Clear Sky Science · en
Women with polycystic ovary syndrome exhibit impaired endometrial receptivity with excessive ERα and histone lactylation
Why the uterine lining matters in a common hormone disorder
Polycystic ovary syndrome (PCOS) is one of the most common hormone-related conditions in women and a leading cause of infertility. Many people assume that PCOS causes infertility mainly because it disrupts ovulation. This study looks beyond the ovaries to the uterus itself, asking a crucial question: even when a good-quality embryo is available, is the uterine lining in women with PCOS ready to allow it to implant and grow?
Looking past the egg to the “soil”
To separate problems with eggs from problems with the womb, the researchers analyzed more than 4,200 frozen embryo transfer cycles, in which single high-quality blastocysts were transferred. By carefully matching women with PCOS to similar women without PCOS (for age, body mass index, infertility duration, and other factors), they could focus on differences in the uterine environment. Even after this strict matching, women with PCOS had lower implantation, pregnancy, and live birth rates, suggesting that the uterine lining itself is less welcoming to embryos.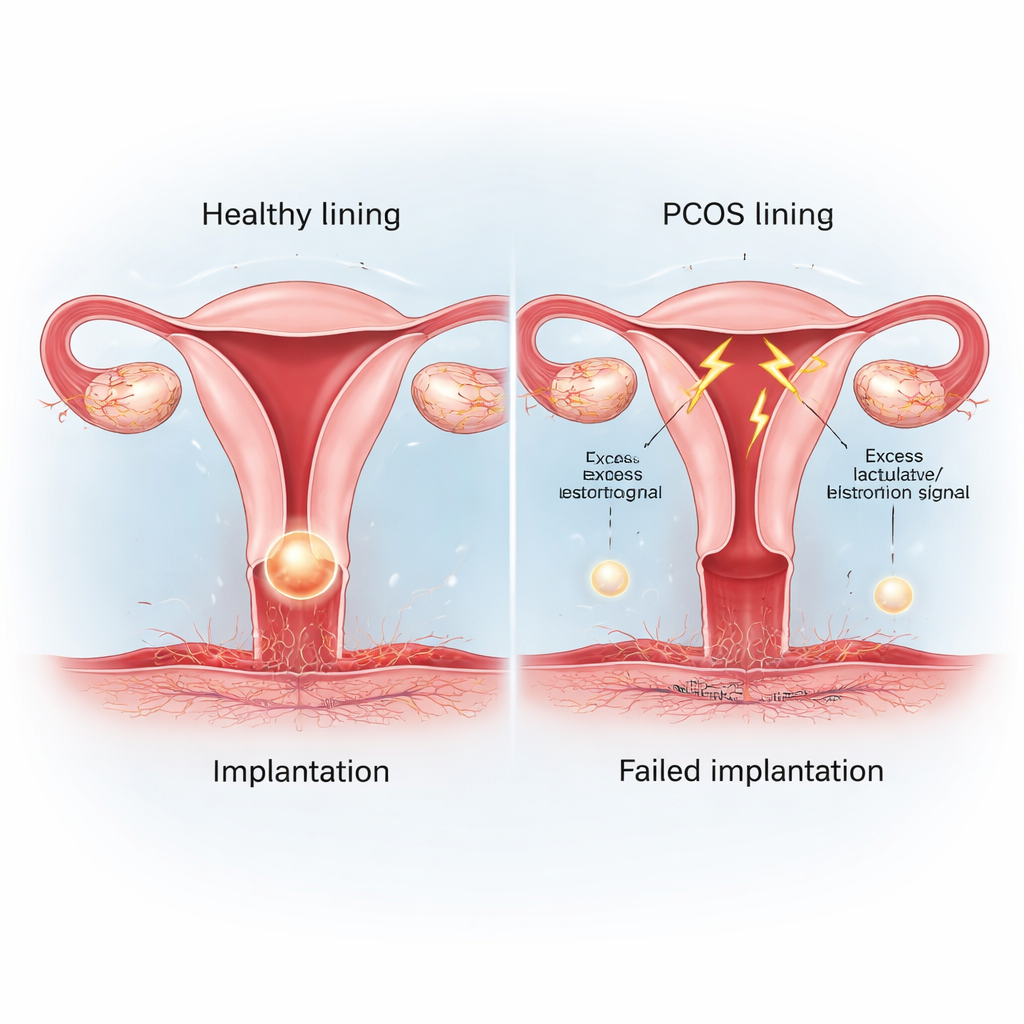
When the lining does not fully prepare
The team then examined samples of endometrium, the tissue that lines the uterus and must change each month to receive an embryo. In women with PCOS, the surface cells were overly busy dividing, while the deeper support cells failed to properly “decidualize” – a transformation that normally creates a nurturing bed for implantation. Key genes and proteins that mark a receptive lining, such as COX2 and HOXA10, were reduced. Together, these changes point to a lining stuck between growth and readiness, leaving the so‑called “window of implantation” narrower or mistimed.
Too much estrogen signal and a new chemical mark
Hormones guide these monthly changes, and balance is critical. The researchers found that in PCOS the estrogen receptor called ERα was unusually abundant in the mid‑cycle endometrium, while the progesterone receptor was not significantly altered. Genes that respond to estrogen were also overactive, hinting that the lining is being pushed too hard by estrogen-like signals. At the same time, the team discovered high levels of “histone lactylation,” a recently described chemical tag on DNA-packaging proteins that is fueled by lactate, a by-product of sugar breakdown. In both women with PCOS and mice with PCOS‑like symptoms, these lactylation marks – especially one called H3K18la – were elevated in the uterine lining, and were enriched near genes involved in hormone responses and cell growth.
