Clear Sky Science · en
Breaking the oxo-wall for Co(IV)-oxo species and their nanoconfined catalytic performance within Ce-Co lamellar membrane
Cleaning Stubborn Pollutants from Water
Many medicines and industrial chemicals slip through ordinary wastewater treatment and end up in rivers and drinking water at trace levels. Even at very low concentrations, these "micropollutants" can harm ecosystems and human health. This study reports a new kind of catalytic membrane that not only filters water but also chemically destroys such pollutants, doing so in a highly efficient and selective way that could help make advanced water treatment more practical.
Why Cobalt Chemistry Hit a Wall
One promising way to break down tough pollutants is to use powerful oxygen‑bearing metal species that behave like targeted oxidizing bullets. For cobalt, the most effective form is a short‑lived complex called Co(IV)=O. In theory, this species can out‑perform similar iron and manganese oxidants. In practice, however, it is very hard to form and keep stable. A long‑standing principle in inorganic chemistry, nicknamed the "oxo‑wall," says that late transition metals such as cobalt should struggle to hold strong double bonds to oxygen in high oxidation states. As a result, conventional cobalt‑based water treatments mostly generate free radicals such as hydroxyl and sulfate radicals, which are less selective, shorter‑lived, and more easily disrupted by other substances in real water.
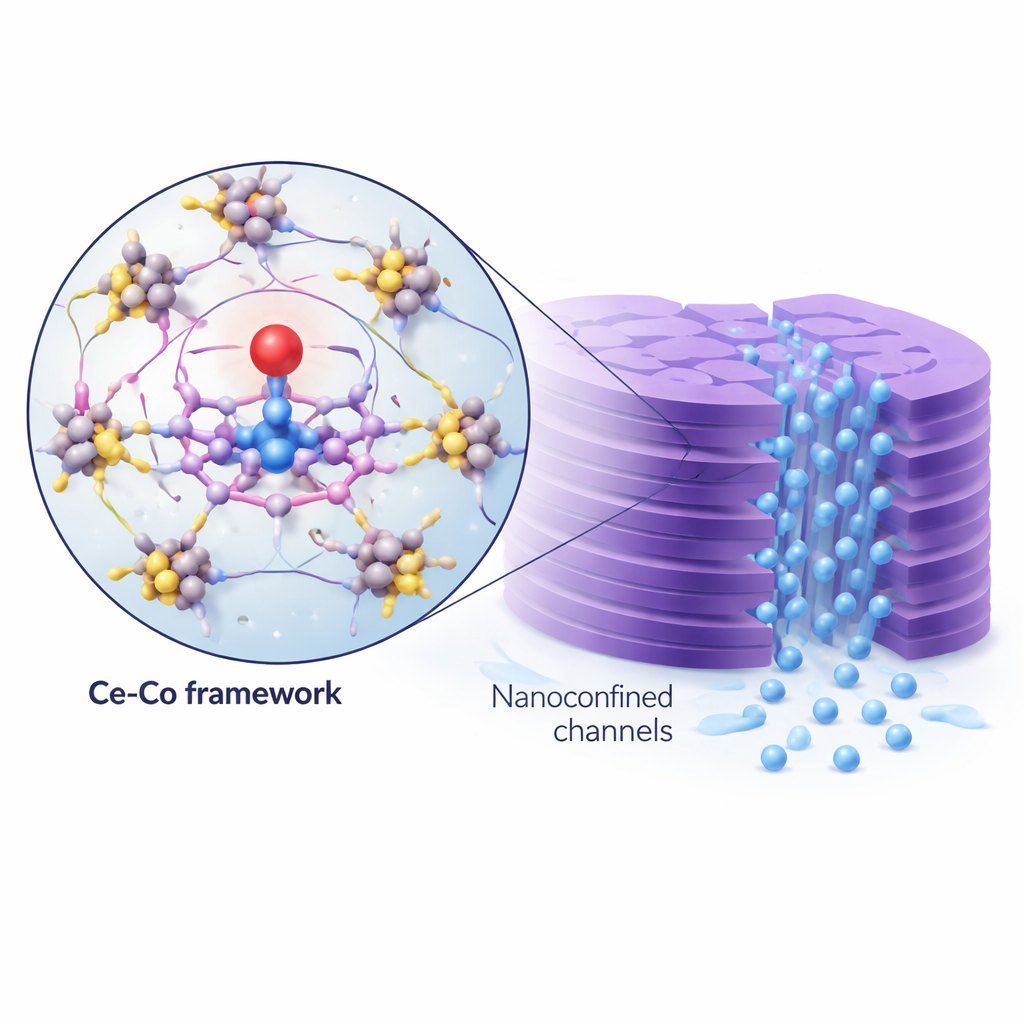
Building a Smart Molecular Scaffold
The researchers tackled this problem by designing a highly ordered framework built from porphyrin molecules – ring‑shaped organic units that can hold metal atoms like a tiny claw. Each ring anchors a single cobalt atom in a well‑defined four‑nitrogen site, and these rings are connected by cerium‑oxide clusters into two‑dimensional sheets. Computer calculations showed that the electron‑withdrawing cerium linkers subtly pull electron density away from the cobalt center through the extended network of bonds. This long‑range tuning leaves more empty cobalt orbitals available for bonding to oxygen, strengthens the cobalt–oxygen bond, and helps bypass the traditional oxo‑wall limitation.
