Clear Sky Science · en
Electric-field-driven CO2 polarization and bioinspired proton blocking unlock CO2 reduction in strong acid without metal cations
Turning a Problem Gas into a Useful Fuel Ingredient
Carbon dioxide (CO2) is the main greenhouse gas driving climate change, but it is also a potential raw material for making fuels and chemicals using renewable electricity. One of the biggest obstacles is that CO2 is stubbornly unreactive, especially in very acidic liquids where unwanted hydrogen gas is formed instead. This study shows how a cleverly shaped gold nanomaterial, wrapped in a bioinspired coating, can overcome these hurdles and turn CO2 into carbon monoxide (CO) efficiently in harsh acidic conditions, without relying on dissolved metal salts that usually cause clogging and waste.

Why Working in Harsh Acid Matters
Most devices that electrically convert CO2 operate in neutral or alkaline liquids. There, however, CO2 tends to react with the liquid and form carbonate and bicarbonate salts, wasting much of the gas and shortening device lifetimes as solids build up. Running the reaction in strong acid could avoid these losses and make better use of every molecule of CO2. The problem is that in acid, positively charged hydrogen ions are everywhere and eagerly combine to make hydrogen gas, outcompeting CO2 for electrons at the electrode surface. At the same time, neutral CO2 molecules do not stick easily to metal surfaces. The authors set out to design a catalyst and surrounding environment that would both attract and activate CO2, and at the same time keep protons at bay, all in a metal‑cation‑free acidic solution.
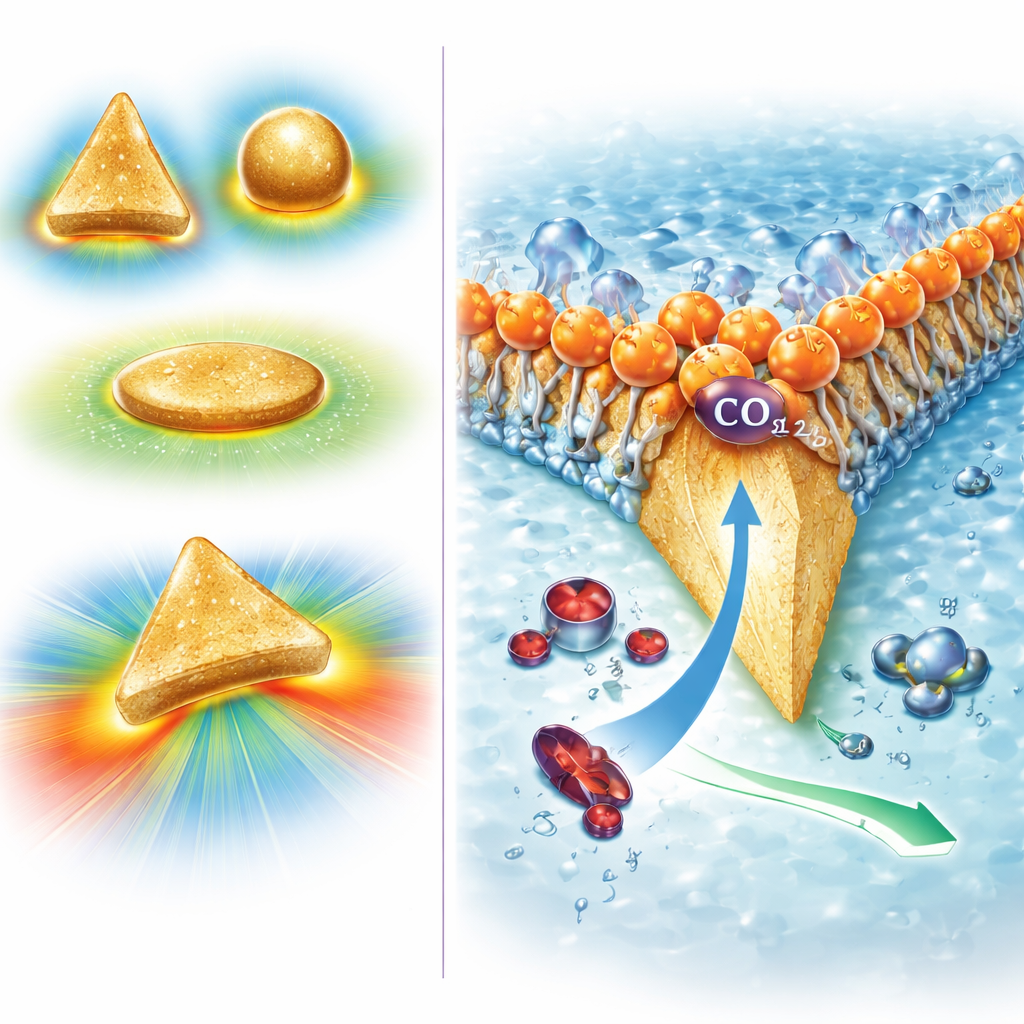
Sharp Gold Triangles that Supercharge CO2
The team created tiny, flat gold triangles only about 70 nanometers across, with very sharp corners. Computer simulations showed that when a voltage is applied, electric charge crowds at these sharp tips, producing extremely strong local electric fields—about ten times stronger than on more rounded particles. These intense fields distort the electron cloud of nearby CO2 molecules, turning them from nonpolar, symmetric species into polarized ones with a measurable dipole. That distortion slightly stretches and bends the carbon–oxygen bonds, making the molecules easier to bind and transform on the gold surface. Calculations and experiments together indicate that this field effect makes CO2 adsorption effectively spontaneous and lowers the energy barrier for the key first step in its conversion to CO, so the reaction proceeds faster and at lower energy cost.
A Bioinspired Coat that Blocks Protons