A harmful feedback loop between signals and metabolism
Digging deeper, the scientists used human uterine cells and mouse models to test how these factors interact. When they artificially increased ERα in cells, lactate levels rose, and histone lactylation increased as well. This in turn boosted ERα activity, creating a positive feedback loop: more estrogen signaling leads to more lactylation, which further amplifies estrogen effects. In PCOS mice, the uterus showed the same pattern – impaired receptivity, high ERα, and high histone lactylation. Importantly, when the researchers blocked ERα or reduced lactate production locally in the uterus, uterine structure improved, implantation sites appeared, and implantation-related genes moved back toward normal levels.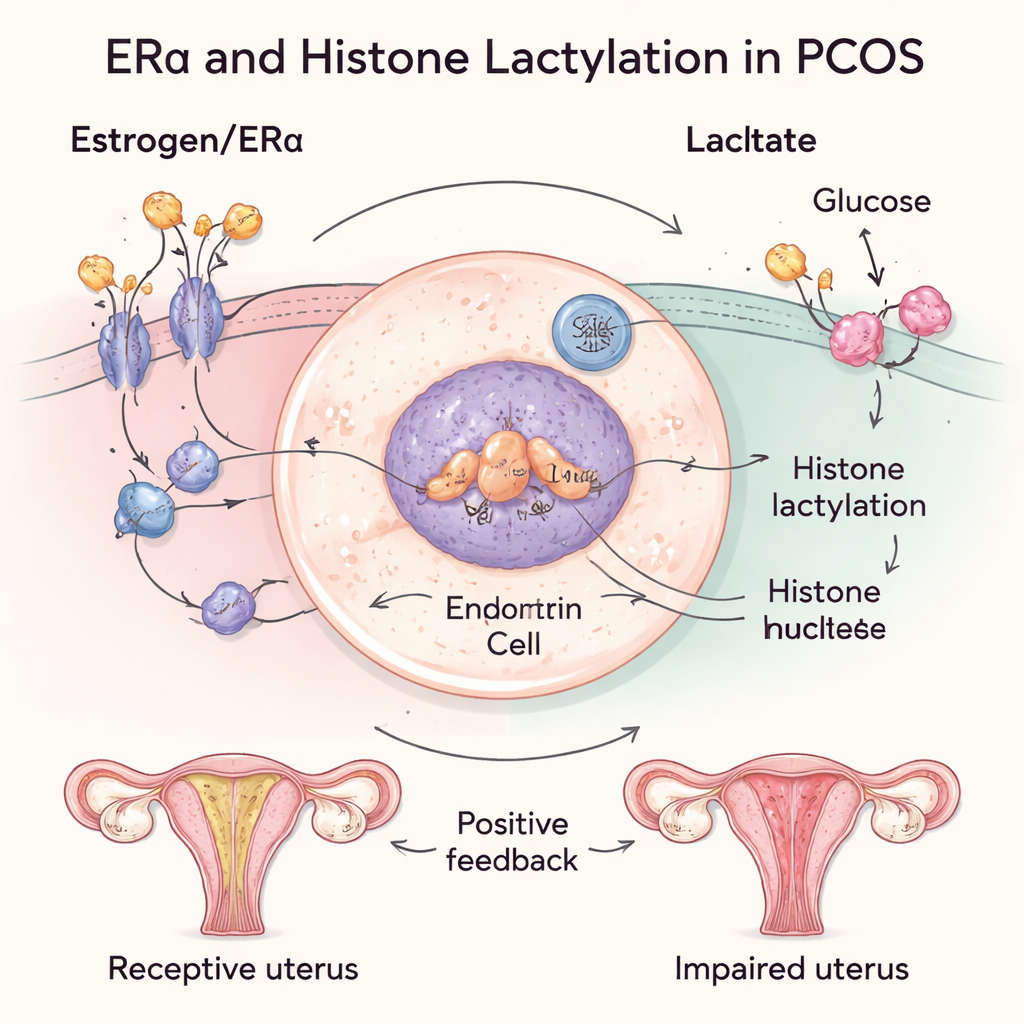
What this means for women with PCOS
For a layperson, the take‑home message is that in PCOS, infertility is not only about producing a good egg. The “soil” – the uterine lining – can also be out of balance. This study shows that an overactive estrogen receptor and an overabundance of lactate-driven histone marks act together to make the lining less receptive to embryos. By dialing down these signals or the lactate that feeds them, at least in mice, implantation and early pregnancy can be rescued. In the future, treatments that fine‑tune this hormone–metabolism loop in the uterus could complement existing ovulation-based therapies, offering new hope for improving fertility in women with PCOS.
Citation: Shan, H., Wang, Y., Liao, B. et al. Women with polycystic ovary syndrome exhibit impaired endometrial receptivity with excessive ERα and histone lactylation. Nat Commun 17, 1739 (2026). https://doi.org/10.1038/s41467-026-68441-0
Keywords: polycystic ovary syndrome, endometrial receptivity, estrogen receptor alpha, histone lactylation, infertility