Clear Sky Science · en
A self-breathing electrode enabled by interface regulation and gradient wettability engineering for industrial H2O2 electrosynthesis
Why a Better Way to Make Peroxide Matters
Hydrogen peroxide is a familiar disinfectant in medicine cabinets, but it is also a workhorse chemical for cleaning water, treating pollution, and making many everyday products. Today, almost all industrial hydrogen peroxide is made in giant plants using a complex, energy-hungry process that creates hazardous by-products and locks production into a few centralized sites. This study explores a very different approach: compact electrochemical devices that can make hydrogen peroxide directly from air, water, and electricity, opening the door to cleaner, cheaper, and more local production.
The Trouble with Flooded Electrodes
At the heart of these devices is a gas diffusion electrode, a thin, porous sheet that must bring together air, liquid water, and an electrically conductive solid so the desired reaction can occur. In conventional designs, a plastic-like binder called PTFE is melted around carbon particles to keep water from drowning the pores. But this “fused” structure tends to create sealed patches and random channels. Under high power, water floods most of the carbon, oxygen can no longer reach the active sites, and the electrode quickly loses its ability to make hydrogen peroxide efficiently.
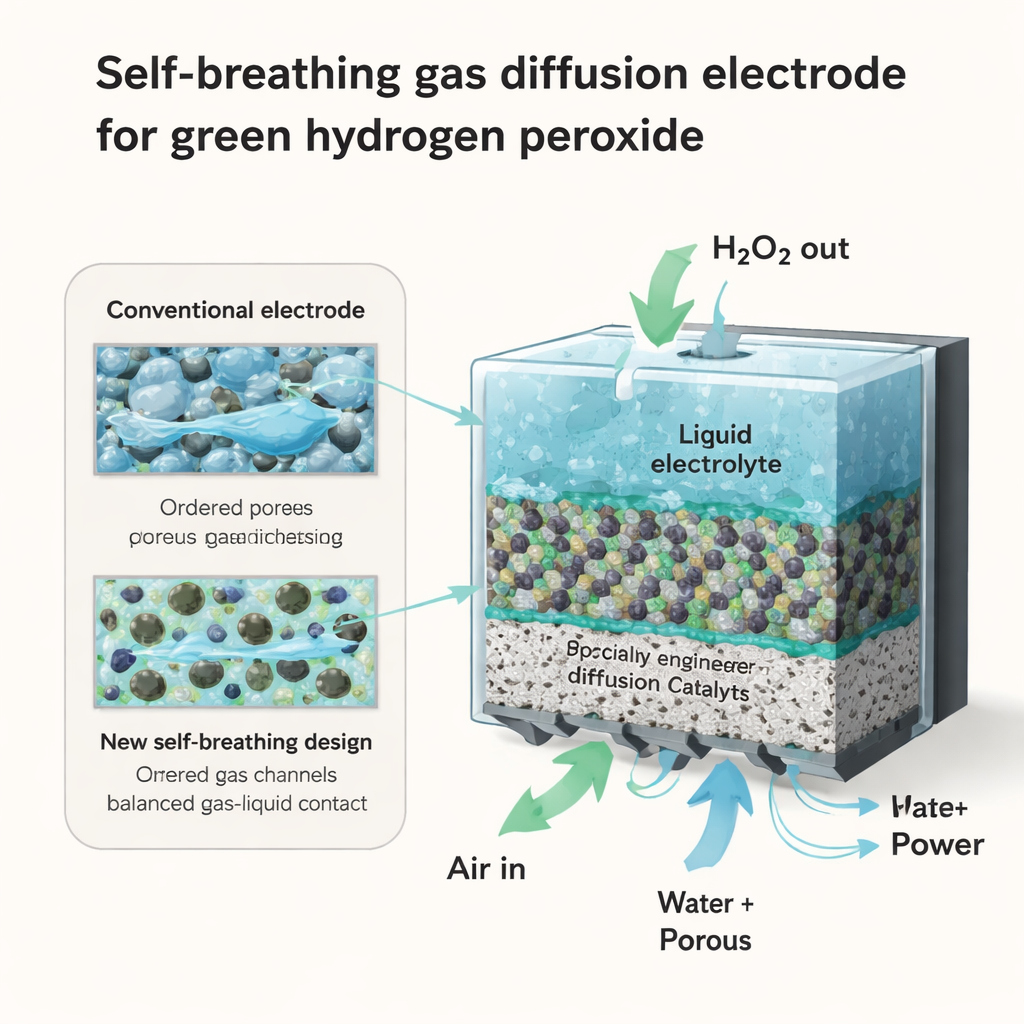
A New Way to Pack the Pieces
The authors propose a different architecture they call a particulate-packed electrode. Instead of melting PTFE into a continuous film, they keep it as tiny, separate particles mixed intimately with carbon. Using advanced 3D imaging and computer simulations, they show that this non-fused structure yields a maze of interconnected pores where hydrophobic PTFE and hydrophilic carbon sit side by side. This creates many stable “three-phase” points where air, liquid, and solid touch at once—exactly the micro-environments where oxygen can be turned cleanly into hydrogen peroxide. Because the pores remain open and well connected, oxygen can travel more freely and flooding is much less severe, even at demanding current levels.
