Clear Sky Science · en
Leveraging glucan-induced trained immunity for the epigenetic and metabolic rewiring of macrophages to enhance colorectal cancer vaccine response
Reawakening the Body’s First Responders
Colorectal cancer is one of the world’s deadliest cancers, and most patients do not benefit from today’s immunotherapies. This study explores an intriguing idea: what if we could “train” the innate immune system—the body’s first responders—to remember danger and help vaccines work better against colorectal tumors? By using a natural sugar from yeast, called beta‑glucan, the researchers show how early immune cells can be rewired so that cancer vaccines trigger stronger, longer‑lasting attacks on tumors.
Why Colorectal Cancer Needs New Defenses
Colorectal cancer causes nearly a million deaths each year worldwide, and cases in younger adults are rising. Standard treatments such as surgery, chemotherapy, and radiation can be effective, but many patients relapse and experience serious side effects. Newer immune‑based drugs, like checkpoint inhibitors, work very well only in a small subset of colorectal cancers that carry special DNA repair defects. For the majority of patients, these drugs and even experimental cancer vaccines fall short because the tumor environment is dominated by cells and signals that dampen the immune attack.
Teaching Innate Immunity New Tricks
The body’s innate immune cells—especially macrophages—normally react quickly but briefly to threats and are thought to lack memory. However, work over the past decade has revealed “trained immunity,” where a strong initial stimulus leaves lasting marks on these cells, allowing them to respond more vigorously later. The authors used beta‑glucan, a component of yeast cell walls, to induce this trained state. In mice, brief treatment with whole beta‑glucan particles reprogrammed macrophages at the level of both gene activity and cell metabolism. When these trained animals later received a colorectal cancer vaccine based on an engineered adenovirus (called PeptiCrad), their immune systems produced more potent killer T cells, which were better at recognizing and attacking tumor‑specific targets. 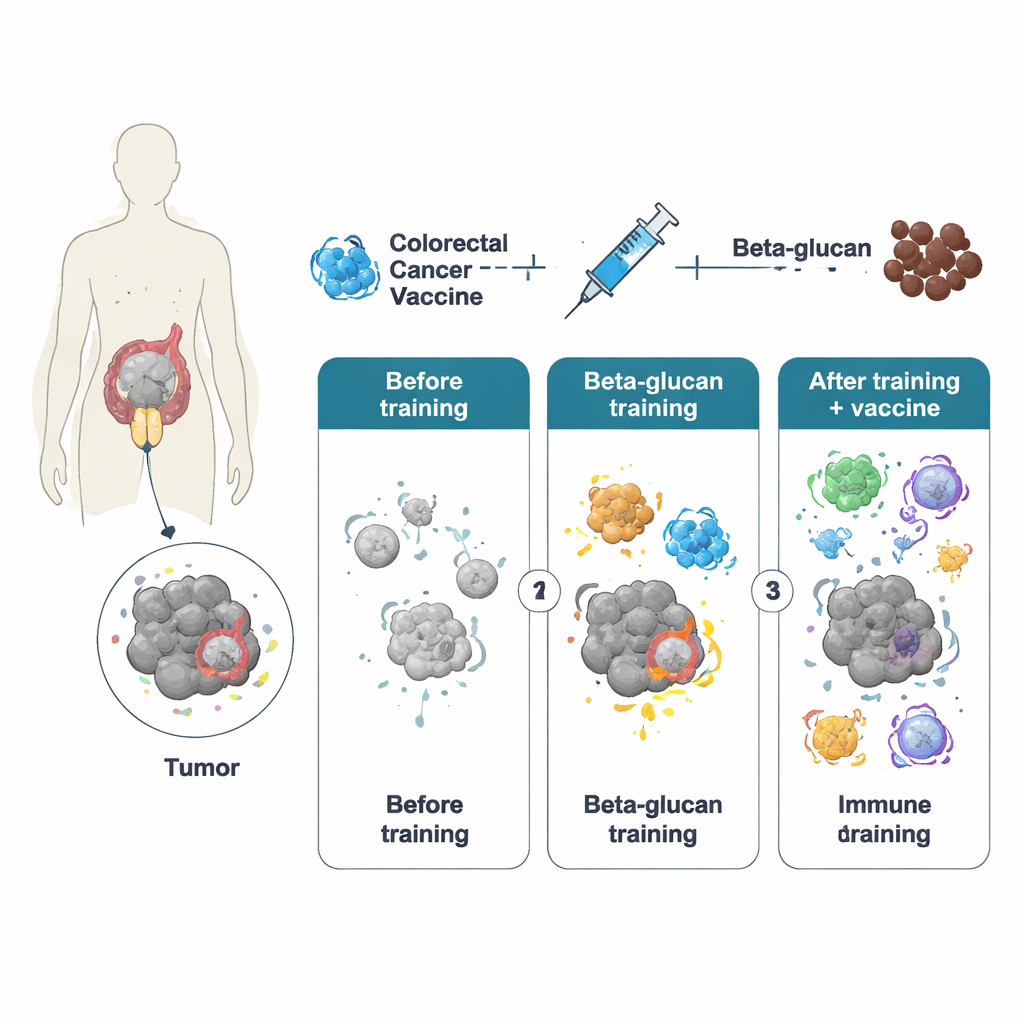
