Clear Sky Science · en
Functional variants at 1p36.23 confer risk of schizophrenia through modulating RERE
Why tiny DNA changes matter for mental health
Schizophrenia is a serious mental illness that affects how people think, feel, and relate to others. It runs strongly in families, but most of the genetic changes involved are small, scattered tweaks in our DNA. This study zooms in on one such region of the genome and shows, step by step, how two subtle DNA variants can alter brain cell development and communication in ways that may raise a person’s risk of schizophrenia. 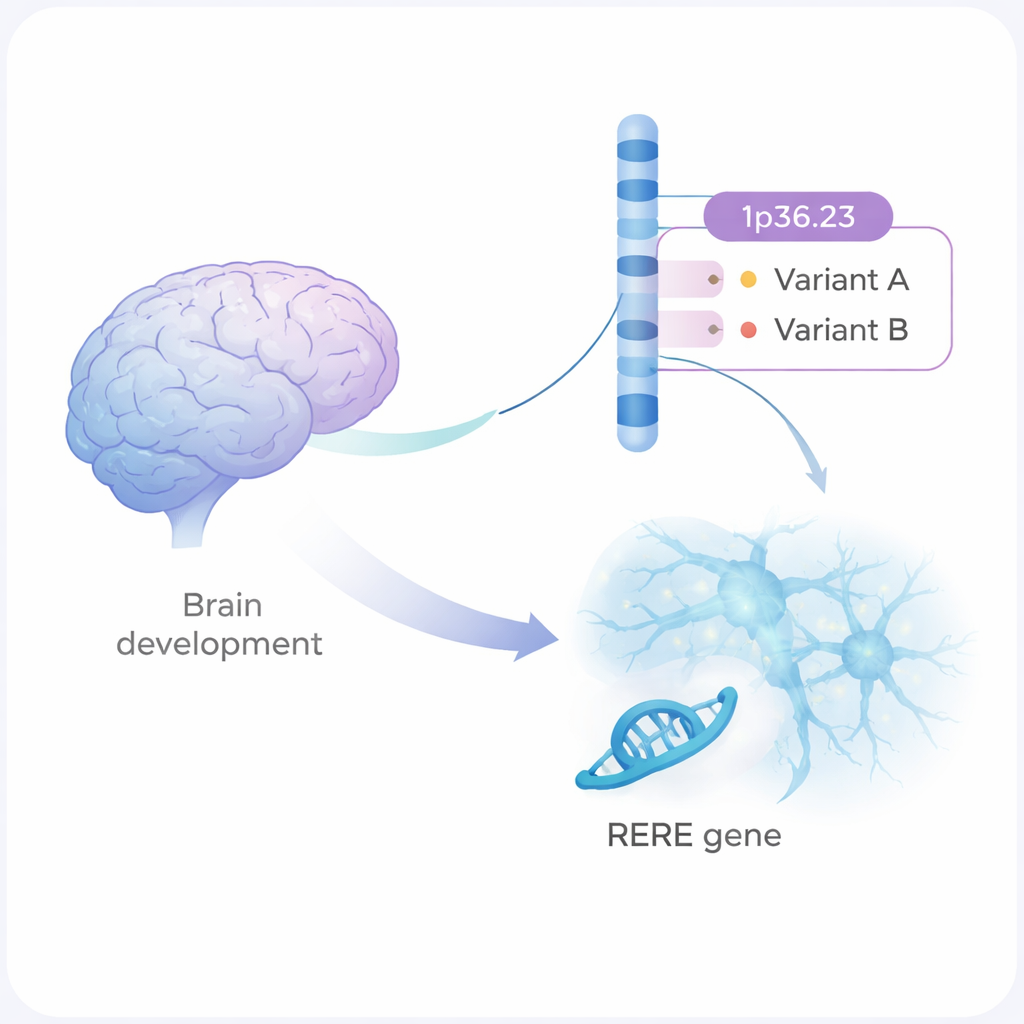
A genetic hot spot on chromosome 1
Large genetic studies have flagged more than 300 regions of the human genome that are linked to schizophrenia. One of these is a stretch of DNA on chromosome 1, called 1p36.23. Until now, scientists did not know which exact changes in this region, or which gene, were doing the real damage. The authors combined statistical genetics with laboratory experiments and singled out two DNA variants, named rs159961 and rs301792, sitting inside a gene called RERE. These variants do not change the RERE protein itself; instead, they sit in regulatory “switch” zones inside the gene that control how strongly RERE is turned on.
How risk variants turn up the volume on RERE
The team first asked whether these two variants actually act as functional switches. Using reporter assays—tests where a piece of DNA drives a light-producing gene—they showed that the schizophrenia-linked versions of rs159961 and rs301792 behave like stronger enhancers (boosters) in nerve-like cells, but not in unrelated cell types. Biochemical binding tests revealed why: the risk form of one variant weakens the grip of REST, a protein that normally damps down gene activity, while the risk form of the other strengthens binding of POLR2A, a core part of the gene-reading machinery. Together, these shifts in protein binding increase the activity of the enhancer segments and push RERE expression upward.
