Clear Sky Science · en
Neural crest cell-derived DKK1 and NEDD4 modulate Wnt signalling in the second heart field to orchestrate outflow tract development
Why tiny heart builders matter
The early embryo’s heart starts as a simple tube, but it must quickly remodel into a complex organ that sends blood to both the body and the lungs. Subtle missteps in this building process can cause serious birth defects, including conditions that require surgery soon after birth. This study uncovers how two groups of cells talk to each other while shaping the heart’s main exit pathway, and reveals a molecular error that can contribute to congenital heart disease in both mice and humans.
Two teams building the heart’s exit ramp
The portion of the heart that carries blood out of the ventricles—the outflow tract—forms from a region called the second heart field. Cells here must stay as flexible “progenitors” long enough to be added to the growing outflow tract, and then switch into mature muscle at the right time. Just beside them travel neural crest cells, a roaming population that helps build the great arteries and valves. Earlier work suggested these neural crest cells somehow influence the second heart field, but how this cross-talk worked was unknown. 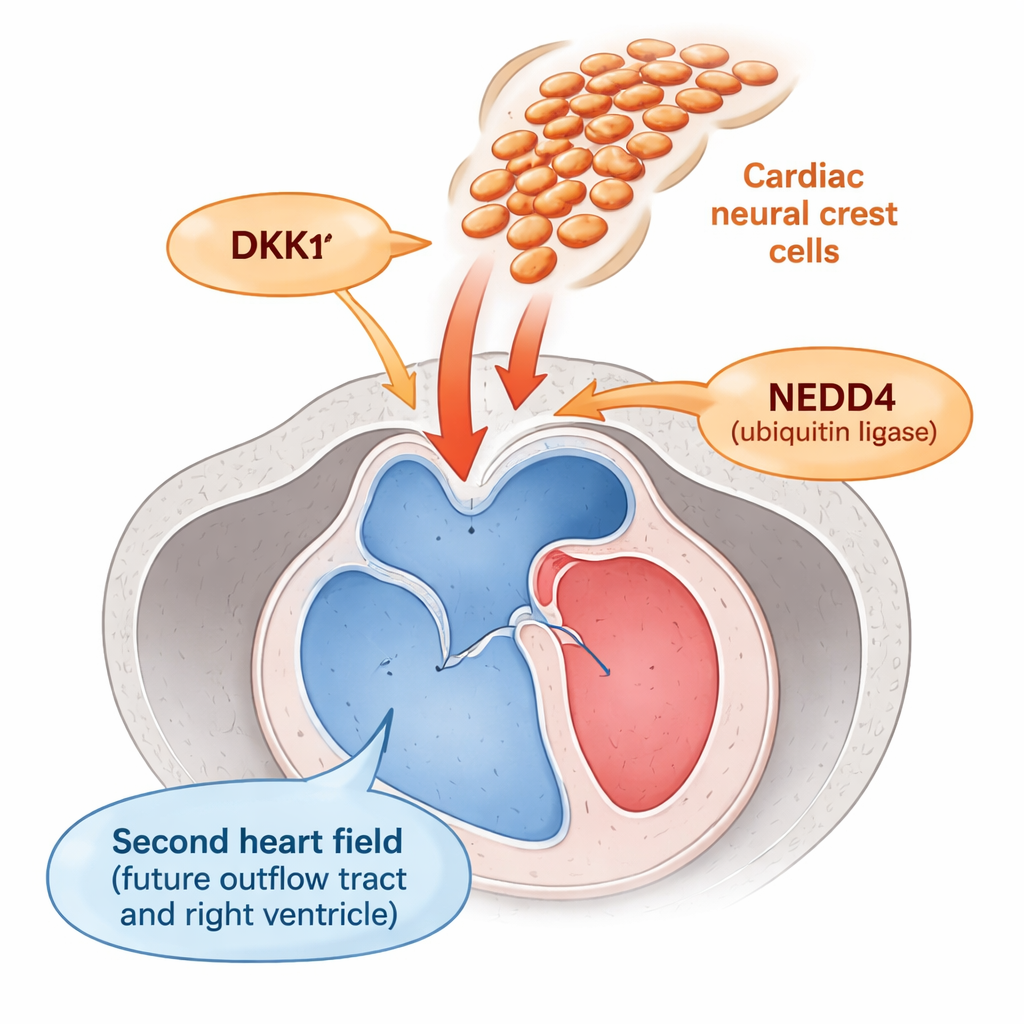
A molecular dimmer switch for growth signals
The authors discovered that neural crest cells act like a dimmer switch for a key growth signal called Wnt, which keeps progenitor cells dividing and delays their maturation. They found that neural crest cells are a major source of DKK1, a small secreted protein that shuts down Wnt signalling in nearby cells. DKK1 itself is normally kept in check by NEDD4, a protein that tags DKK1 for disposal. In mouse embryos lacking Nedd4 specifically in neural crest cells, DKK1 built up to abnormally high levels. This excess DKK1 dialed down Wnt activity in the neighbouring second heart field, as shown by reduced nuclear beta‑catenin—a standard readout of Wnt signalling—and lower levels of multiple Wnt‑responsive genes.
When timing goes wrong, the heart’s geometry fails
Too much DKK1 and too little Wnt had a clear consequence: second heart field cells started turning into heart muscle too soon. Markers of mature muscle appeared early in this progenitor zone, and there were fewer undifferentiated cells left to extend the outflow tract. By tracking dividing cells over time, the team showed that fewer second heart field cells were added into the outflow tract in mutants. As a result, the outflow tract was shorter and rotated incorrectly, leading to misalignment between the great arteries and the ventricles. These structural errors resembled human conotruncal defects such as double‑outlet right ventricle and related malformations seen in full Nedd4 knockout mice.