Proving a New Reaction Pathway
To activate the system, the team used peroxymonosulfate, a common oxidant in advanced water treatment. In a conventional cobalt framework used for comparison, this oxidant mostly produced a mixture of free radicals. In contrast, the cerium‑modified framework showed almost no detectable radicals. Through a combination of specialized spectroscopy, chemical quenching tests, and probe molecules that react preferentially with Co(IV)=O, the authors demonstrated that a high‑valent cobalt‑oxo species dominates the reaction in their new material. Detailed quantum‑chemical calculations revealed why: on the cerium‑linked scaffold, the oxidant binds in a way that allows an internal shift of a proton and a cooperative two‑electron transfer from cobalt, leading to Co(IV)=O via an overall energy‑lowering route that is not available in the control material.
Trapping Chemistry in Nano‑Sized Channels
Turning this chemistry into a practical device, the team stacked the two‑dimensional sheets into a thin lamellar membrane. The gaps between the sheets form nanometer‑scale channels lined with isolated cobalt sites. As polluted water is pushed through, molecules of the oxidant and the target contaminants are forced into these tight spaces, greatly increasing how often they collide with the catalytic sites. Measurements showed that this membrane, paired with peroxymonosulfate, could almost completely remove the test pollutant ranitidine in about a minute, with water flowing through at rates suitable for treatment applications. Computer simulations supported the idea that nanoconfinement concentrates reactants and shortens diffusion distances, raising the local Co(IV)=O level by roughly a thousand‑fold compared with a simple particle suspension.
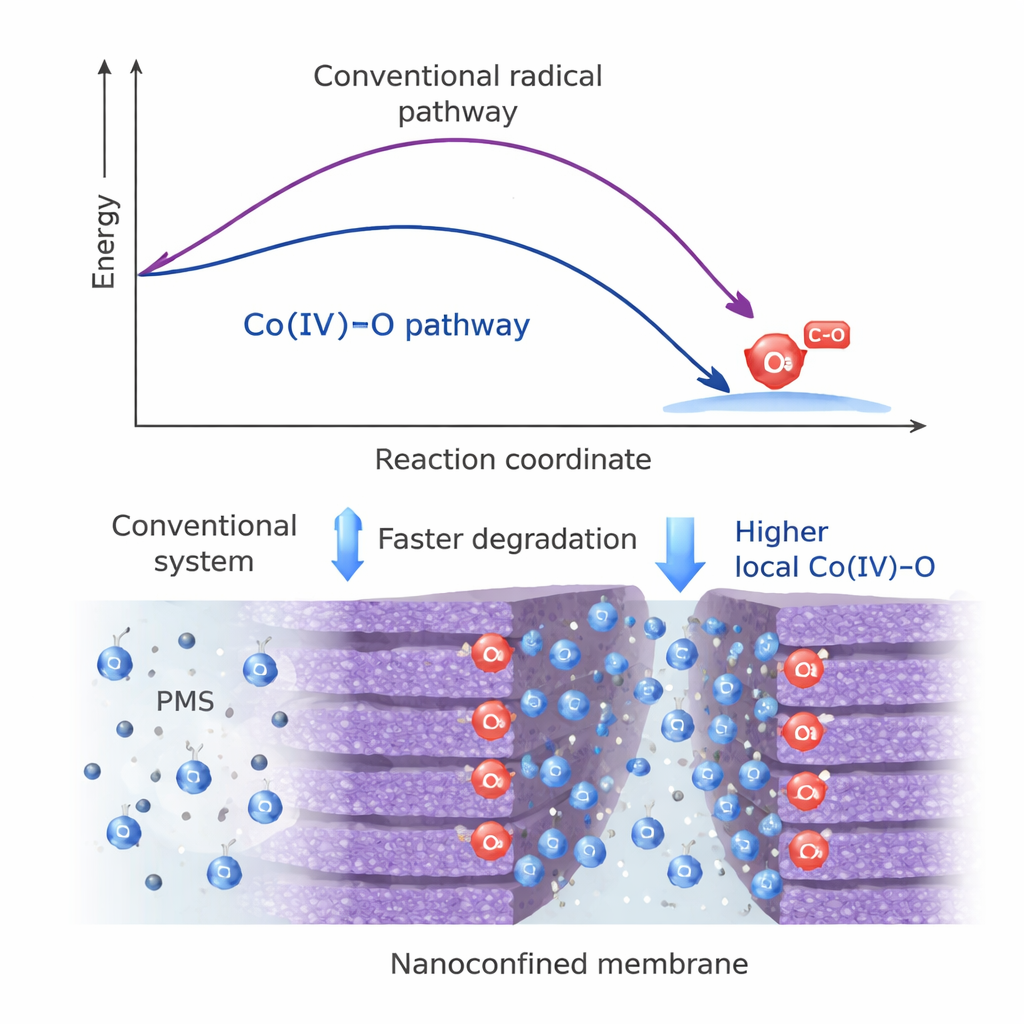
Selective, Stable, and Safer Water Treatment
The membrane worked well in different water types, including tap and lake water, and was resilient to common dissolved salts. It selectively attacked pollutants with electron‑rich groups, such as many antibiotics, while leaving more resistant molecules largely untouched – a hallmark of the targeted Co(IV)=O pathway. Long runs of nearly four days showed stable water flow and high removal, with very low cobalt leakage and only gradual loss of activity that could be restored by a mild chemical treatment. Toxicity tests indicated that the breakdown products of ranitidine were significantly less harmful than the original drug. Overall, the study demonstrates a strategy to overcome a fundamental chemical barrier and harness highly reactive cobalt‑oxo species inside nanostructured membranes, pointing toward more efficient and sustainable technologies for cleaning complex wastewater.
Citation: Tian, M., Zhang, H., Liu, Y. et al. Breaking the oxo-wall for Co(IV)-oxo species and their nanoconfined catalytic performance within Ce-Co lamellar membrane. Nat Commun 17, 1767 (2026). https://doi.org/10.1038/s41467-026-68471-8
Keywords: water purification, advanced oxidation, catalytic membrane, cobalt oxo chemistry, micropollutants