Clear Sky Science · en
Rigid ionic-bonding networks boosting organic room temperature phosphorescence
Glowing in the Dark, Without the Heat
Imagine a material that keeps glowing long after the lights go out, without needing rare metals or extreme cold. This study shows how chemists can coax common organic molecules to produce a long-lasting afterglow at room temperature by locking them into invisible “ionic cages.” Such materials could power next‑generation security inks, glowing displays, and medical imaging tools that work safely inside the body.
Why Afterglow Is Hard to Get
Long-lived glow, known as room‑temperature phosphorescence, depends on fragile excited states called triplet excitons. In ordinary organic molecules, these states are hard to create and even harder to protect: they vanish when the molecules jiggle and collide at room temperature. Traditional approaches add heavy atoms like bromine directly to the glowing molecule or pack molecules tightly into crystals and polymers. These tricks can help, but they often demand painstaking molecular design, and each new color or application may require starting from scratch.
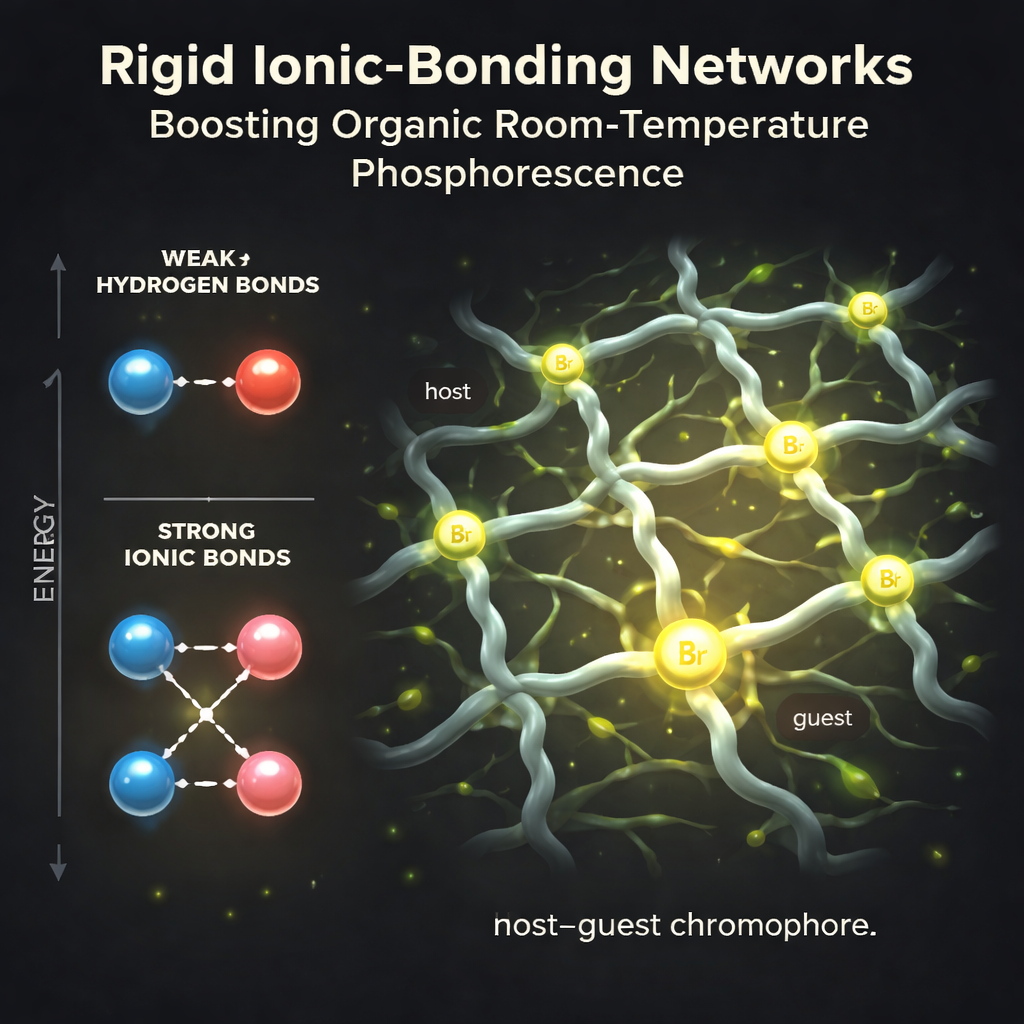
Building a Rigid Ionic Cage
The authors tackle this problem by separating the roles of “glow” and “structure.” They design a family of flexible host molecules made from simple alkyl (carbon) chains tipped with charged ammonium groups and counterions such as bromide or chloride. Into this host, they dissolve tiny amounts of brightly emissive guest molecules that carry a matching charged tail. When the solvent is removed, the positive and negative ions from host and guest attract each other strongly, assembling into a rigid ionic network. The host provides a stiff framework, while the guests act as light‑emitting centers held in place like bulbs in a lattice.
