Clear Sky Science · en
Antigravity confined interfacial self-assembly approach for the synthesis and characterization of nanofilms
Building Films That Grow Against Gravity
Many high‑tech products, from advanced filters to smart coatings, rely on ultra‑thin films only a few billionths of a meter thick. Making such films is surprisingly hard because gravity tends to pull heavy ingredients downward, limiting how we can stack and stabilize materials. This paper introduces an "antigravity" way to grow nanometer‑scale films that are strong, smooth, and large in area, opening paths to greener oil recovery, better insulation, and new soft materials.
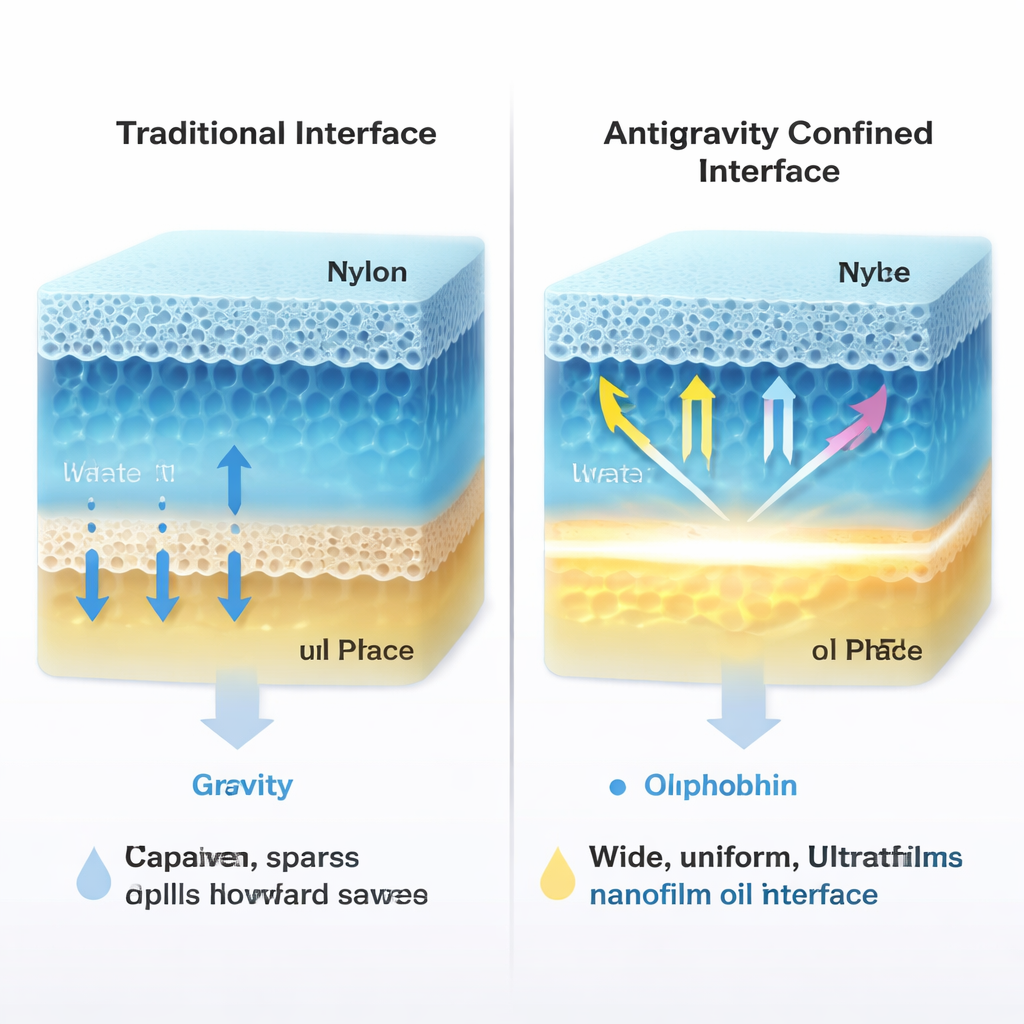
Why Gravity Is a Problem for Tiny Layers
Whenever two liquids meet, the molecules at their boundary can sometimes organize themselves into a thin film. But under normal conditions, gravity causes heavier molecules to sink and lighter ones to float, creating a vertical layering that fights many useful designs. If engineers want a dense component to sit on top, for example, they have to force the system out of its natural balance, which can make films fragile and short‑lived. Traditional methods that rely on simple liquid layers or droplets often produce films that are patchy, supported by rigid substrates, or too weak to be peeled off and used on their own.
Trapping Liquids to Beat Gravity
The researchers solve this by confining two immiscible liquids—water and oil—inside a pair of porous membranes that act like thin sponges. A hydrophilic nylon membrane holds a water‑based solution of cyclodextrins, doughnut‑shaped sugar molecules widely used in food and medicine. A hydrophobic PTFE membrane holds an oil, such as dodecane. When the two soaked membranes are pressed together, the liquids meet in a narrow, hidden gap. Inside the tiny pores, capillary forces—the same forces that pull water up a paper towel—overwhelm gravity and lock the liquids in place. This creates a flat, stable "antigravity" interface where molecules can arrange themselves with unusual precision.