From boosted RERE to altered brain cell growth
Next, the researchers asked what extra RERE does inside the brain. They found that people who died with schizophrenia showed higher RERE levels in their brain tissue compared with unaffected individuals. To model this, they artificially raised RERE levels in mouse neural stem cells—the immature cells that give rise to neurons and glia. When RERE was overproduced, these stem cells divided less, got stuck in a late stage of the cell cycle, and produced fewer mature neurons while leaving other cell types mostly unchanged. In cultured neurons, too much RERE reshaped their branching patterns and reduced the number and type of tiny protrusions called dendritic spines, where synapses form. These changes fit with long-standing evidence that schizophrenia involves disrupted brain development and spine loss. 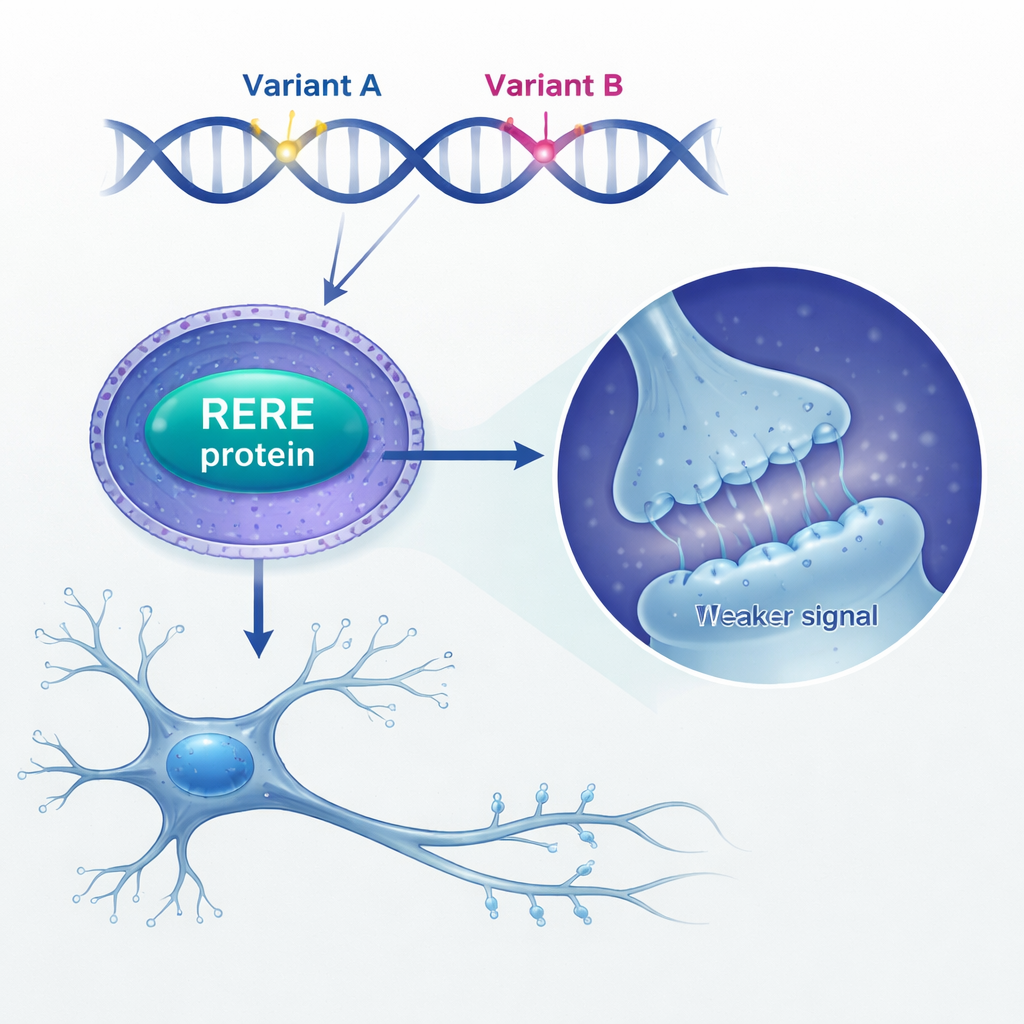
Disrupting the brain’s glutamate “conversation”
Looking more closely at gene activity, the team found that RERE overexpression disturbed networks of genes involved in dendrite growth and major chemical signaling systems, especially the glutamate pathway. One key target stood out: the gene GRIN2A, which encodes a crucial subunit (GluN2A) of NMDA-type glutamate receptors, long implicated in schizophrenia. The authors showed that RERE partners with two other nuclear proteins, RARB and RXRA, to bind directly to the GRIN2A promoter and dampen its activity. In neurons with excess RERE, GluN2A levels dropped and electrical recordings revealed weaker NMDA receptor–mediated synaptic currents, even though the frequency of synaptic events was unchanged. In other words, the “volume” of individual excitatory signals was turned down.
Connecting DNA variants to brain function
By weaving together genetics, molecular biology, cell culture, and electrophysiology, this work outlines a clear causal chain: risk DNA variants at 1p36.23 enhance regulatory elements inside the RERE gene, leading to higher RERE expression in brain cells. Elevated RERE in turn impairs the growth and maturation of neurons, alters the shape and number of their synaptic spines, and weakens glutamate-based signaling through NMDA receptors—especially those containing GluN2A. For a lay reader, the take-home message is that very small DNA changes can slightly shift the activity of a single gene, and over many brain cells and many years, this can nudge brain development and communication off course in ways that contribute to schizophrenia.
Citation: Liu, Y., Wang, J., Yang, H. et al. Functional variants at 1p36.23 confer risk of schizophrenia through modulating RERE. Nat Commun 17, 1742 (2026). https://doi.org/10.1038/s41467-026-68449-6
Keywords: schizophrenia genetics, RERE gene, neurodevelopment, glutamate signaling, synaptic function