Clear Sky Science · en
Suppressing solvent adducts via coordination competition enables scalable perovskite photovoltaics
Why better solar films matter
Perovskite solar cells are one of the most promising contenders to deliver cheaper, more efficient solar power than today’s silicon panels. But while researchers can make tiny, record-breaking devices in the lab, turning the same materials into large, factory-scale solar modules has proven much harder. This study tackles a hidden bottleneck in scaling up perovskite films and offers a chemistry-based fix that pushes big, blade‑coated modules to efficiencies suitable for real‑world deployment.
From spinning drops to factory blades
Most top‑performing perovskite cells are made by spin coating—spreading a liquid solution across a small wafer by spinning it at high speed. This method forces the solvent to fly off quickly, then an extra “antisolvent” is added to trigger sharp, well‑controlled crystal growth. Industrial methods, however, must coat large glass sheets with simple tools like moving blades, relying on slow solvent evaporation instead of rapid spinning. The authors show that this difference in fluid behavior leads to very different crystal growth, and that recipes fine‑tuned for spin coating do not automatically transfer to scalable blade coating.
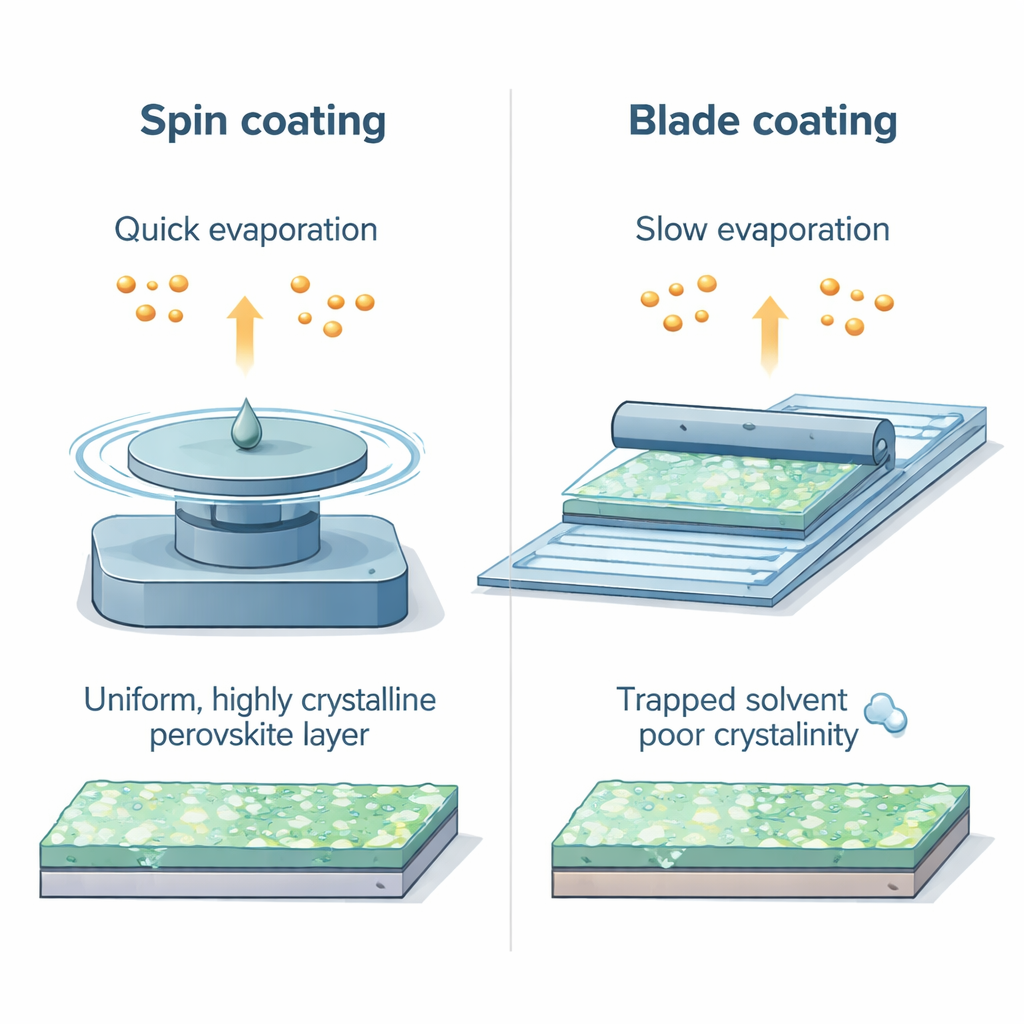
A hidden clock in the wet paint
The team identifies a key but previously overlooked variable: the time that solvent molecules stay tightly bound to the perovskite ingredients in the wet film, which they call the solvent–precursor interaction time. In blade‑coated films, the slower drying leaves the solvent strongly attached for longer, forming stubborn “solvent adduct” phases and trapping residues inside the film. X‑ray measurements and chemical analysis reveal that these solvent‑rich intermediates are much more pronounced in blade‑coated layers than in spin‑coated ones, and the result is poorer crystal order and more electronic defects—both bad news for solar cell performance.
Letting the right molecules win
Rather than forcing the solvent out with harsher processing, the researchers design a subtle molecular competition. They introduce a small additive molecule bearing two hydroxyl (OH) groups, nicknamed 2OH, into the perovskite “ink.” This molecule is tailored to bind more strongly to lead ions than the common processing solvent N‑methyl‑2‑pyrrolidone (NMP). A battery of techniques—including infrared spectroscopy, X‑ray absorption, and diffraction—shows that 2OH successfully outcompetes the solvent at lead sites, weakens the solvent–lead grip, and shifts the balance toward free solvent that can evaporate more easily. At the same time, 2OH helps organize the organic components of the perovskite, steering them toward the desired crystal phase.
