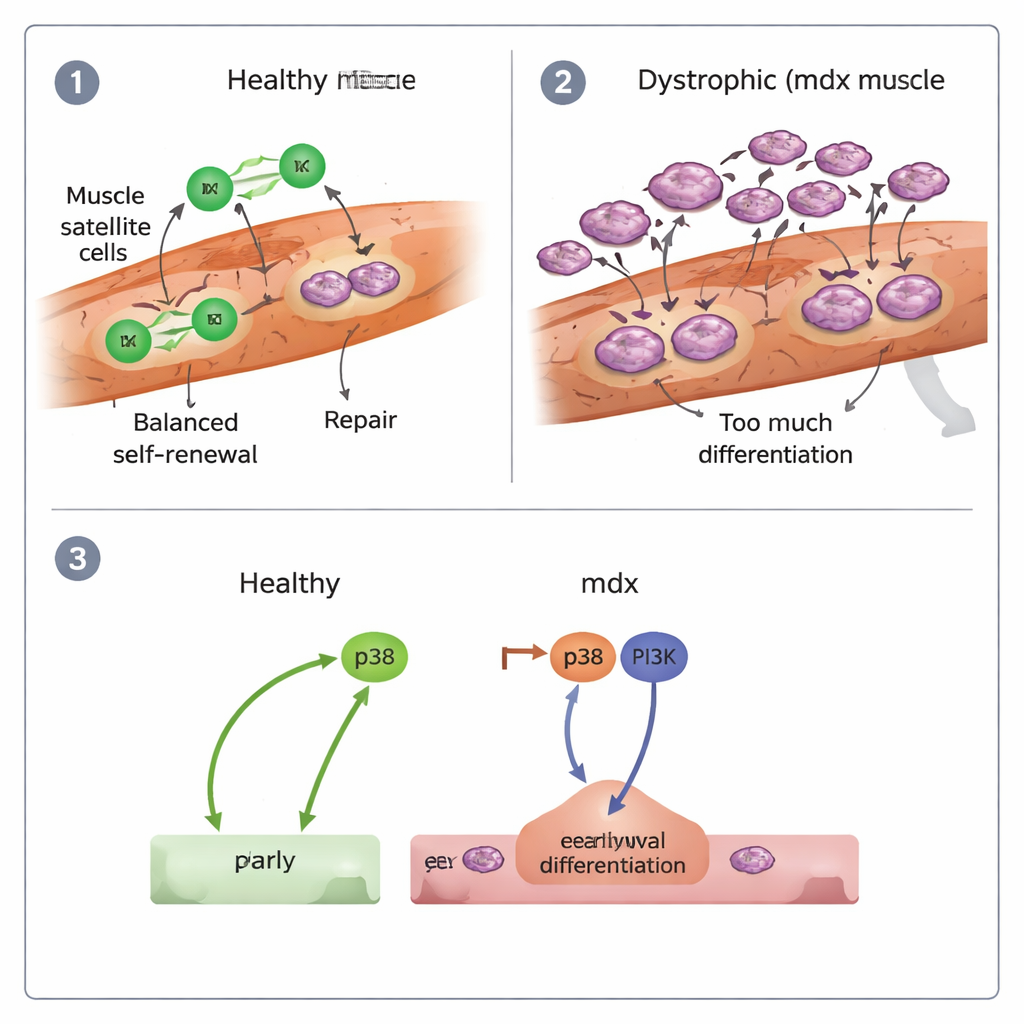
Inside injured healthy muscles, stem-cell–derived myogenic cells elongated and crawled along fibers with relatively straight, directed paths, dividing and spreading efficiently. In contrast, dystrophic (mdx) cells often became round and sluggish. They moved more slowly and with less direction, and a noticeable fraction remained almost immobile. The team also found that mdx cells fused into new muscle fibers earlier than normal, and many daughter cells migrated together instead of separating after division. Overall, mdx muscles showed signs of “precocious differentiation”: stem cells rushed into becoming muscle cells and forming fibers, rather than maintaining a robust pool for ongoing repair.
Unbalanced cell divisions and a confused neighborhood
On isolated fibers, the researchers could classify how each stem cell divided. In healthy muscles, most divisions were symmetric but balanced—many produced two proliferating daughters that could keep expanding the repair pool, and fewer produced two terminally differentiating cells. Asymmetric splits, where one daughter stayed a stem cell and the other committed to repair, were relatively rare but present. In mdx muscles, the pattern shifted dramatically: symmetric divisions skewed toward producing two differentiating cells, with far fewer divisions generating two renewing cells. Cross-grafting experiments, where healthy stem cells were placed on dystrophic fibers and vice versa, showed that migration defects were largely dictated by the damaged fiber environment, while the overly differentiative behavior of mdx stem cells followed the cells themselves, shaped in part by their inflammatory history.
Miswired signals and what it means for therapy
The authors also probed the molecular switches that push stem cells to differentiate. They focused on two signaling routes, known as p38 and PI3K. In healthy cells, blocking p38 strongly reduced differentiation, while inhibiting PI3K had little effect. In mdx cells, however, both pathways had to be blocked together to curb the excessive rush into specialization, and even then their ability to keep proliferating remained poor. This suggests that DMD stem cells are driven into “early burnout” by combined p38 and PI3K activity, contributing to regeneration that starts fast but cannot be sustained.
A new view of Duchenne: a stem cell disease too
To a lay reader, the core message is that DMD is not just a problem of fragile muscle fibers; it is also a problem of the repair system meant to fix them. In dystrophic muscle, stem cells move less efficiently, divide in a way that depletes the reserve, and respond abnormally to key growth signals. By pinpointing which aspects are caused by the damaged muscle environment and which are built into the stem cells’ altered state, this work suggests new strategies: therapies that temper p38 and PI3K signaling, restore balanced cell divisions, or improve the local niche could help rebuild muscle more effectively and extend the benefits of gene or cell-based treatments for people living with Duchenne muscular dystrophy.
Citation: Sarde, L., Letort, G., Varet, H. et al. Impaired stem cell migration and divisions in Duchenne muscular dystrophy revealed by live imaging. Nat Commun 17, 1769 (2026). https://doi.org/10.1038/s41467-026-68474-5
Keywords: Duchenne muscular dystrophy, muscle stem cells, cell migration, stem cell division, regenerative medicine