Clear Sky Science · en
Near-infrared-driven photocatalytic CO2 reduction to C2 hydrocarbons by bis(terpyridine)-metal functionalized lead halide frameworks
Turning Carbon Pollution into Useful Fuel
Most of the sunlight that reaches Earth arrives not as visible light, but as invisible near‑infrared glow. Yet today’s solar‑powered chemistry struggles to use that low‑energy light, especially for hard jobs like transforming carbon dioxide (CO2) into multi‑carbon fuels. This study reports a new class of solid materials that can soak up near‑infrared light and directly convert CO2 into valuable two‑carbon hydrocarbons such as ethylene and ethane, pointing toward more complete use of sunlight in future artificial photosynthesis technologies.
Why Near‑Infrared Light Matters
CO2 emissions from burning fossil fuels are a main driver of climate change, and more than 130 countries have pledged to reach carbon neutrality. One appealing approach is to use sunlight to turn CO2 back into energy‑rich molecules, closing the carbon loop. However, this strategy faces two big obstacles. First, about half of the sun’s energy lies in the near‑infrared region, which most photocatalysts cannot absorb efficiently. Second, even when CO2 is reduced, the products are usually one‑carbon molecules like carbon monoxide or methane, not more valuable multi‑carbon (C2+) hydrocarbons that industry relies on. The new materials described here are designed to tackle both problems at once by harvesting near‑infrared light and promoting the key step where two carbon‑containing fragments join to form a C–C bond.
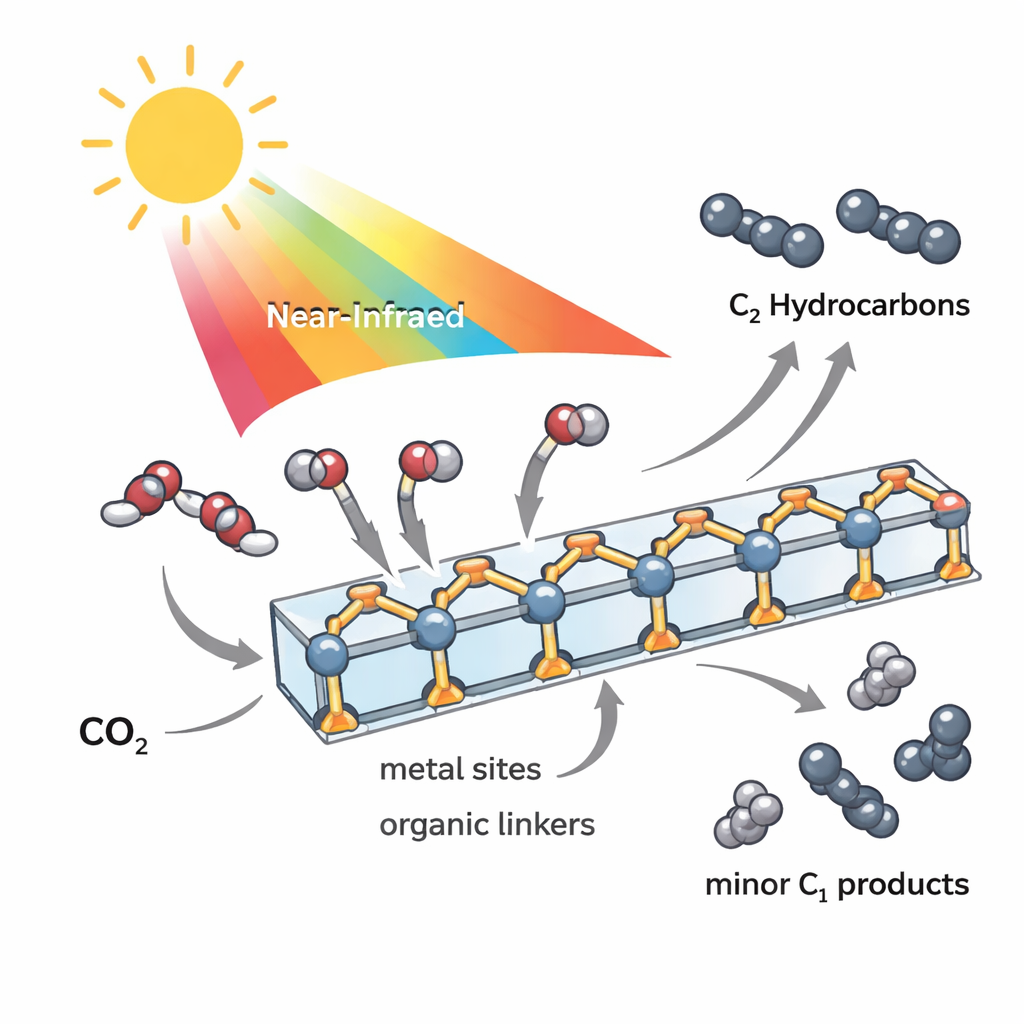
Building a Light‑Harvesting Crystal
The researchers started from hybrid lead halide materials, a family already known for strong light absorption and good charge transport, and then engineered them into robust, three‑dimensional frameworks. They linked tiny clusters of lead and halide ions (chloride, bromide, or iodide) with large “antenna” molecules built around iron, cobalt, or nickel. These organic antennas, based on terpyridine units, are excellent at absorbing light and holding excited electrons long enough for chemistry to happen. By connecting the metal‑halide clusters to the antennas through sturdy carboxylate bonds, the team created nine single‑component crystalline frameworks that remain stable in various solvents, across a range of acidity, and up to roughly 220 °C.