Rewiring Energy and Communication Inside Tumors
Digging deeper, the researchers isolated immune cells from tumors and profiled their metabolism and gene activity one by one. They found that trained macrophages switched into a high‑gear metabolic mode resembling the “Warburg effect,” where cells burn sugar rapidly and divert resources into building blocks for growth and function. These cells also boosted creatine metabolism—better known from sports supplements—which helped supply the extra energy needed to produce inflammatory molecules. At the same time, specific chemical tags on DNA‑associated proteins (notably an increase in H3K4me3) marked genes involved in chemokine production, effectively locking in a pro‑inflammatory, tumor‑fighting program that persisted over time.
Building an Immune Relay Team
Single‑cell sequencing revealed a stepwise relay among different immune cells inside the tumor. First, trained macrophages released high levels of two key signals, CXCL9 and CXCL10, which attracted natural killer (NK) cells through a receptor called CXCR3. Once recruited, these NK cells did not just kill cancer cells; they also secreted another messenger, CCL5, that drew in a specialized dendritic cell subset known as cDC1. These dendritic cells excel at presenting tumor pieces to T cells. As a result, more CD8 T cells became activated and shifted into effector memory cells—cells that respond quickly and robustly when they reencounter the tumor. Blocking creatine use, depleting macrophages or NK cells, or interrupting these chemokine pathways weakened tumor control, underscoring how dependent vaccine success was on this macrophage–NK–dendritic cell–T cell circuit. 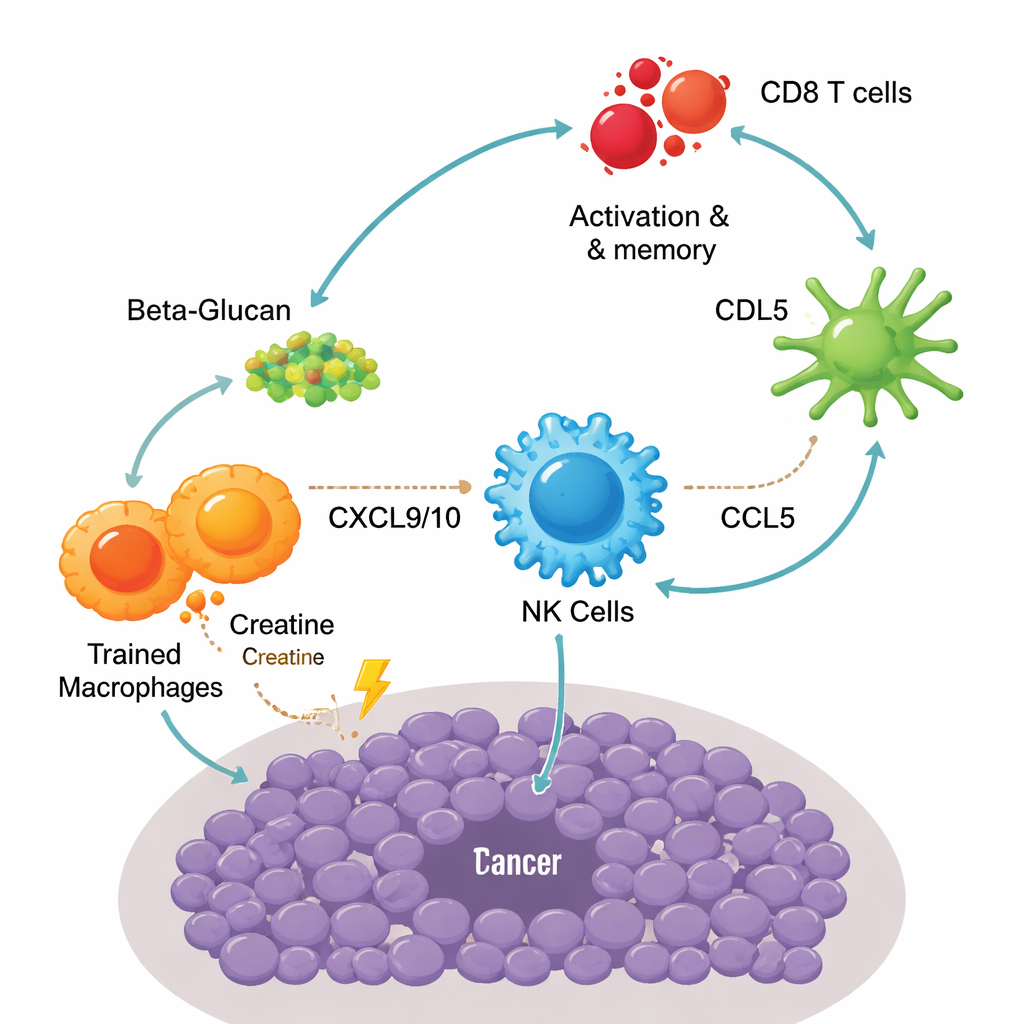
