Clear Sky Science · en
Improving erectile function in diabetic male mice by rescuing depalmitoylated FBP1 to reduce cavernosal lactate
Why this matters for men with diabetes
Erectile problems are common in men with diabetes, often appearing earlier and responding poorly to standard pills like Viagra. This study in mice uncovers a surprising culprit: a buildup of lactic acid (lactate) inside the erection tissue of the penis. The researchers not only trace how this buildup damages the tissue but also test an experimental RNA-based therapy delivered in tiny fat bubbles that restores erectile function. Their work hints at a future in which treating diabetic erectile dysfunction means fixing the underlying metabolism, not just boosting blood flow for a few hours.
From energy waste to tissue damage
Lactate is often thought of as a simple “waste” from hard-working muscles, but it is also a powerful signaling molecule. In men with diabetes-related erectile dysfunction, the authors found higher lactate levels in the blood than in age-matched controls. In a diabetic mouse model, lactate inside the spongy erectile tissue of the penis (the corpus cavernosum) rose steadily over time while the pressure generated during an erection fell. When healthy mice were given extra lactate, their penile tissue accumulated lactate, their erectile pressure dropped, and microscopic exams showed loss of smooth muscle and more collagen scar tissue. In cultured smooth muscle cells from the penis, high lactate slowed cell movement and pushed them from a contractile, “work-ready” state into a synthetic, scar-forming state, in part by overactivating self-digestion pathways known as autophagy.
A key enzyme goes missing
To understand why lactate builds up, the team compared genes involved in two opposing energy pathways: glycolysis, which produces lactate, and gluconeogenesis, which recycles lactate back into glucose. They saw no signs that glycolysis was overactive. Instead, a central gluconeogenic enzyme called FBP1 was consistently reduced in penile tissue from diabetic mice, diabetic rats, and in single-cell data from human men with diabetes-related erectile dysfunction. When the researchers genetically lowered Fbp1 in mice, lactate in the penis rose and erectile performance declined, even under relatively low lactate conditions. In smooth muscle cells, adding back working FBP1 helped protect against the harmful effects of high lactate, whereas an inactive FBP1 variant did not. This showed that FBP1’s enzyme activity is crucial for keeping lactate in check and maintaining healthy erection tissue.
Epigenetic locks and fatty “tags” on FBP1
Why is FBP1 reduced in diabetes? The study reveals a two-layered control system. First, the Fbp1 gene is clamped down at the DNA level. Chemical marks on histone proteins—specifically two “off” switches called H3K9me3 and H3K27me3—accumulate at the Fbp1 promoter in diabetic penile tissue. These marks are written by enzymes such as SUV39H1 and EZH2 and read by a protein called CBX3, forming a repressive complex that keeps Fbp1 switched off. Drugs and designer degraders that target these enzymes could partially lift this repression in cells but produced only modest improvements in diabetic mice. Second, even when FBP1 protein is present, it can be disabled by a fatty modification called palmitoylation at a single amino acid, Cys282. The team identified the enzyme ZDHHC13 as the palmitoyltransferase that attaches this fatty group. Palmitoylation subtly changes FBP1’s shape near a critical residue, blunting its ability to process lactate. Blocking Zdhhc13 in cells and mice improved metabolism and erectile function only slightly, underscoring that neither epigenetic drugs nor palmitoylation blockers alone are enough.
