Clear Sky Science · en
Construction of remote dual stereocenters by electrochemical cobalt-catalyzed enantioselective desymmetrization
Shaping molecules for better medicines
Chemists have long known that the 3D shape of a molecule can make or break a drug’s effect in the body. Many successful medicines and catalysts work only because certain atoms point in just the right directions in space. Yet precisely arranging two distant “control points” in a single step has been extremely difficult. This study introduces an electricity-driven method that uses a single cobalt-based catalyst to sculpt molecules with two far-apart stereocenters—key 3D features—opening new possibilities for designing drugs and specialized chemical tools.
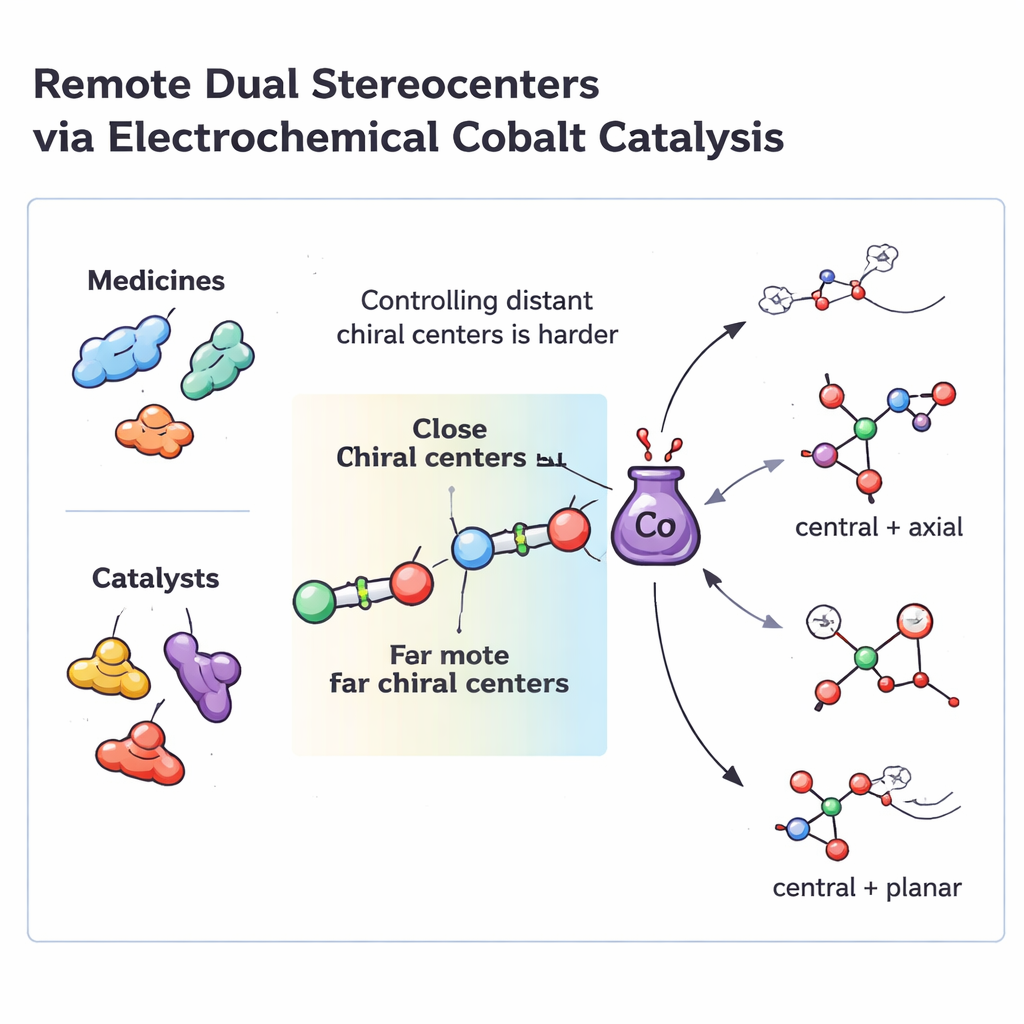
Why distant control points matter
Many modern drugs and high-performance catalysts contain two non-adjacent stereocenters—specific atom arrangements that can exist as left- or right-handed versions. These remote control points are often crucial for how a molecule fits into a biological target or a metal center in a catalyst. Traditional asymmetric methods are very good at building neighboring stereocenters, where the two points are right next to each other. But when those points are separated by five or more atoms, the usual “steering” models break down, and chemists often need multi-step routes or two different catalysts working together. Such multi-catalyst systems are hard to tune, prone to incompatibilities, and usually tailored to narrow families of starting materials.