Matching Chains for Maximum Glow
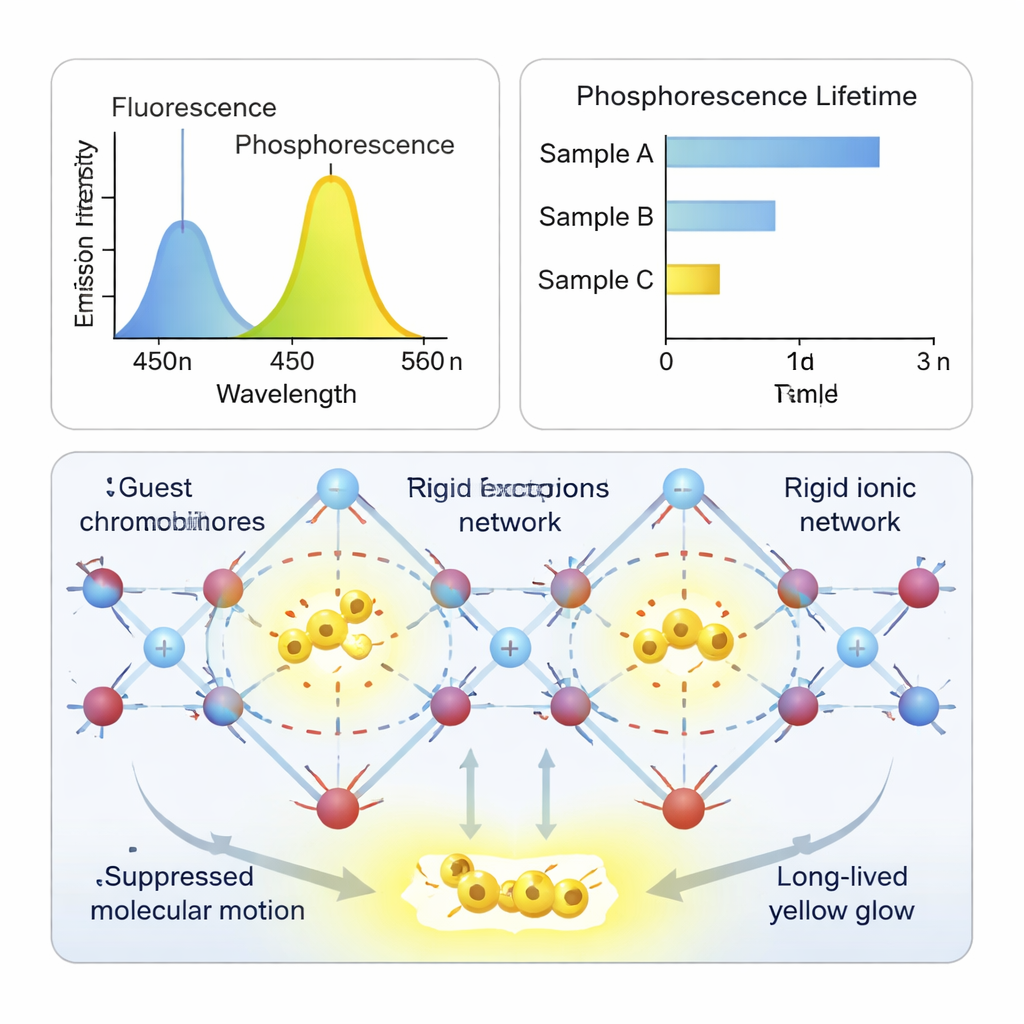
By carefully tuning the length of the alkyl chains on host and guest, the team found they could create highly ordered networks that best immobilize the glowing molecules. When the chains match, the ionic nodes line up and form an organized, cross‑linked structure. Single‑crystal X‑ray measurements reveal that bromide ions sit at key junctions, and the guest molecules are further pinned by close contacts between hydrogen, oxygen, and bromine atoms. This rigid environment suppresses energy‑wasting vibrations and prevents the guests from clumping together in ways that would quench the glow.
Heavy Atoms Without Heavy Design
The ionic framework does more than just hold the guests still. Bromide ions at the ends of the host chains act as “external heavy atoms,” subtly enhancing the conversion of ordinary excited states into the triplet states that power phosphorescence. Control experiments show how crucial these features are: if the guest lacks charge, if the host is not ionic, or if the bromide is replaced by less effective partners, the long‑lived glow weakens or disappears. In the optimized system, the researchers achieve bright yellow afterglow visible to the naked eye and measure lifetimes up to about half a second or more—an impressive span for purely organic materials.
Tuning Colors and Hiding Messages
Because the host framework is largely the same for different guests, the team can swap in various phosphorescent molecules to cover colors from blue to orange‑red while still benefiting from the same ionic cage. Lifetimes can be adjusted from a few milliseconds to over half a second, simply by changing the guest. To demonstrate real‑world potential, the authors press the powders into thin tablets and pattern them with masks. Under ultraviolet light, shapes like maple leaves or numbers appear; when the light is turned off, hidden afterglow images emerge, acting as simple encryption or anti‑counterfeiting features. They even use a solution of charged guests like an “ink” that activates afterglow only where it touches the ionic host film.
What This Means for Everyday Technology
In essence, the researchers show that you don’t need exotic chemistries to get stable, long‑lasting glow at room temperature. By using strong ionic bonds to build a rigid cage and strategically placing heavy ions at the right spots, they create a universal platform that works with many different glowing molecules. For non‑experts, the takeaway is straightforward: if we can reliably lock light‑emitting molecules into these ionic networks, it becomes much easier to design safer, tunable, and low‑cost afterglow materials for security printing, displays, and bio‑friendly imaging tools.
Citation: Ye, W., Huang, C., Lv, A. et al. Rigid ionic-bonding networks boosting organic room temperature phosphorescence. Nat Commun 17, 1759 (2026). https://doi.org/10.1038/s41467-026-68468-3
Keywords: room temperature phosphorescence, ionic bonding networks, organic afterglow materials, host guest systems, security inks