Clear Sky Science · en
Serine inhibits granulosa cell ferroptosis to maintain ovarian function
Why this research matters for women’s health
Modern cancer treatments save many lives but can come with a painful trade-off: loss of fertility and early menopause. At the same time, more young women report sleep-deprived, high-stress lifestyles and unexplained drops in ovarian function. This study uncovers a shared biological link between chemotherapy, staying up late, and premature ovarian insufficiency—and points to a surprisingly simple nutrient, the amino acid serine, as a potential way to protect the ovaries without weakening cancer therapy.
A quiet crisis in ovarian health
Premature ovarian insufficiency (POI) happens when a woman’s ovaries lose function before age 40, causing irregular or absent periods, infertility, and menopausal symptoms. Chemotherapy with the widely used drug cyclophosphamide can trigger POI in many breast cancer patients, but a large number of young women develop similar ovarian problems without any medical treatment—so-called idiopathic POI. The authors set out to understand whether a common, overlooked factor might connect these two groups and whether it could be safely targeted to protect fertility.
A drop in serine tracks with weakened ovaries
The team followed 27 young breast cancer patients through their first round of cyclophosphamide chemotherapy. After just one cycle, most women showed reduced levels of key ovarian hormones, including estrogen and anti-Müllerian hormone (AMH), clear signs that the ovaries were already under stress. At the same time, detailed blood analysis revealed a sharp fall in several amino acids, with serine standing out as especially reduced. In 25 of the 27 women, hormone declines and serine drops went hand in hand, suggesting that lower serine might be tied to emerging ovarian damage.
Low serine and late nights in otherwise healthy women
To see if this connection extended beyond chemotherapy, the researchers studied 124 young women being evaluated for infertility. Those with poor ovarian function, defined by low AMH and fewer egg-containing follicles on ultrasound, consistently had lower blood serine than women with normal ovaries. When the team dug into lifestyle data, one factor stood out: staying up late. Women who frequently went to bed late—often because of sleep disorders or prolonged screen use—had lower serine and AMH levels, and the more often they stayed up, the worse both measures became. In mice kept awake for prolonged periods, the ovaries shrank, hormone balance was disturbed, and iron built up inside ovarian cells, mirroring the patterns seen with chemotherapy.
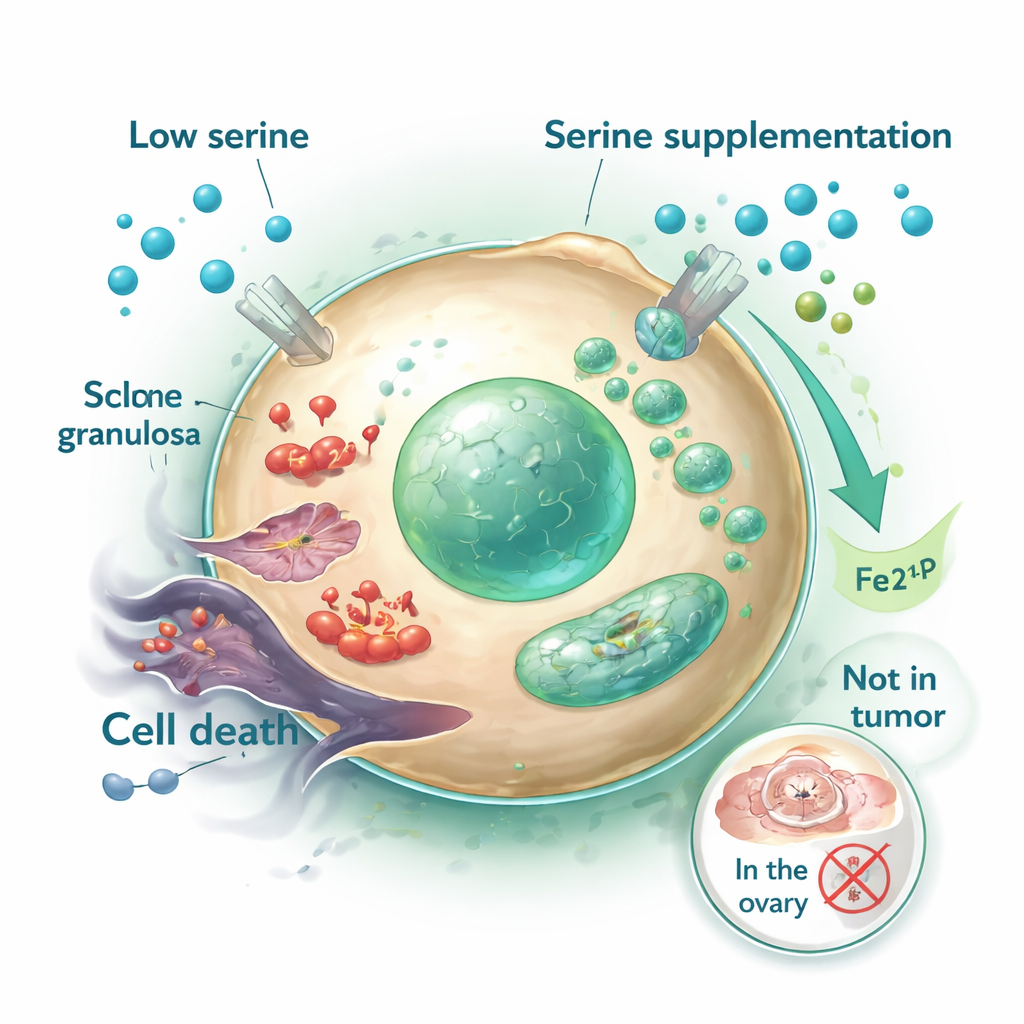
