Clear Sky Science · en
In vivo CRISPR screening identifies SAGA complex members as key regulators of hematopoiesis
Why keeping blood production in balance matters
Every day, your body quietly manufactures hundreds of billions of new blood cells that carry oxygen, fight infections, and stop bleeding. This nonstop production depends on rare "mother" cells in the bone marrow, called blood-forming stem cells. When the machinery that controls these cells goes wrong, people can develop anemia, weakened immunity, or cancers such as leukemia. This study asks a simple but powerful question: among the thousands of genes in our genome, which ones are truly essential to keep blood cell production healthy and balanced?
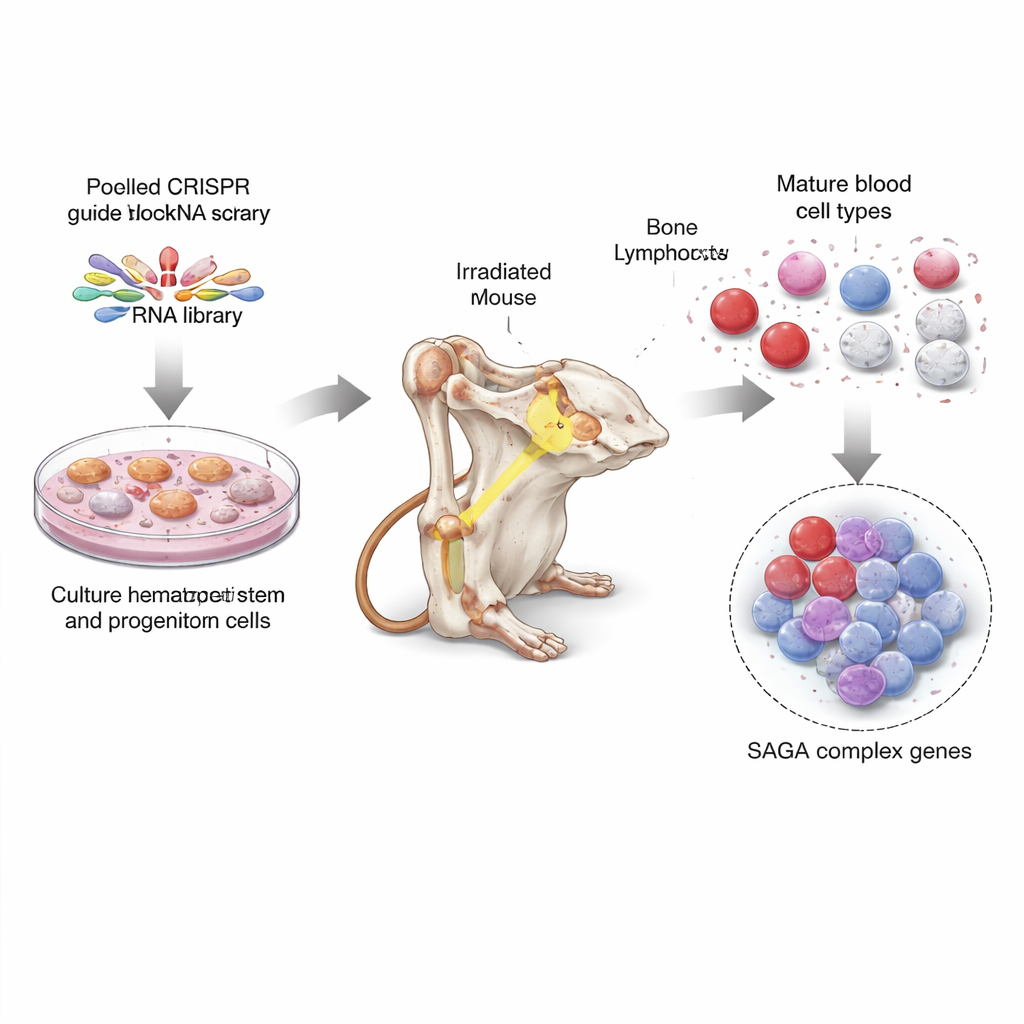
A genome-wide treasure hunt in mouse stem cells
To tackle this, the researchers used CRISPR, the gene-editing tool often likened to molecular scissors, to switch off almost every gene in the mouse genome, ten guides per gene. They first grew large numbers of bone-marrow stem and progenitor cells in special culture conditions, then infected these cells with pooled CRISPR guides so that each cell carried a knockout of a different gene. Next, they transplanted these mixed, edited cells into dozens of irradiated mice, allowing the edited cells to rebuild the animals’ blood systems. Months later, they purified various mature blood cell types and stem-cell–like cells from the bone marrow and read out which CRISPR guides were over- or under‑represented. This revealed which gene losses favored immature cells, and which supported the production of fully formed blood cells.
A hidden control hub called SAGA comes into focus
Among many known regulators of blood formation, a surprising set of hits emerged: several structural pieces of a large protein assembly called the SAGA complex, including genes named Tada2b, Taf5l, and Tada1. SAGA does not make blood cells directly; instead, it helps turn other genes on or off by chemically marking DNA-packaging proteins called histones. When the team individually knocked out these SAGA components and transplanted the edited cells into mice, they saw a consistent pattern. Immature bone-marrow cells piled up, but their ability to mature into circulating white blood cells, red blood cells, and certain immune cells was sharply reduced. In essence, the stem and progenitor cells were stuck in a bottleneck, unable to complete the journey to fully functional blood cells.