A one-catalyst, electricity-assisted shortcut
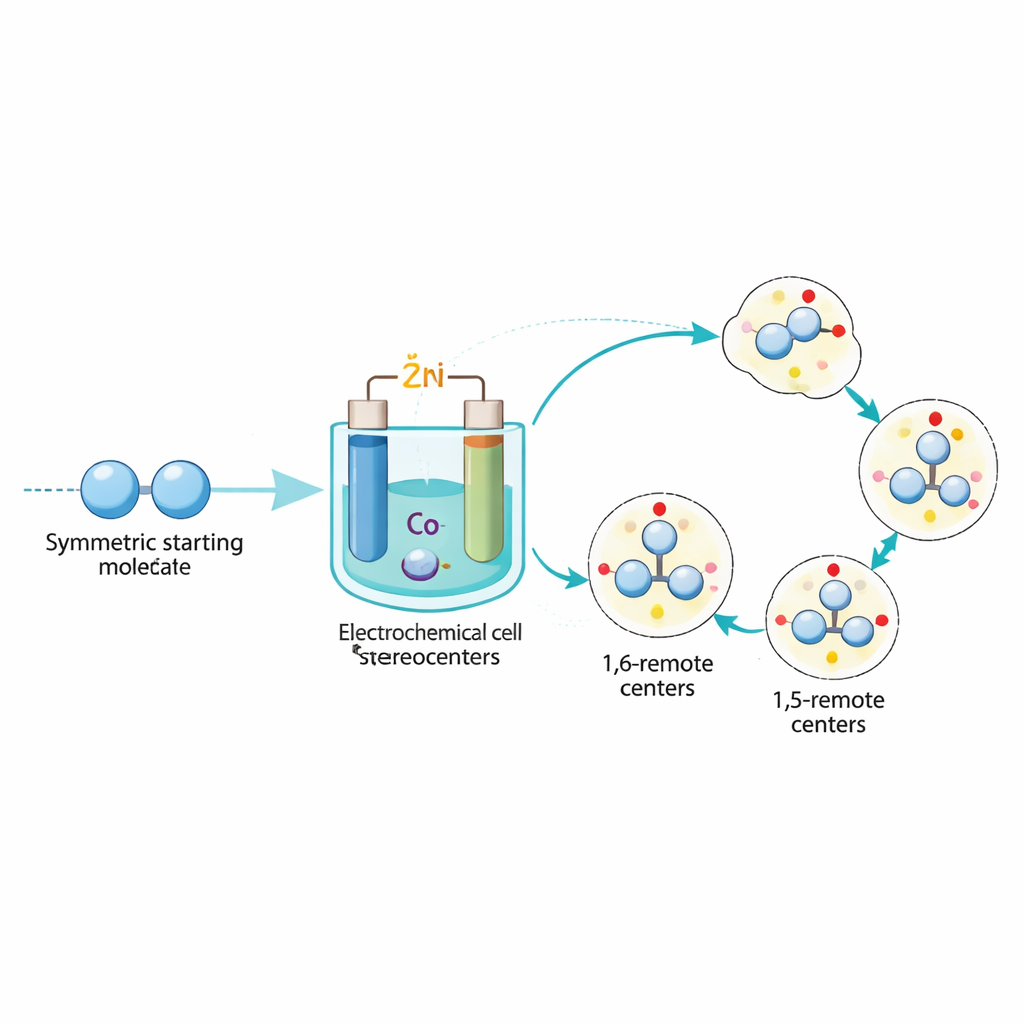
The authors tackled this challenge by combining electrochemistry with a chiral cobalt catalyst. Instead of using chemical reducing agents, they apply a small electric current in a simple cell equipped with zinc and nickel electrodes. This current turns a cobalt complex bearing a chiral ligand into a reactive, low-valent species that can bind and reshape simple starting materials: symmetric dialdehydes and so-called enynes, which contain both a double and a triple bond. The key idea is desymmetrization: starting from molecules that have two equivalent “ends” and using the chiral cobalt to break this balance in a controlled way, so each end becomes part of a well-defined 3D structure.
Turning symmetry into diversity
Under the optimized conditions, this electrochemical process reliably converts a wide variety of dialdehydes and enynes into products that contain two distinct chiral elements at remote positions. Depending on the starting framework, the team can generate four different types of 3D arrangements in the same unified setup: a central stereocenter paired with a twisted C–C axis, a central stereocenter with a twisted C–O axis, and two kinds of planar chirality based on [2.2]paracyclophane and ferrocene scaffolds. In practice, that means they can make families of molecules whose shapes are locked in space for years, with very high selectivity for one 3D form over all others, and with many different substituents tolerated on the aromatic rings.
Peeking under the hood of the reaction
To understand how this process works, the researchers used labeling experiments and mechanistic probes. By replacing certain hydrogen atoms with deuterium (a heavier isotope of hydrogen), they showed that these atoms end up exactly where expected in the final product, and that no scrambling occurs between different molecules. This rules out some competing reaction pathways and supports a stepwise mechanism in which the cobalt catalyst first forms a ring-like intermediate with the enyne, establishing the first stereocenter. The dialdehyde then inserts into this intermediate to create the second stereocenter, followed by steps that release the product and regenerate the active cobalt species. The team also demonstrated that the products can be modified further—oxidized, coupled, or turned into ligands—without losing their finely tuned 3D control.
From lab method to useful molecular building blocks
In accessible terms, this work shows how electricity and a single cobalt catalyst can carve highly specific 3D shapes into otherwise simple and symmetric starting molecules. Instead of designing a different catalytic system for each type of chiral architecture, the same electrochemical platform can produce multiple classes of chiral products with two remote control points. Because such structures are common in top-selling drugs and advanced catalysts, this strategy offers chemists a powerful and flexible way to build complex, shape-sensitive molecules more directly and efficiently.
Citation: Li, Y., Liu, S., Yuan, B. et al. Construction of remote dual stereocenters by electrochemical cobalt-catalyzed enantioselective desymmetrization. Nat Commun 17, 743 (2026). https://doi.org/10.1038/s41467-026-68437-w
Keywords: asymmetric catalysis, electrochemistry, cobalt catalysis, chiral molecules, remote stereocenters