Guiding Water and Peroxide with Gradients
Building on this insight, the team goes beyond simply mixing particles and deliberately shapes both the pore sizes and surface wetness through the thickness of the electrode. They construct layered catalyst coatings in which the side facing the air is very water-repellent and finely porous, while the side facing the liquid is more wettable and contains larger channels. Simulations and microfluidic experiments show that this gradient acts like a built-in pump: capillary forces push electrolyte and newly formed hydrogen peroxide toward the more open, hydrophilic region, while leaving dry pathways for oxygen elsewhere. This combination of a hydrophobic “shield” and a directional “drain” helps the electrode resist flooding and continuously move product away from reaction sites.
From Lab Concept to Working Hardware
Electrodes built with this gradient design maintain high selectivity for hydrogen peroxide—over 80 to 85 percent of the electric current goes to the desired product—at industrially relevant current densities of 300 to 400 milliamps per square centimeter, and they do so for hundreds of hours without external oxygen feeding. The authors then integrate many of these electrodes into a four-cell stack roughly the size of a small cabinet. With integrated pumps, heat management, and power electronics, the system produces concentrated hydrogen peroxide solutions continuously while drawing oxygen directly from air. A cost analysis suggests that the peroxide can be made for well under one dollar per kilogram, competitive with current large-scale methods but in a far smaller, more flexible footprint.
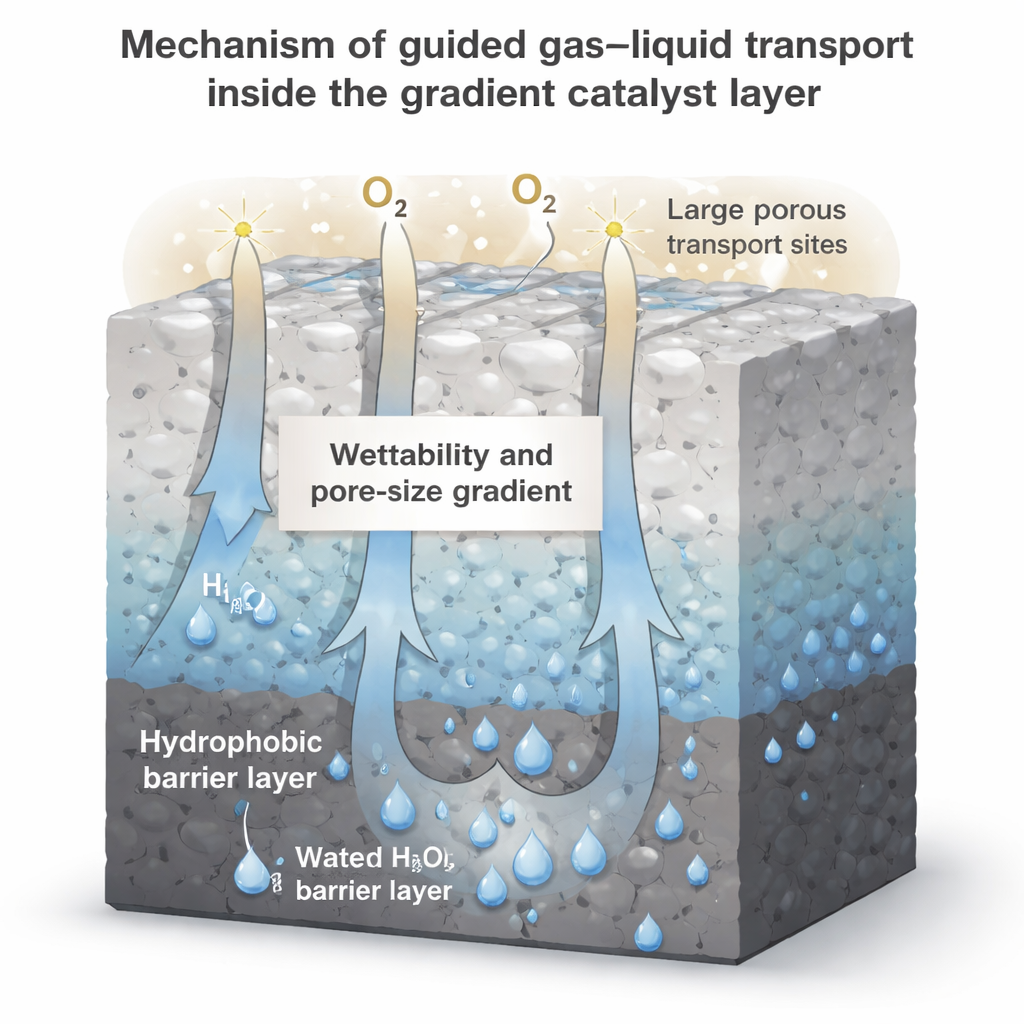
What This Means for Everyday Use
For non-specialists, the key message is that this work turns an abstract materials tweak into a practical machine: by carefully arranging tiny pores and tailoring how water does—or does not—wet those pores, the researchers create an electrode that “breathes” on its own and keeps working at high rates. Such self-breathing electrodes could power on-site hydrogen peroxide generators for factories, farms, or water treatment plants that simply plug into renewable electricity and ambient air. If deployed widely, this approach could cut the environmental footprint of a common but critical chemical while making clean oxidants available wherever they are needed.
Citation: Tian, Y., Pei, L., Wang, S. et al. A self-breathing electrode enabled by interface regulation and gradient wettability engineering for industrial H2O2 electrosynthesis. Nat Commun 17, 1735 (2026). https://doi.org/10.1038/s41467-026-68436-x
Keywords: hydrogen peroxide, gas diffusion electrode, electrochemical synthesis, wettability gradient, decentralized chemistry