Clear Sky Science · en
Control of telomerase recruitment and end protection by independent shelterin components
How Cells Keep Their Chromosome Ends Safe
Every time a cell divides, the very tips of its chromosomes—called telomeres—are shaved down a little. To avoid losing vital genetic information, cells use a special enzyme, telomerase, and a group of protective proteins to maintain these ends. This study explores how two of those proteins, TPP1 and POT1, manage the delicate balance between letting telomerase in to rebuild telomeres and keeping chromosome ends safely sealed off from damage.
The Problem of Aging Chromosome Ends
Telomeres act like plastic caps on shoelaces, preventing chromosomes from fraying or being mistaken for broken DNA. If the cell misreads a chromosome end as damage, it can trigger emergency repair systems that glue chromosomes together or stop cells from dividing. At the same time, telomeres must occasionally open up so telomerase can lengthen them, especially in stem cells that divide many times. Biologists have long imagined telomeres switching between a “closed” protected state and an “open” state that allows telomerase access but risks temporary damage signals. Whether these two states reflect different physical shapes of the telomere or truly separate control mechanisms has remained an open question.
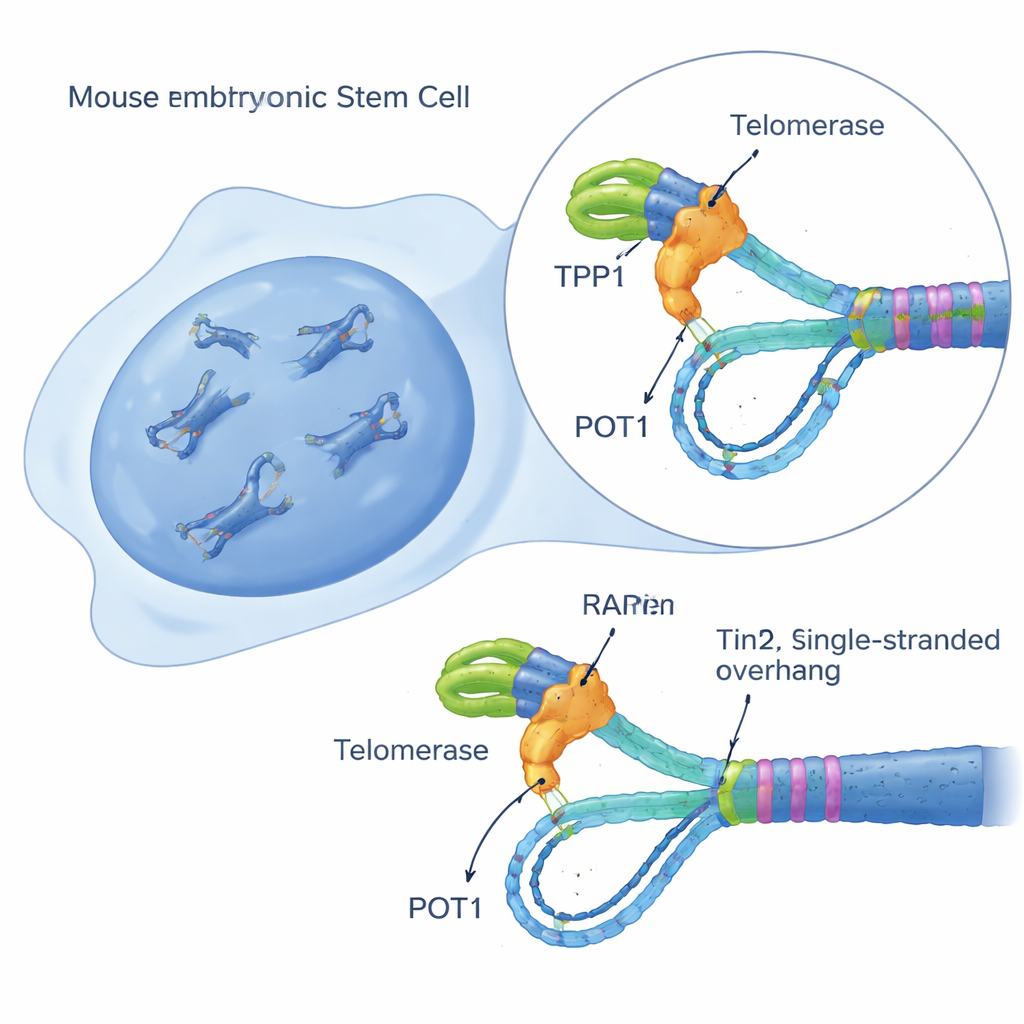
Building a Single-Cell Telomerase Activity Sensor
To untangle this puzzle, the researchers created a sensitive live-cell test, called iTAP (inducible Telomerase Activity Probing), in mouse embryonic stem cells. They engineered the cells to produce a slightly altered version of telomerase’s RNA template only when given the drug doxycycline. This “mutant” template causes telomerase to add a modified DNA sequence at telomeres that can be visually distinguished from normal telomere DNA. Using fluorescent probes, DNA blots, and a sequencing method that enriches telomere fragments, the team could directly see, cell by cell, when and where telomerase was active. Crucially, this system did not harm cell growth or spur a damage response on its own, allowing clean measurements of telomerase behavior inside otherwise healthy cells.