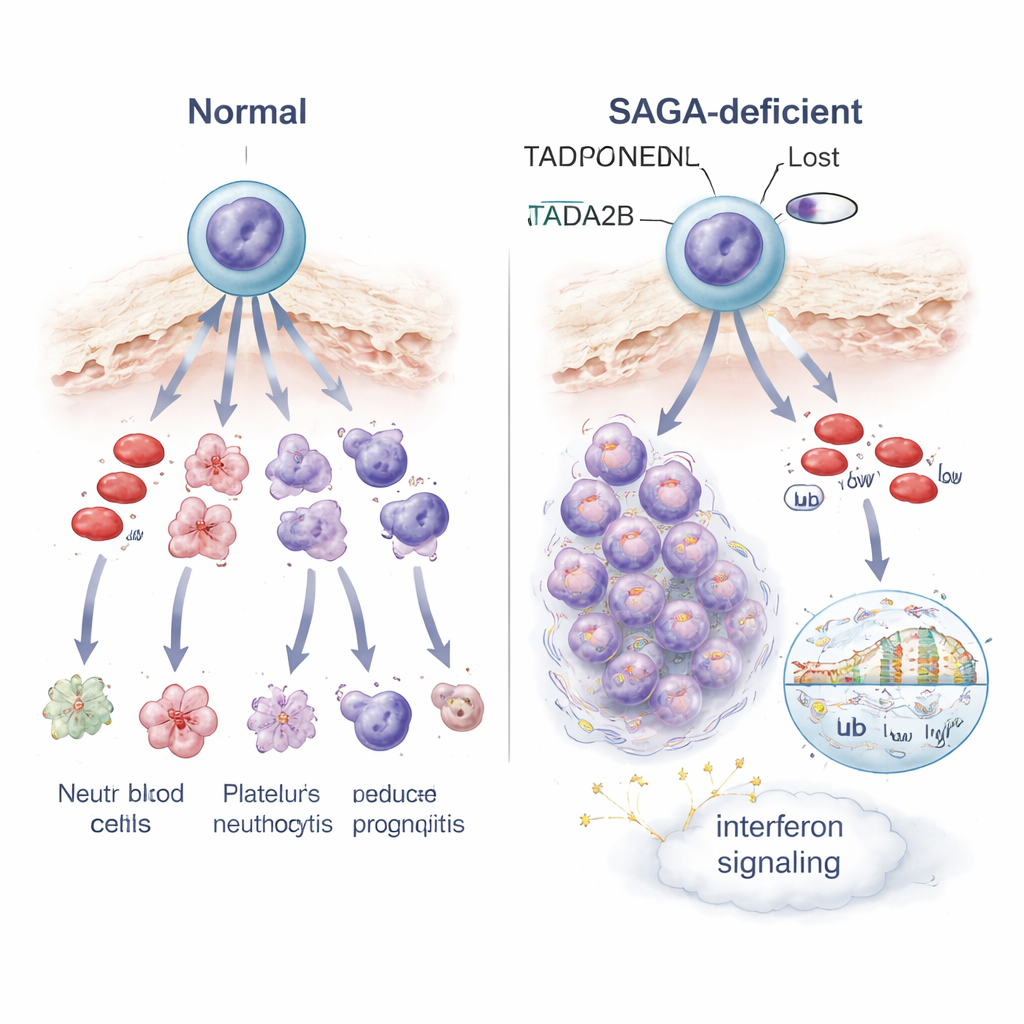
How disrupted gene control alters cell fate and stress signals
To understand what goes wrong inside these blocked cells, the scientists profiled gene activity one cell at a time and in bulk. Loss of Tada2b or Taf5l rewired the activity of many genes in stem cells, with two striking themes. First, genes involved in interferon signaling—normally part of the body’s antiviral defense—were switched on, indicating an internal alarm or stress state. Second, the cells’ mitochondria, the tiny power plants that fuel cellular work, became less active, and the altered cells grew more sensitive to a drug that targets mitochondrial energy production. At the level of DNA packaging, cells lacking Tada2b showed reduced levels of a histone mark linked to open, active chromatin, and changes in another mark tied to how genes are read. These shifts in chromatin chemistry likely help explain why many genes, including those that guide proper blood maturation, become misregulated.
From basic mechanisms to disease models
The team then asked whether dialing SAGA activity up or down could reverse these effects. Forcing cells to make extra Tada2b lowered interferon-related gene activity and reduced the proportion of stem-like cells, the mirror image of the knockout. Treating normal stem cells with a drug that inhibits SAGA’s key enzymatic subunits reproduced much of the Tada2b-loss signature, reinforcing the idea that SAGA’s chromatin-modifying activity is central to its role. Finally, the researchers turned to a human cell model of myelodysplastic syndrome, a pre-leukemia disorder in which blood production falters and interferon pathways are often elevated. When they knocked out SAGA components in these human cells and transplanted them into immunodeficient mice, the edited cells gained a growth advantage and again lit up interferon and myeloid gene programs, suggesting that this regulatory hub also shapes disease behavior.
What this means for blood health and future therapies
For a lay reader, the core message is that this study reveals a powerful control center— the SAGA complex—that helps blood-forming stem cells decide when to stay immature, when to mature, and how to avoid harmful stress responses. When key SAGA parts go missing, stem cells accumulate but fail to produce enough working blood cells, while stress and antiviral-like signals rise and mitochondria falter. These discoveries not only deepen our understanding of how healthy blood production is maintained over a lifetime, but also hint that subtle changes in SAGA activity could contribute to age-related blood problems and disorders like myelodysplastic syndrome. In the long run, mapping this control circuitry could help scientists design therapies that nudge faulty stem cells back toward balanced, resilient blood production.
Citation: Shankar, A., Olender, L., Hsu, I. et al. In vivo CRISPR screening identifies SAGA complex members as key regulators of hematopoiesis. Nat Commun 17, 1756 (2026). https://doi.org/10.1038/s41467-026-68465-6
Keywords: hematopoietic stem cells, CRISPR screening, SAGA complex, interferon signaling, myelodysplastic syndrome