Clear Sky Science · en
Intranasal administration of broad-spectrum macrocyclic peptide inhibitor protects against SARS-CoV-2 Omicron variants
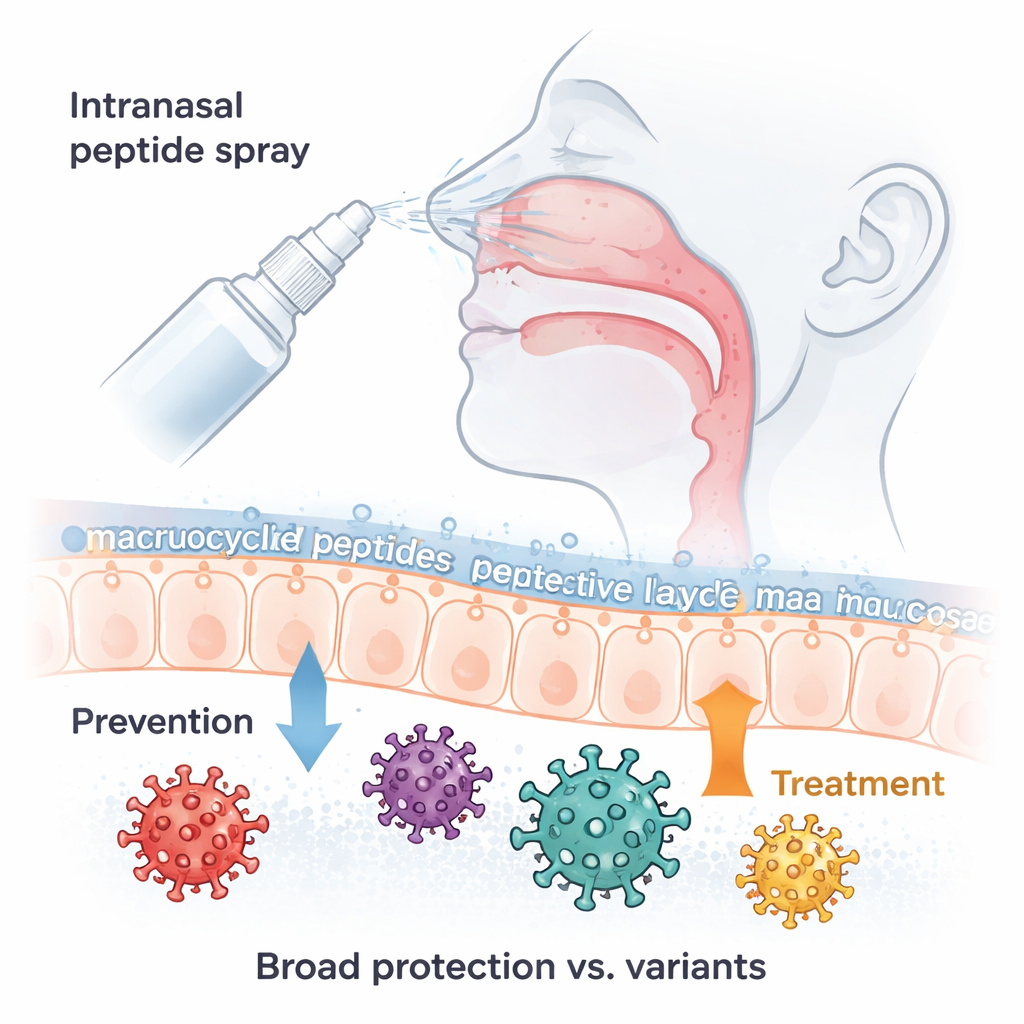
Why a nasal spray for COVID still matters
Even though the COVID-19 pandemic emergency has passed, the virus that causes it, SARS-CoV-2, continues to infect and kill people worldwide, especially older adults and those with weak immune systems. New variants such as Omicron keep evolving ways to dodge our vaccines and antibody drugs. This study describes a new kind of medicine: a tiny, ring-shaped molecule delivered as a simple nasal spray that can block a wide range of Omicron and other SARS-CoV-2 variants before they take hold, and can also help treat infection after it has started.
Turning designer rings into virus blockers
The researchers focused on macrocyclic peptides—small, ring-shaped protein fragments already viewed as promising drug candidates because they can grip their targets tightly, slip into tissues, and are relatively easy to manufacture. Using a powerful screening platform called the RaPID system, they built and tested huge libraries of such rings to find those that latch onto a critical part of the coronavirus spike protein, the receptor-binding domain (RBD). From this search they identified a lead compound called 6L3 and then improved it stepwise through precise changes in its amino acid building blocks, eventually creating much more potent variants named 6L3-3P and 6L3-3P11K that strongly block infection by many Omicron subvariants in cell cultures.