Proving the role of the signal, and a link to human disease
To confirm that altered Wnt signalling really caused these problems, the researchers nudged the pathway with drugs in pregnant mice. Blocking Wnt in otherwise healthy embryos pushed them toward the same premature differentiation and rotation defects seen when Nedd4 was lost, while reducing Dkk1 gene dosage in the Nedd4‑deficient background partially rescued outflow tract size and rotation. Finally, the team identified a child with Tetralogy of Fallot who carried a rare inherited change in NEDD4 that weakened its ability to tag DKK1 for breakdown. Mice engineered to carry the same Nedd4 variant developed right‑sided heart wall and septal defects, supporting the idea that faulty NEDD4–DKK1 control can contribute to human congenital heart disease. 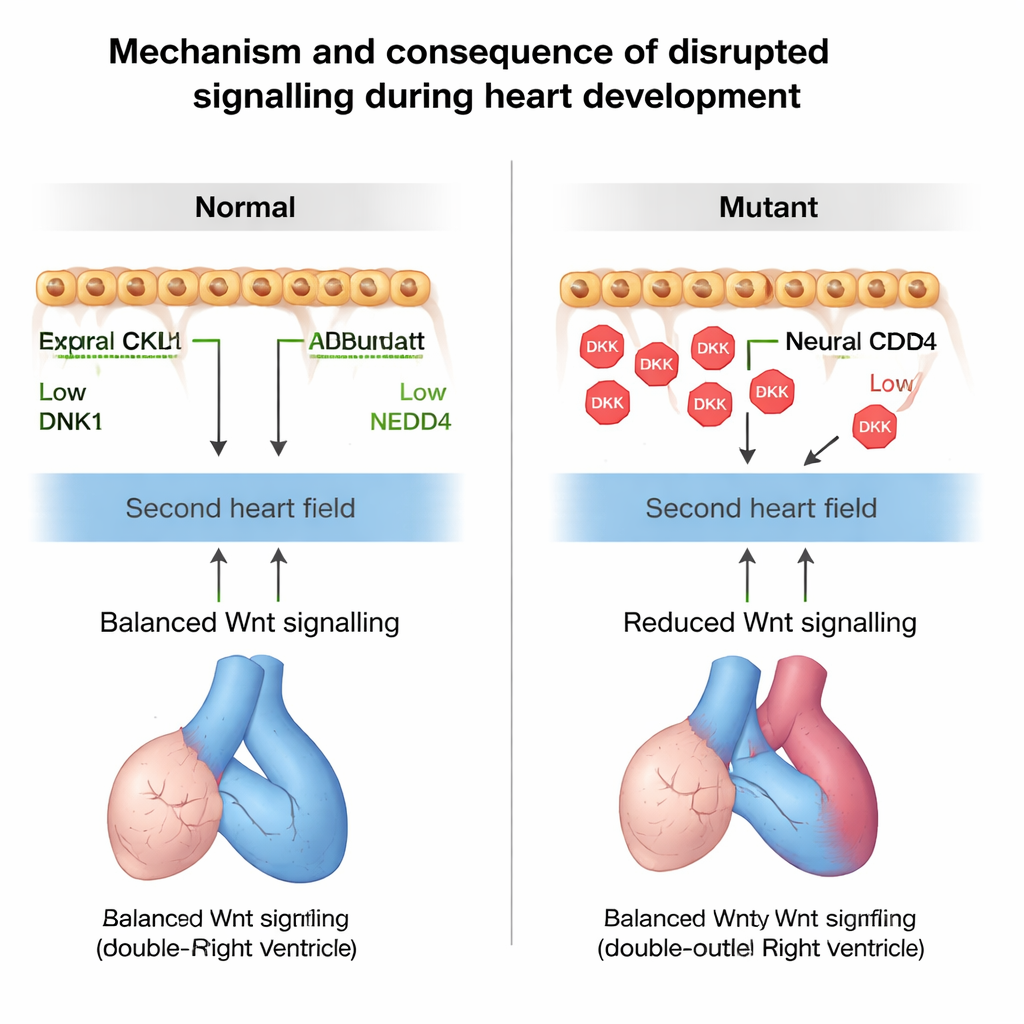
What this means for understanding heart defects
For non-specialists, the key message is that the heart’s “exit ramp” depends on precise timing of when its building blocks stop multiplying and start becoming muscle. This timing is controlled not just from within the heart tissue itself, but also by neighbouring neural crest cells that fine‑tune a growth signal via the NEDD4–DKK1 pair. When this molecular dimmer switch is stuck too low, the outflow tract is underbuilt and misaligned, leading to serious birth defects. By mapping this pathway and tying it to a human gene variant, the study offers new clues to how some congenital heart defects arise and highlights potential molecular targets for future diagnosis or intervention.
Citation: Wiszniak, S., Alankarage, D., Lohraseb, I. et al. Neural crest cell-derived DKK1 and NEDD4 modulate Wnt signalling in the second heart field to orchestrate outflow tract development. Nat Commun 17, 1751 (2026). https://doi.org/10.1038/s41467-026-68459-4
Keywords: congenital heart disease, heart development, neural crest cells, Wnt signalling, cardiac outflow tract