How serine shields ovarian cells from iron-driven damage
Using mouse models, the scientists tested whether adding serine to the diet could blunt the harmful effects of cyclophosphamide. Whether delivered in drinking water or by oral dosing, serine helped preserve normal ovarian size and structure, maintained estrogen, AMH, and follicle-stimulating hormone at healthier levels, and supported the survival and growth of granulosa cells—the cells that nourish developing eggs. Inside these cells, cyclophosphamide was found to trigger a specific form of cell death called ferroptosis, driven by an overload of iron and destructive lipid oxidation. Serine countered this by boosting production of a lipid messenger called sphingosine-1-phosphate (S1P). S1P, in turn, dampened a stress pathway in the nucleus and reduced the activity of HO-1, a protein that otherwise helps release iron. With less free iron and fewer damaged mitochondria, granulosa cells were far less likely to die.
Protecting ovaries without helping tumors
A natural worry is that feeding extra serine might also nourish cancer cells and blunt chemotherapy. To address this, the researchers used a breast cancer mouse model and several tumor cell lines. Encouragingly, serine did not reduce cyclophosphamide’s ability to shrink tumors or kill cancer cells, even while it clearly protected ovarian tissue. The drug’s damaging effects on the ovary and its cancer-killing action in tumors appear to rely on different mechanisms; iron overload and ferroptosis were prominent in ovarian cells but not in tumor tissue under the same treatment conditions.
What this could mean for patients
This work suggests that a simple, inexpensive nutrient—serine—may be a key guardian of ovarian health. In both chemotherapy-treated patients and women with unexplained ovarian problems linked to chronic late nights, lower blood serine tracks closely with reduced ovarian function. In animals, restoring serine levels prevents iron-driven cell death in the ovary without protecting tumors from chemotherapy. While clinical trials are now needed to test safety, dosage, and long-term outcomes in people, the study raises the possibility that managing lifestyle factors like sleep and thoughtfully supplementing serine could one day become part of routine strategies to help young women preserve their fertility and delay premature ovarian decline.
Citation: Gu, HC., Zhuo, YQ., Wang, LF. et al. Serine inhibits granulosa cell ferroptosis to maintain ovarian function. Nat Commun 17, 1738 (2026). https://doi.org/10.1038/s41467-026-68440-1
Keywords: premature ovarian insufficiency, serine, chemotherapy and fertility, sleep deprivation, granulosa cell ferroptosis