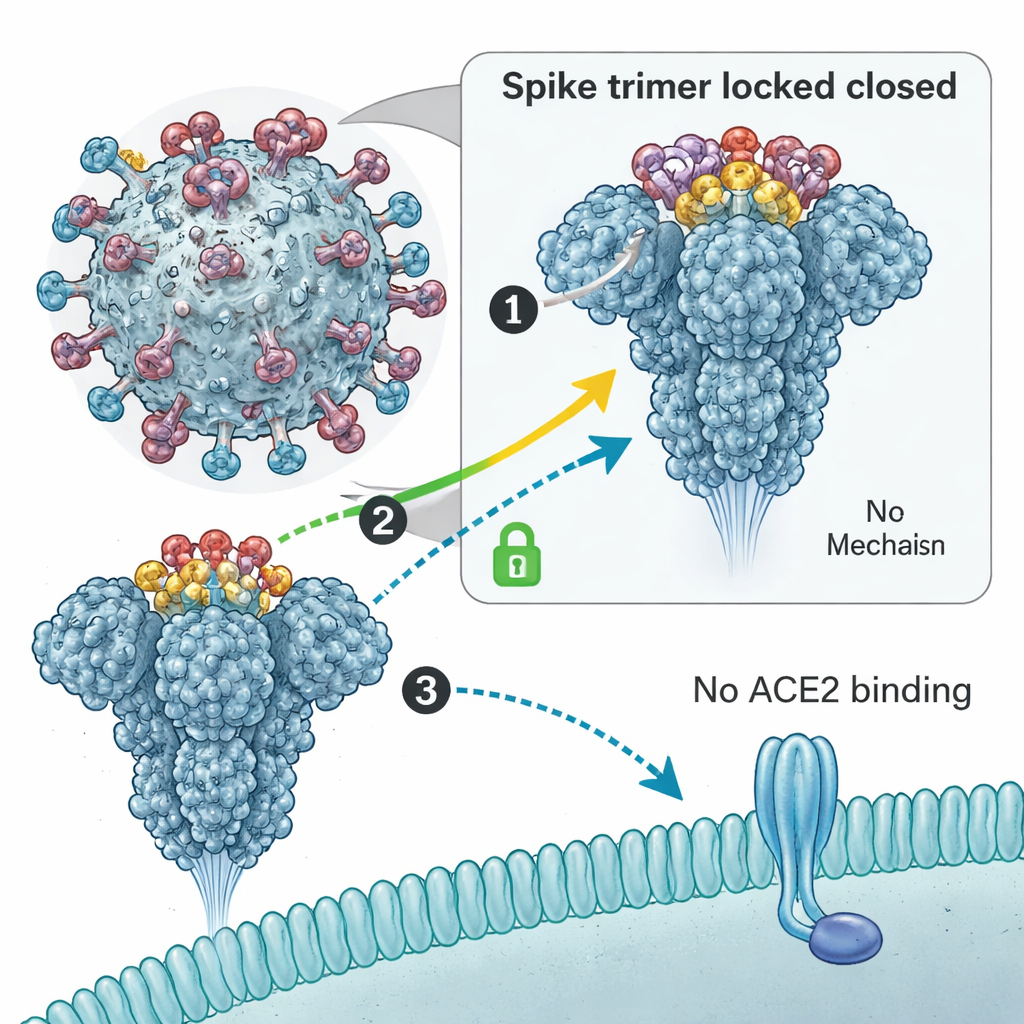
Gluing the spike shut
To understand how these rings stop the virus, the team used cryo–electron microscopy to visualize them bound to the spike protein in high detail. They found that three copies of the macrocyclic peptide come together as a small trimer that plugs into a cavity at the top of the spike made of three RBDs. This acts like molecular glue, locking all three RBDs into a “down” or closed position. In this posture, the spike cannot flip one of its RBDs “up” to grab the ACE2 receptor on our cells—a necessary first step for infection. Biophysical measurements confirmed that when the peptide is present, spike proteins lose their ability to bind ACE2. Importantly, the peptide targets a conserved region that is not the usual antibody binding site, meaning that common mutations in variants have so far left this pocket largely unchanged.
From lab dish to human-like tissue and mice
Armed with this structural insight, the scientists fine-tuned the molecule to make it both stronger and more stable in the body. A final optimized version, 6L3-1F3P11hR, resists breakdown by enzymes, tolerates heat and different acidity levels, and remains mostly in the nasal passages when sprayed into mice, minimizing exposure to the rest of the body. In lab-grown human nasal organoids—three-dimensional cultures that mimic real nasal lining—this peptide sharply reduced replication of recent Omicron variants even at low doses. In genetically engineered mice that express human ACE2 and develop severe COVID-like lung disease, intranasal dosing with the peptide either shortly before or after infection cut virus levels in the nose and lungs, protected lung tissue from damage, and performed comparably to the approved antiviral pill nirmatrelvir (part of Paxlovid) in these tests.
Staying ahead of future variants
Because the peptide binds a highly conserved “non-receptor-binding” patch of the spike and works through a physical locking mechanism rather than by targeting one viral enzyme, it may be harder for the virus to escape without harming its own ability to infect cells. The authors also show that simple point changes in the peptide sequence can tune its strength and spectrum, suggesting a path to adapt the drug against related coronaviruses in the future. Their pharmacokinetic studies indicate that the compound concentrates where the virus first lands—the nasal passages—making it a logical candidate for a preventive spray or early treatment that could be taken at home.
What this could mean for everyday protection
For non-specialists, the bottom line is that this work delivers a well-characterized, nose-delivered antiviral candidate that physically clamps the coronavirus spike shut, blocking it from docking onto our cells. In cells, in human-like nasal tissues, and in a susceptible mouse model, the optimized macrocyclic peptide sharply lowered virus levels and lung damage across many Omicron subvariants and some earlier strains. While human trials are still needed, this strategy points toward a future where a shelf-stable nasal spray could offer on-demand protection and treatment against current and emerging variants of SARS-CoV-2, and possibly other related coronaviruses.
Citation: Wang, M., Yang, J., Tan, Y. et al. Intranasal administration of broad-spectrum macrocyclic peptide inhibitor protects against SARS-CoV-2 Omicron variants. Nat Commun 17, 1753 (2026). https://doi.org/10.1038/s41467-026-68462-9
Keywords: COVID-19, SARS-CoV-2 variants, nasal spray antiviral, spike protein inhibitor, macrocyclic peptide