How Sugar Rings and Oil Chains Build a Film
At this confined interface, cyclodextrin molecules diffuse from the water‑rich side toward the oil. Their hollow, water‑repelling interiors capture straight oil molecules, forming host–guest pairs that behave like tiny surfactants: one side likes water, the other likes oil. As more pairs gather, they lower the tension between the liquids and pack tightly at the boundary. Neighboring complexes then link together through hydrogen bonds, knitting themselves into a continuous nanofilm only tens of nanometers thick. By tuning the size of the membrane pores, the concentration of cyclodextrin, and the waiting time, the team can optimize how quickly these films form and how strong they become. Measurements of gas pressure needed to break through the film show that certain combinations—especially beta‑cyclodextrin with dodecane—produce films with particularly high mechanical stability.
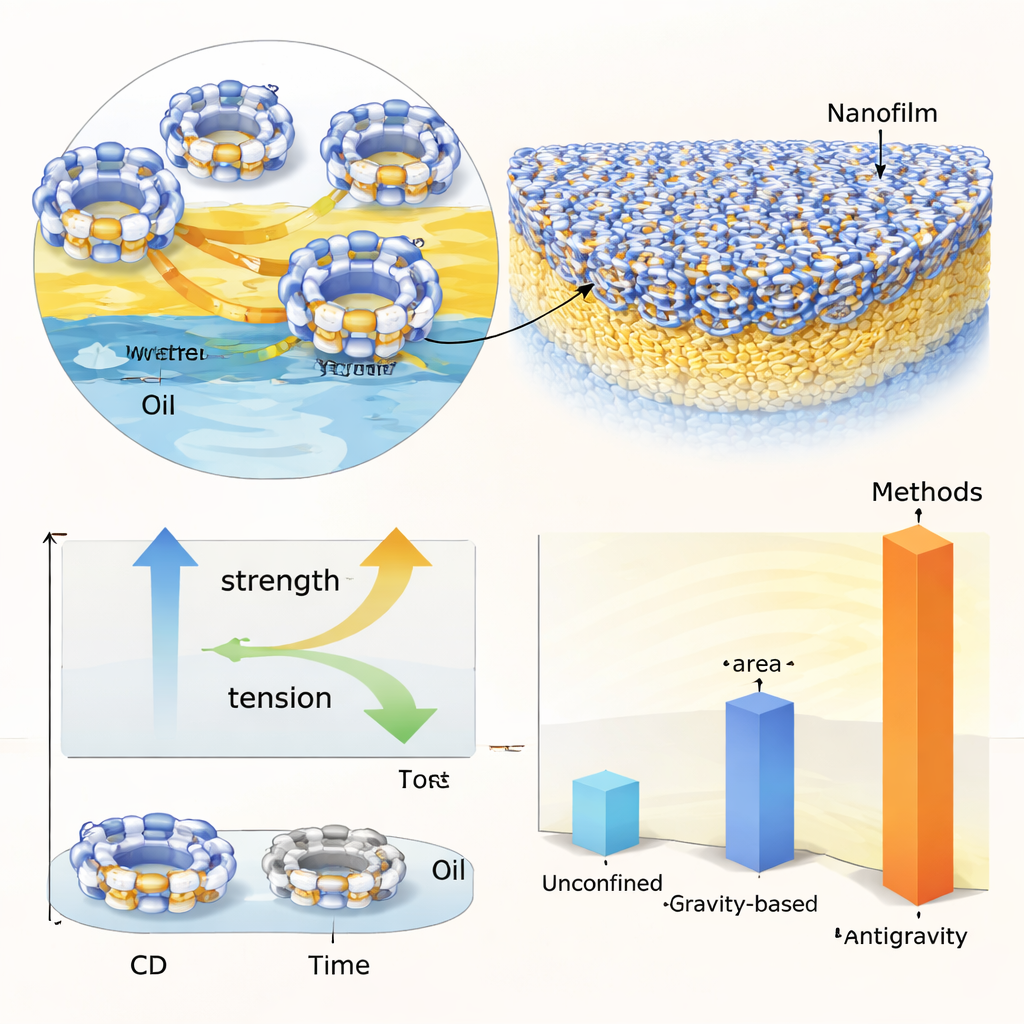
Making Larger, Stronger, and Smarter Films
Because the interface is spread across the entire contact area of the membranes, this method can create films that are far larger than those formed by ordinary liquid layering. With the same small amount of liquid, the antigravity setup yields films about 17 times larger than those grown under gravity and over 100 times larger than those made without confinement. The films can even repair themselves: if pressure briefly ruptures them, the building blocks at the interface reassemble once the stress is removed. The team also shows that changing membrane shapes—circles, stars, leaves—directly imprints the film outline, and that the same principle works with other liquid pairs, including food‑like systems and crude oils.
From Oil Fields to Everyday Materials
To show real‑world promise, the authors test these films in model oil‑recovery setups. When cyclodextrin films form in the tiny channels of rock, they raise the pressure needed for water to break through easy pathways, steering flow into smaller pores that still hold oil and boosting recovery. The same films slow heat loss in simple insulation tests and help create stable emulsions, which are important in foods, cosmetics, and pesticides. Overall, the study demonstrates a general strategy: by using confined, antigravity interfaces, it is possible to grow ultra‑thin, self‑supporting films with tunable strength, shape, and area, making nanofilm design more predictable and practical for a wide range of technologies.
Citation: Zhou, Z., Lei, J., Zhang, Z. et al. Antigravity confined interfacial self-assembly approach for the synthesis and characterization of nanofilms. Nat Commun 17, 1741 (2026). https://doi.org/10.1038/s41467-026-68447-8
Keywords: nanofilms, self-assembly, cyclodextrin, oil recovery, emulsions