Clear Sky Science · en
Macrocycle-assembled membranes for high-salinity organic wastewater treatment
Cleaning up salty, colorful wastewater
Industries such as textiles, petrochemicals and pharmaceuticals generate huge volumes of wastewater that is both very salty and loaded with brightly colored organic dyes. This mix is hard to treat: methods that remove dyes often also remove the salts, making treatment energy-hungry and expensive. This article reports a new type of “smart” membrane that lets water and dissolved salts pass while holding back large dye molecules, offering a more efficient way to clean and reuse highly contaminated industrial water.
Why salty dye wastewater is so difficult
Conventional treatment plants struggle when organic pollutants and salts appear together at high concentrations. Today’s polymer membranes, typically made from tightly crosslinked polyamides, act like extremely fine sieves. They tend to reject almost everything larger than a water molecule, including both dyes and salts. That sounds good, but in practice it means high operating pressure, high energy use, and large volumes of brine that still need disposal. To reuse water and recover valuable salts, engineers need membranes with pores that are large and well connected enough to pass small ions while still blocking bulky organic molecules.
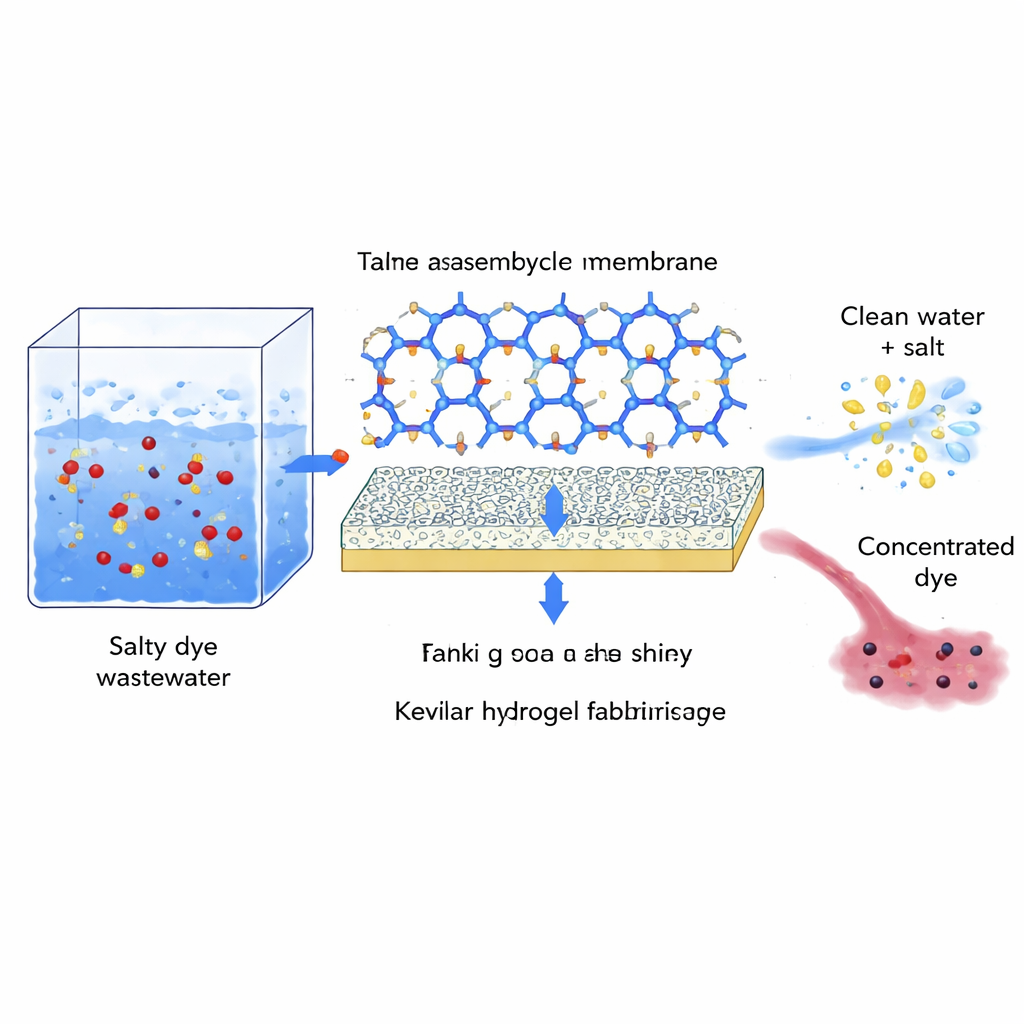
Building a membrane from molecular rings
The researchers tackled this problem by designing a membrane around a specialized ring-shaped molecule called a macrocycle. Their chosen building block, a calixarene bearing four aldehyde groups (TACA), has a rigid three-dimensional “cup” shape with an internal cavity. TACA is oil-loving and stays in an organic liquid, while a small water-loving diamine (MPD) starts in water. Using a technique called unidirectional diffusion-assisted interfacial polymerization, the team places a water-rich Kevlar hydrogel between the two liquids. MPD slowly diffuses through the hydrogel and reacts only where it meets TACA at the interface, linking many TACA rings together into an ultrathin film on the Kevlar support.
Controlling film growth for ideal pores
The Kevlar hydrogel does more than just hold the membrane: it acts as a gentle reactor that evens out monomer supply, absorbs reaction heat and slows diffusion. This produces a smooth, defect-free selective layer about 90 nanometers thick, made of densely packed, hollow nodules whose internal cavities connect into a network of nanochannels. By tuning reaction time and the concentrations of TACA and MPD, the authors control film thickness and compactness, achieving pores about 3.4 nanometers across—large enough for water and hydrated salt ions, but restrictive for bulky dye assemblies. Chemical analyses confirm the intended imine linkages and show many oxygen-containing groups that attract water inside an otherwise hydrophobic framework.