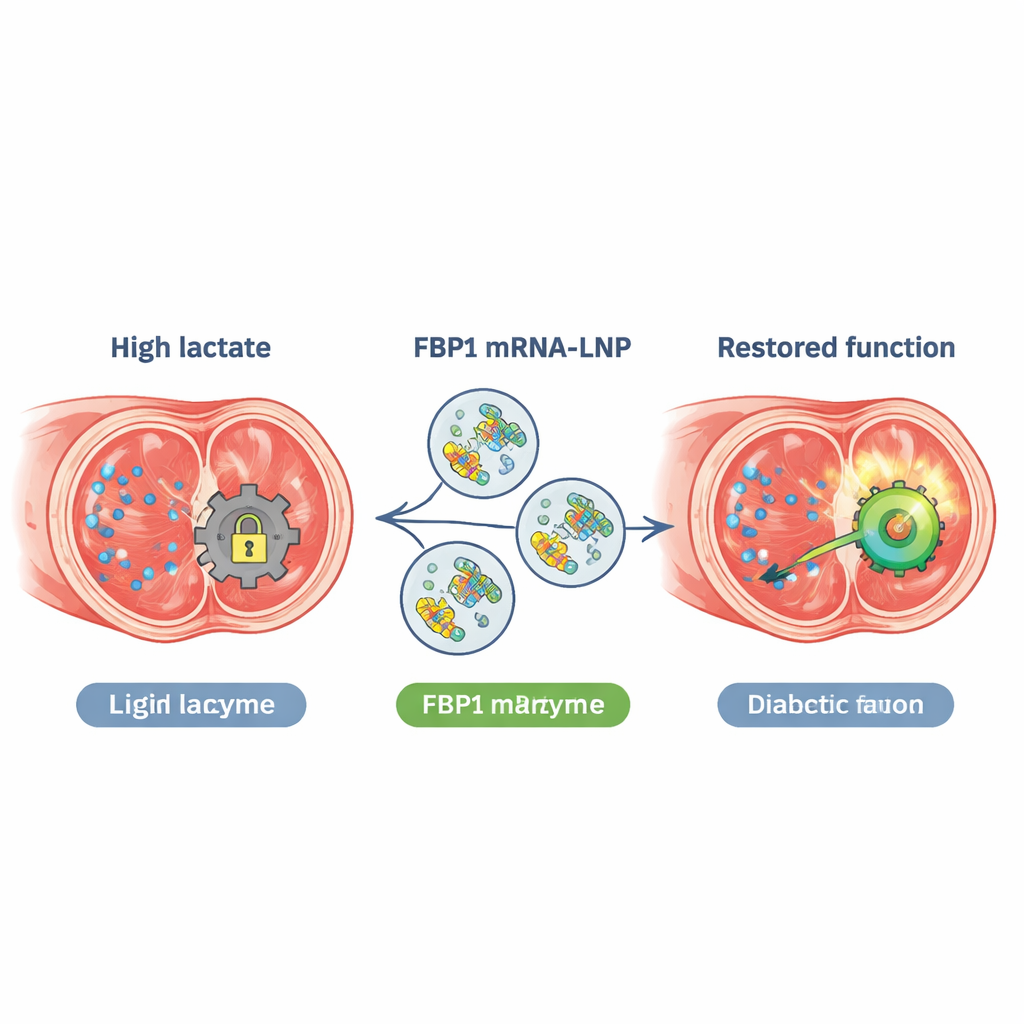
RNA-filled nanoparticles as a repair kit
To tackle both problems at once, the researchers engineered an mRNA that encodes a palmitoylation-resistant Fbp1 mutant (C282S) and packaged it inside lipid nanoparticles—similar in spirit to mRNA COVID-19 vaccines. Injected directly into the penile tissue of diabetic mice, these particles were efficiently taken up by smooth muscle cells. The treated mice showed higher FBP1 protein levels, sharply lower cavernosal lactate, better erectile pressure, more smooth muscle, and less fibrosis. A mid-range dose worked best; higher dosing did not add benefit. The therapy also normalized the overactive autophagy seen in diabetes models and improved erectile outcomes across multiple types of diabetic mice, with no clear liver or kidney toxicity during the study period.
What this could mean for future treatments
In plain terms, this work reframes troublesome erections in diabetes as a problem of “sour” metabolism inside the penis: too much lactate and too little functioning FBP1. By restoring a non-palmitoylated, active version of FBP1 exactly where it is needed, the mRNA–nanoparticle therapy repairs the tissue’s energy balance, preserves smooth muscle, and allows normal erections to return—at least in mice. While many questions remain about long-term safety, dosing, and translation to humans, the study points toward a new class of treatments that rebuild the metabolic health of erectile tissue and may one day complement or rescue patients who do not respond to current drugs.
Citation: Xiao, M., Guo, W., Zeng, R. et al. Improving erectile function in diabetic male mice by rescuing depalmitoylated FBP1 to reduce cavernosal lactate. Nat Commun 17, 1740 (2026). https://doi.org/10.1038/s41467-026-68443-y
Keywords: diabetic erectile dysfunction, lactate metabolism, FBP1 enzyme, lipid nanoparticle mRNA therapy, penile smooth muscle