Clear Sky Science · en
H3K27me3-dependent imprinting and transcriptional regulation in early mouse embryos requires EZHIP-mediated restriction of PRC2 activity
Why tiny embryos need careful genetic timing
Every mammal begins life as a single cell that carries one set of chromosomes from the mother and one from the father. Although these two genetic packages contain almost the same DNA letters, they arrive wrapped in different chemical tags that help decide which genes turn on or off. This study explores how one such tagging system is kept in balance during the first days after fertilization in mice, and why disturbing that balance can derail normal development.
Two parents, two differently packed genomes
In mammals, certain genes behave differently depending on whether they come from the mother or the father, a phenomenon called genomic imprinting. Classic imprinting relies on chemical marks on DNA itself, but in recent years scientists discovered a second, more short-lived type that depends on a modification of histone proteins called H3K27me3. These histones act like spools around which DNA is wound, and H3K27me3 tends to shut nearby genes down. In normal mouse embryos, the maternal genome carries more of this mark than the paternal genome right after fertilization, helping to silence specific maternal gene copies while allowing paternal copies to work.
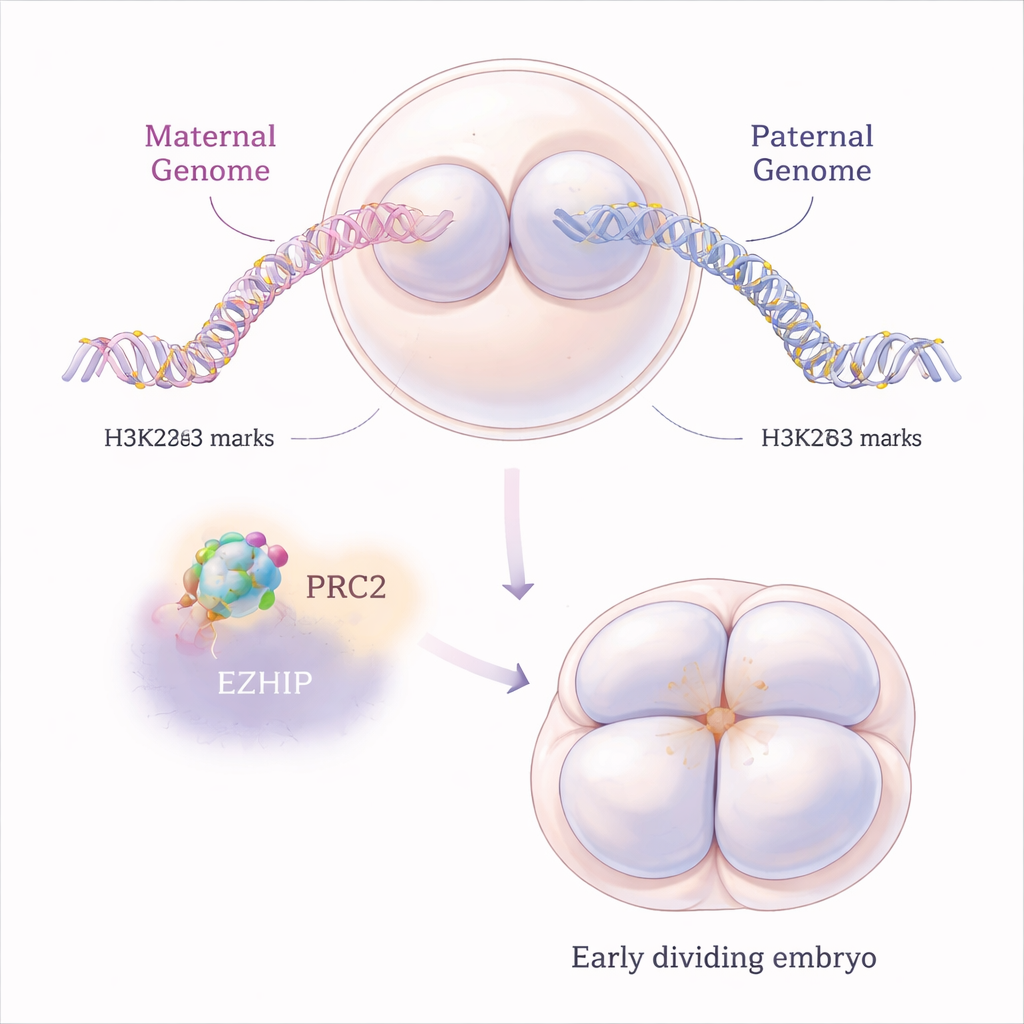
A molecular brake on a powerful repressor
H3K27me3 is placed on chromatin by a large protein machine called PRC2. The authors focused on EZHIP, a lesser-known protein that binds PRC2 and slows it down. Mouse egg cells produce a lot of Ezhip RNA, and the resulting EZHIP protein lingers in embryos through the first few cell divisions. By studying mice whose mothers lack Ezhip, the researchers asked what happens when this natural brake is removed. They found that, without EZHIP, eggs and early embryos gained extra H3K27me3 marks, but in a surprisingly “flattened” way: instead of sharp, well-defined domains, the repressive mark spread more broadly and blurred normal patterns, affecting both maternal and paternal chromosomes.