To solve the second problem—excess hydrogen formation—the researchers took inspiration from aquaporins, proteins in the cell membranes of certain acid‑loving microorganisms. Aquaporins allow neutral water molecules through while blocking protons using precisely placed positive charges. Mimicking this idea, the authors coated their gold nanotriangles with a layer of a positively charged surfactant called CTAC. This layer forms a soft, ordered shell whose charged head groups repel incoming protons but do not hinder neutral CO2. Experiments showed that when this cationic coating is present, almost all of the electric current goes into producing CO rather than hydrogen, whereas bare or differently coated gold produces much more hydrogen. Computer models confirmed that the charged layer slows proton transport, raises the local pH right next to the catalyst, and thus suppresses the side reaction.
Performance that Lasts
When the sharp, CTAC‑capped gold triangles were tested in a flowing electrolyzer at pH 1, they produced CO with nearly 100% selectivity over a wide voltage range and kept working for at least 100 hours at high current density. The energy efficiency reached about 60%, which is competitive with or better than many systems that rely on metal salts in less acidic environments. Comparisons with smoother gold shapes and with “rounded‑tip” versions of the triangles showed that both the two‑dimensional triangular form and, crucially, the sharp corners are needed to reach this performance. The work demonstrates a true synergy: geometry‑enhanced electric fields draw in and activate CO2, while the bioinspired charged coating shapes the local chemistry to keep protons away.
What This Means for Future Clean Energy Devices
For non‑specialists, the main message is that this research offers a new recipe for turning CO2 into a useful feedstock under conditions that previously seemed unfavorable. By borrowing ideas from biology and exploiting the physics of sharp tips, the authors show that it is possible to run CO2 conversion in strong acid without added metal ions, avoiding salt buildup and improving CO2 usage. If scaled up and integrated with renewable power, such catalysts could help transform CO2 from a waste product into a building block for carbon‑neutral fuels and chemicals, while making electrochemical devices more robust and easier to operate.
Citation: Chen, L., Guo, Z., Huang, HZ. et al. Electric-field-driven CO2 polarization and bioinspired proton blocking unlock CO2 reduction in strong acid without metal cations. Nat Commun 17, 1734 (2026). https://doi.org/10.1038/s41467-026-68435-y
Keywords: CO2 electroreduction, acidic electrolyzer, gold nanocatalyst, electric field enhancement, proton blocking