Catching More of the Sun’s Spectrum
Optical measurements showed that all nine frameworks absorb light from the ultraviolet through the visible and well into the near‑infrared, up to about 1150 nanometers. The iron‑based versions have the narrowest bandgaps, meaning they can use the lowest‑energy photons. Detailed electronic studies and computer calculations revealed that the organic antennas mainly supply the “starting” electronic states, while the lead‑halide units accept the excited electrons. When light hits, electrons move from the terpyridine units into the lead sites, helping separate charge and preventing wasteful recombination. The iodide‑based frameworks stand out further: the way solvent molecules bind to the lead iodide clusters creates an asymmetric local environment, polarizing the charge at neighboring lead sites and setting the stage for efficient C–C bond formation.
From Gas to Two‑Carbon Fuels
In photocatalytic tests, the materials were suspended in a CO2‑saturated solution and illuminated with a xenon lamp. The chloride and bromide versions mainly produced carbon monoxide and methane, and did not form detectable C2 products. In contrast, the iodide frameworks, especially the iron‑based TJU‑60(I)‑Fe(tpy)2, favored two‑carbon hydrocarbons: under full‑spectrum light they generated appreciable amounts of ethylene and ethane with high selectivity. Even under strictly near‑infrared light (wavelengths above 700 nm), where photons carry less energy, the same material still converted CO2 into a mixture of products dominated by C2 hydrocarbons, reaching C2 selectivity of up to 86% on an electron basis. Control experiments with isotopically labeled 13CO2 confirmed that all carbon in the products originated from CO2, and repeated reaction cycles showed that the crystals retained their structure while releasing only trace amounts of lead.