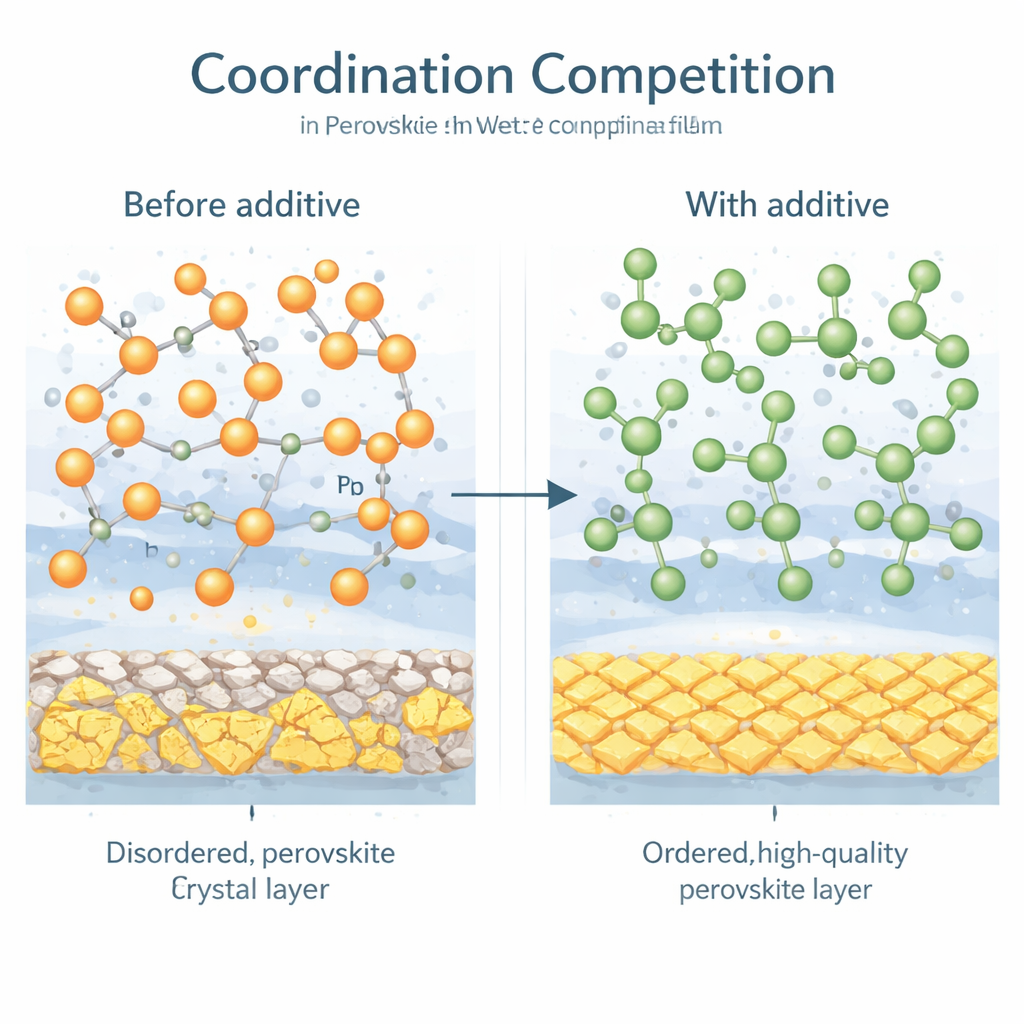
Cleaner films, bigger devices
To test how this coordination competition translates into real devices, the authors vary additives with zero, one, or two OH groups. As the number of OH groups rises, residual solvent in the film drops, the lead–solvent bonds become weaker, and the perovskite crystals become more ordered and defect‑poor. Solar cells made with the 2OH additive reach a power conversion efficiency of 26.5% on small test cells, with noticeable gains in voltage and fill factor. Crucially, the same strategy scales: blade‑coated mini‑modules of 20.8 square centimeters hit 22.9% efficiency, and a pre‑pilot sub‑module of 728.0 square centimeters is certified at 22.58%, putting blade‑coated perovskites into a performance class previously reserved for more delicate lab methods.
More power, longer life
Better crystals not only boost efficiency but also stability. Devices made with 2OH keep 92% of their initial performance after nearly 1,000 hours of continuous illumination, compared with 80% for control devices. Electrical imaging further reveals that large‑area modules with the additive show more uniform light emission and fewer “hot spots,” signatures of reduced shunting and fewer defects. Measurements of charge flow and recombination confirm that the films lose less energy through unwanted pathways, helping explain the improved voltage and durability.
A practical route to scalable perovskites
For non‑specialists, the takeaway is that the authors have found a simple chemical “knob” that lets manufacturers tune how long the solvent clings to the forming perovskite crystals in large‑area coating. By introducing a molecule that nudges the chemistry away from sticky solvent complexes and toward clean, well‑ordered crystals, they achieve high efficiency and stability using industrially friendly blade coating. This approach offers a realistic path to mass‑produced perovskite solar panels that are both powerful and manufacturable at scale.
Citation: Jin, L., Zhang, S., Zhou, J. et al. Suppressing solvent adducts via coordination competition enables scalable perovskite photovoltaics. Nat Commun 17, 1737 (2026). https://doi.org/10.1038/s41467-026-68439-8
Keywords: perovskite solar cells, blade coating, thin-film photovoltaics, crystal growth control, solar module scaling