When too much repression breaks imprinting
Using sensitive genome-mapping methods, the team showed that the usual asymmetry between the maternal and paternal H3K27me3 landscapes is largely lost in embryos that develop from Ezhip-deficient eggs. Many regions that normally keep H3K27me3 on only one parental copy now gained it on both, or had their patterns smeared out. This change had important consequences for imprinted genes that are normally controlled by H3K27me3 rather than DNA methylation. In control embryos, these genes tend to be expressed mostly from the paternal allele. In embryos lacking maternal EZHIP, however, the same genes were often turned on from both parental copies, and overall RNA levels for many of them increased instead of being doubly silenced.
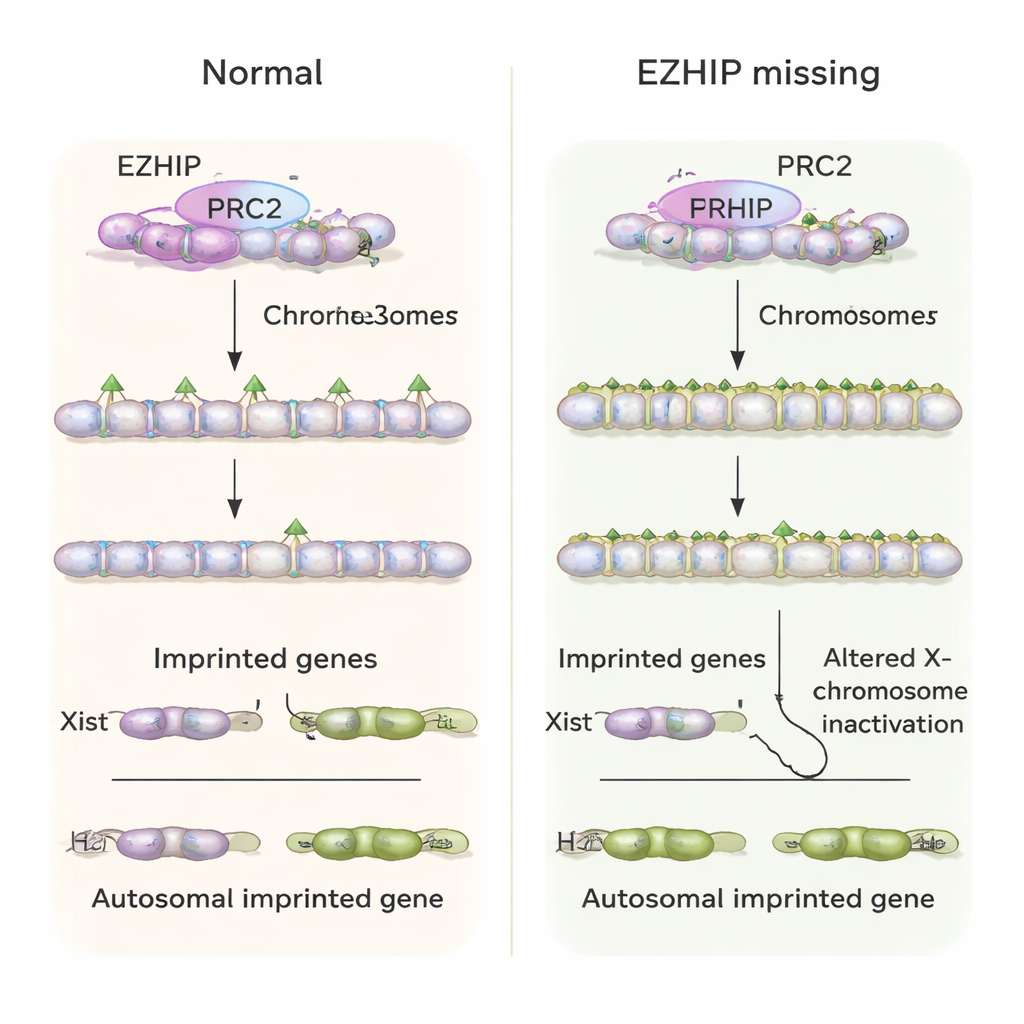
Disrupted X-chromosome control and embryo health
One key H3K27me3-dependent imprinted gene is Xist, a long RNA that coats and inactivates one X chromosome in female cells to balance gene dosage with males. Normally, early mouse embryos express Xist only from the paternal X in females, and not at all in males. In the absence of maternal EZHIP, the authors observed that female embryos frequently expressed Xist from both X chromosomes, while male embryos “inappropriately” turned on Xist from their single X. This led to abnormal inactivation patterns of X-linked genes. At the whole-animal level, mice born to Ezhip-deficient mothers were fewer in number, and embryos showed altered growth and problems in extraembryonic tissues that support the placenta, consistent with long-lasting developmental stress caused by early imprinting failure.
Too much of a good thing can be harmful
To a non-specialist, it may seem intuitive that more of a silencing mark like H3K27me3 would simply quiet more genes. Instead, this work shows that overloading the genome with such marks in the wrong pattern can actually undermine their ability to act as precise switches. By removing EZHIP, the researchers unleashed PRC2, causing H3K27me3 to spread so broadly that imprinting signals were blurred and critical regulators like Xist were miscontrolled. The study highlights that early embryos depend not just on having the right molecules, but on keeping their activities tightly tuned so that maternal and paternal genomes are read in the proper balance.
Citation: Diop, S., Richart, L., Petitalot, A. et al. H3K27me3-dependent imprinting and transcriptional regulation in early mouse embryos requires EZHIP-mediated restriction of PRC2 activity. Nat Commun 17, 1758 (2026). https://doi.org/10.1038/s41467-026-68467-4
Keywords: epigenetics, genomic imprinting, early embryonic development, X-chromosome inactivation, Polycomb PRC2