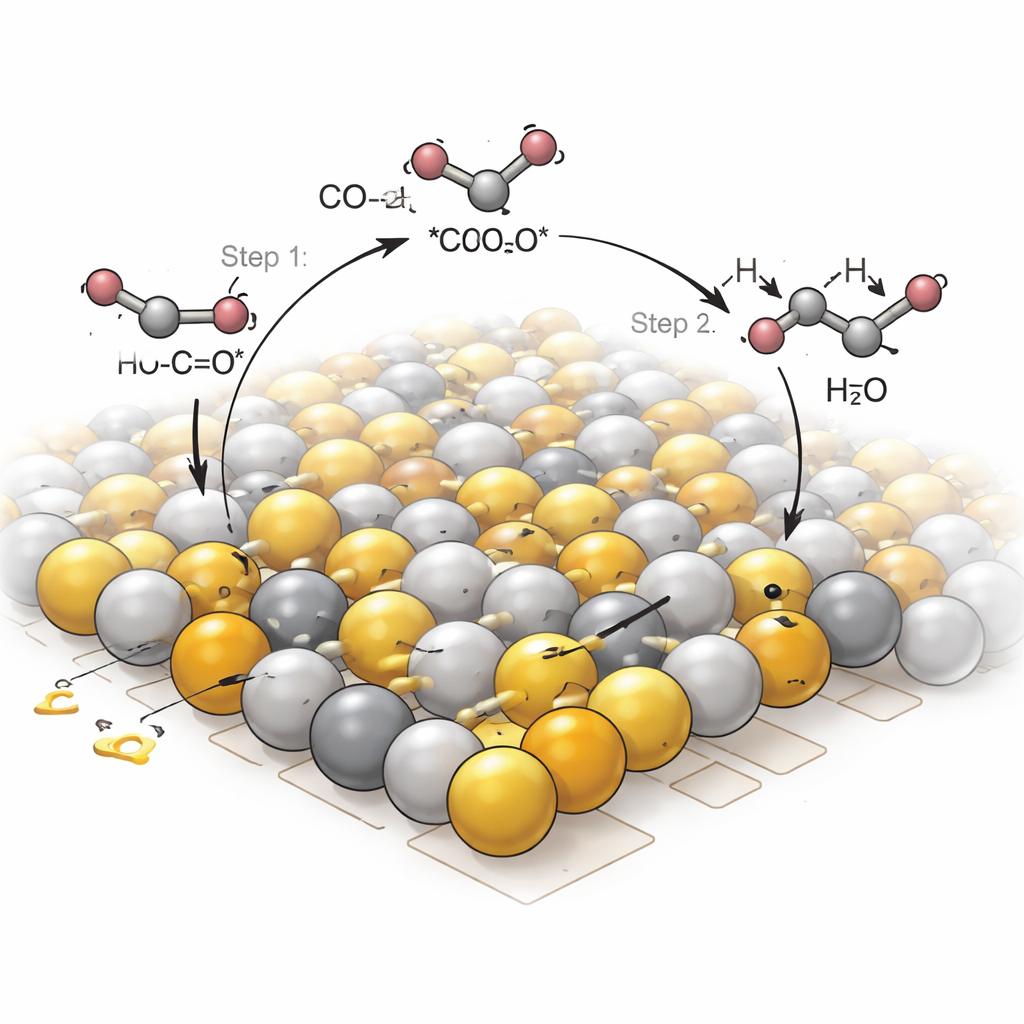
How the Material Guides the Reaction
To understand why the iodide frameworks behave so differently, the team combined advanced spectroscopy with quantum‑chemical modeling. They found that, after light absorption, electrons move from the terpyridine antennas to the lead iodide clusters, where they accumulate on two neighboring, but unequally charged, lead sites. CO2 molecules bind to these sites in a bent, activated form, with their bonds stretched and ready to react. Infrared measurements during reaction revealed a series of fleeting intermediates, including one in which two carbon‑based fragments join to form a *COCOH species—a hallmark of the first C–C bond. Calculations showed that the polarized pair of lead sites stabilizes both partners and lowers the energy barrier for this coupling step, steering the reaction toward two‑carbon products rather than stopping at single‑carbon gases.
What This Means for Solar Fuels
In simple terms, the researchers have built a crystal that acts like a tiny solar refinery, able to harvest not just visible light but also the weaker near‑infrared glow, and to channel that energy into stitching two carbon atoms together from CO2. By carefully shaping the local charge pattern around lead atoms, they turned a passive light absorber into an active site for carbon–carbon bond formation. While these materials are not ready for industrial deployment—and their lead content will require careful handling—they demonstrate a powerful concept: with smart molecular design, it is possible to tap nearly the full solar spectrum and selectively turn a greenhouse gas into more complex, energy‑rich fuels.
Citation: Li, Y., Wang, Z., He, X. et al. Near-infrared-driven photocatalytic CO2 reduction to C2 hydrocarbons by bis(terpyridine)-metal functionalized lead halide frameworks. Nat Commun 17, 1743 (2026). https://doi.org/10.1038/s41467-026-68450-z
Keywords: CO2 reduction, near-infrared photocatalysis, artificial photosynthesis, lead halide frameworks, C2 hydrocarbons