Letting salts through while trapping dyes
In filtration tests, the optimized membrane showed very high water permeability and almost complete rejection of several common dyes, including Congo red and Direct red 23, while allowing most of the dissolved salts to pass. Dyes tend to clump together in water and carry negative charge, so they experience both size-based blockage and electrostatic repulsion from the negatively charged TACA cavities. By contrast, small inorganic ions zip through the interconnected channels. In a realistic test using a salty dye solution, the membrane supported an efficient diafiltration process that reduced salt levels with far less water and energy than a commercial nanofiltration membrane, all while keeping dye losses minimal over many hours of continuous operation.
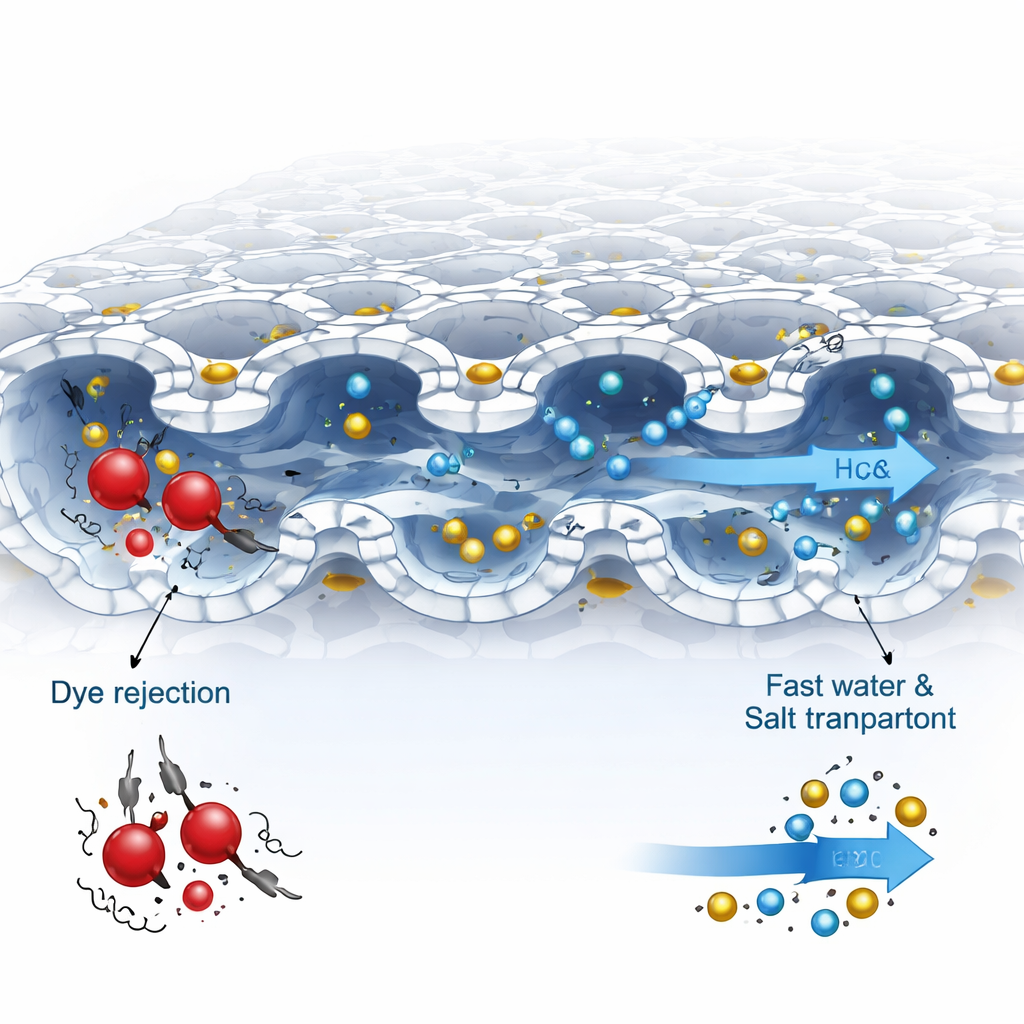
What happens inside the membrane
Computer simulations helped reveal why this structure works so well. Calculations show that water molecules move favorably from the central cavities of the TACA rings down toward rows of hydroxyl groups, forming a low-friction pathway that speeds transport. Molecular dynamics models of the full polymer network highlight a highly porous, interconnected free volume where small ions diffuse readily, while large dye molecules become trapped near the membrane surface. Combined with experimental evidence of negligible material leaching and good thermal stability, these results indicate that the macrocycle-based film is both robust and highly selective.
A gentler route to reusable industrial water
For a non-specialist, the key takeaway is that the authors have turned carefully shaped molecular rings into a kind of programmable sieve. By assembling these rings into a thin, stable membrane with well-controlled pores, they created a filter that can separate dyes from salts in very harsh wastewaters using relatively low pressure and energy. This approach could help factories recover clean water and useful salts from streams that are currently difficult and costly to treat, moving industry closer to true water reuse and a more circular economy.
Citation: Li, Y., Duan, Y., Yuan, J. et al. Macrocycle-assembled membranes for high-salinity organic wastewater treatment. Nat Commun 17, 1731 (2026). https://doi.org/10.1038/s41467-026-68430-3
Keywords: wastewater treatment, membrane filtration, dye removal, salt separation, macrocyclic materials