From Mouse Models to Human Tumor Organoids
The team tested their approach in several mouse models of colorectal cancer, including a particularly stubborn type that resists common immunotherapies. In each case, combining beta‑glucan training with the vaccine slowed tumor growth more effectively than either treatment alone. Importantly, they also moved beyond mice. Human blood cells exposed to beta‑glucan became better at responding to a colorectal cancer vaccine peptide, producing more immune‑stimulating cytokines. When these trained immune cells were expanded and co‑cultured with miniature patient‑derived colorectal tumors (organoids), they killed more cancer cells than non‑trained cells. Removing human monocytes and macrophages from the system erased this benefit, confirming that these trained cells are essential organizers rather than direct tumor killers.
What This Could Mean for Future Cancer Vaccines
In simple terms, this work shows that a safe, yeast‑derived sugar can “coach” innate immune cells to support more effective colorectal cancer vaccination. By rewiring how macrophages use energy and which genes they keep ready for action, beta‑glucan training sets off a domino effect: macrophages call in NK cells, NK cells recruit expert dendritic cells, and together they build a stronger army of memory CD8 T cells. While much remains to be tested in clinical trials—including safety, timing, and best vaccine combinations—trained immunity emerges here as a promising adjuvant strategy to turn more cold, suppressive colorectal tumors into ones that the immune system can recognize and control.
Citation: Hamdan, F., Gandolfi, S., D’Alessio, F. et al. Leveraging glucan-induced trained immunity for the epigenetic and metabolic rewiring of macrophages to enhance colorectal cancer vaccine response. Nat Commun 17, 1757 (2026). https://doi.org/10.1038/s41467-026-68466-5
Keywords: colorectal cancer immunotherapy, trained immunity, beta-glucan, macrophages and NK cells, cancer vaccines