TPP1 Brings In Telomerase, POT1 Holds It Back
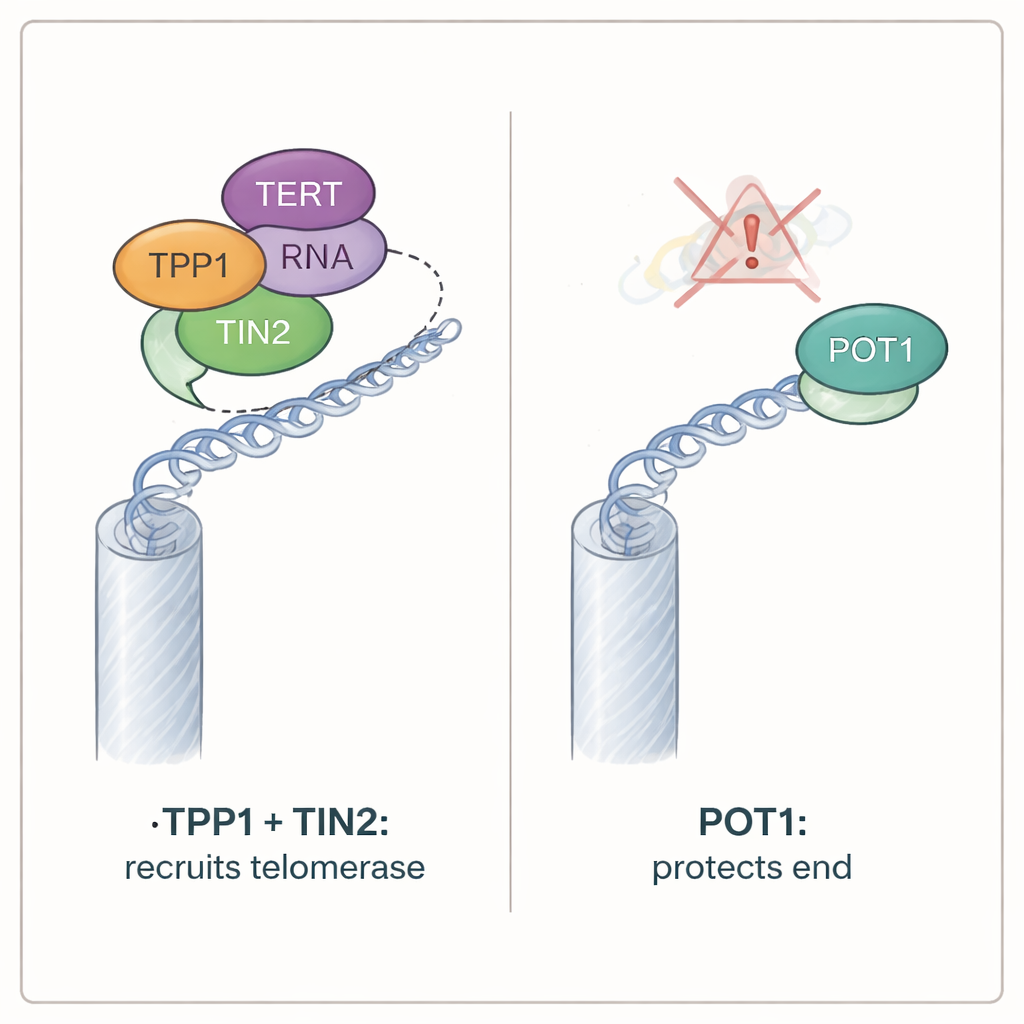
Armed with iTAP, the authors tested how two key telomere proteins, TPP1 and POT1, contribute to telomerase action. Both belong to a larger complex known as shelterin, which coats telomeres. When they removed TPP1 using gene-editing tools, mutant telomere sequences nearly disappeared, even though the mutant telomerase RNA was still produced at normal levels. Further experiments showed that TPP1 must physically link to another shelterin component, TIN2, to recruit telomerase; disrupting TPP1’s connection to TIN2 shut down telomerase activity. Surprisingly, breaking TPP1’s interaction with POT1 had no such effect, and deleting POT1 itself did not reduce telomerase action. In fact, cells lacking POT1 showed a modest increase in the amount of mutant sequence added, suggesting that POT1 normally acts as a brake—limiting how often telomerase engages telomeres, rather than helping it.
POT1 Guards the Ends, While TPP1 Can Be Dispensable
Although POT1 was unnecessary for telomerase recruitment, it proved vital for protecting chromosome ends. Cells lacking both versions of POT1 in mice rapidly stopped proliferating and displayed all the hallmarks of exposed telomeres: DNA damage markers piled up at chromosome tips, a damage-sensing enzyme called ATR switched on, and chromosome ends fused together. By contrast, cells without TPP1 continued to grow and showed little sign of telomere distress, even though they had lost normal telomerase activity. Additional tests showed that POT1’s defensive role depends on its ability to bind the single-stranded overhang at telomeres. Mutant POT1 proteins that could no longer grasp this exposed DNA failed to quell damage signals, even if they could still interact with TPP1. Conversely, versions of POT1 that could bind the overhang but no longer attached to TPP1 still protected chromosome ends.
A New, Modular View of Telomere Control
These findings overturn the simple picture of telomeres flipping between a single “open” state for telomerase and a single “closed” state for protection. Instead, the study shows that telomerase recruitment and end protection are controlled by separable molecular circuits. TPP1, working through TIN2, is the main gateway that brings telomerase to telomeres, while POT1 independently guards the exposed DNA at chromosome ends and keeps damage responses in check. For a layperson, the takeaway is that cells do not rely on a single universal switch at chromosome tips. Rather, they use separate but coordinated knobs—one for letting telomerase in, and another for keeping the ends safe—to maintain genome stability over a lifetime.
Citation: Sandhu, R., Tricola, G.M., Lee, S.Y. et al. Control of telomerase recruitment and end protection by independent shelterin components. Nat Commun 17, 1733 (2026). https://doi.org/10.1038/s41467-026-68433-0
Keywords: telomeres, telomerase, stem cells, DNA damage, chromosome protection