Clear Sky Science · en
Light-driven restructuring generates nanoisland NiIr alloy for efficient methane dry reforming
Turning Greenhouse Gases into Useful Fuel
Methane and carbon dioxide are two of the most important greenhouse gases warming our planet. This study explores a way to turn both of them, at the same time, into a useful gas mixture called syngas, which can be used to make fuels and chemicals. By harnessing concentrated light instead of burning more fossil fuels for heat, the researchers aim to turn a climate problem into an energy resource.
A New Spin on an Old Industrial Reaction
Industry already knows how to convert methane (the main component of natural gas) and carbon dioxide into syngas through a process called dry reforming. The catch is that it usually needs blast-furnace temperatures of 700–1000 °C, demanding huge energy inputs and often causing the metal catalyst to foul with carbon deposits, or “coke,” that shut the reaction down. The authors of this paper tackle both problems at once. They design a catalyst that uses light to help drive the chemistry and that can resist the slow damage that normally plagues metals under such harsh conditions.
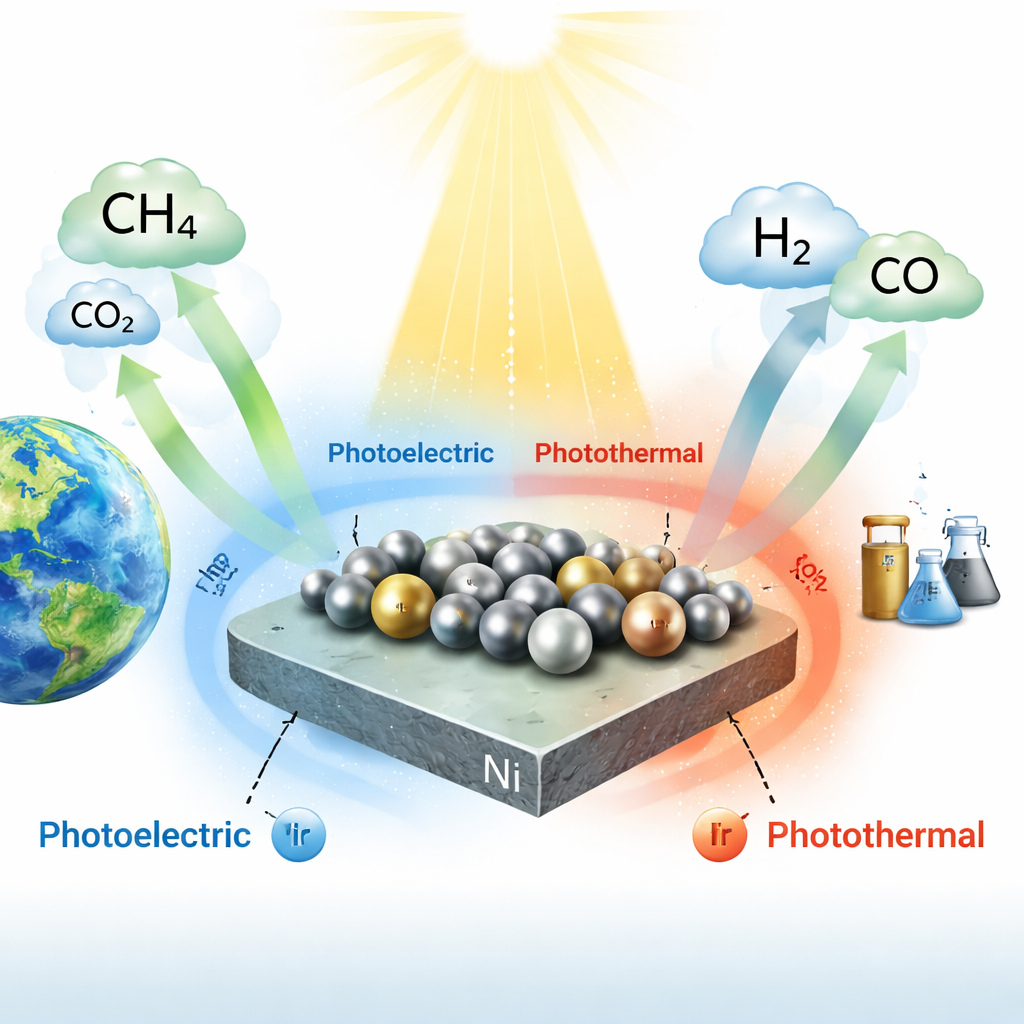
Tiny Metal Islands That Rearrange Under Light
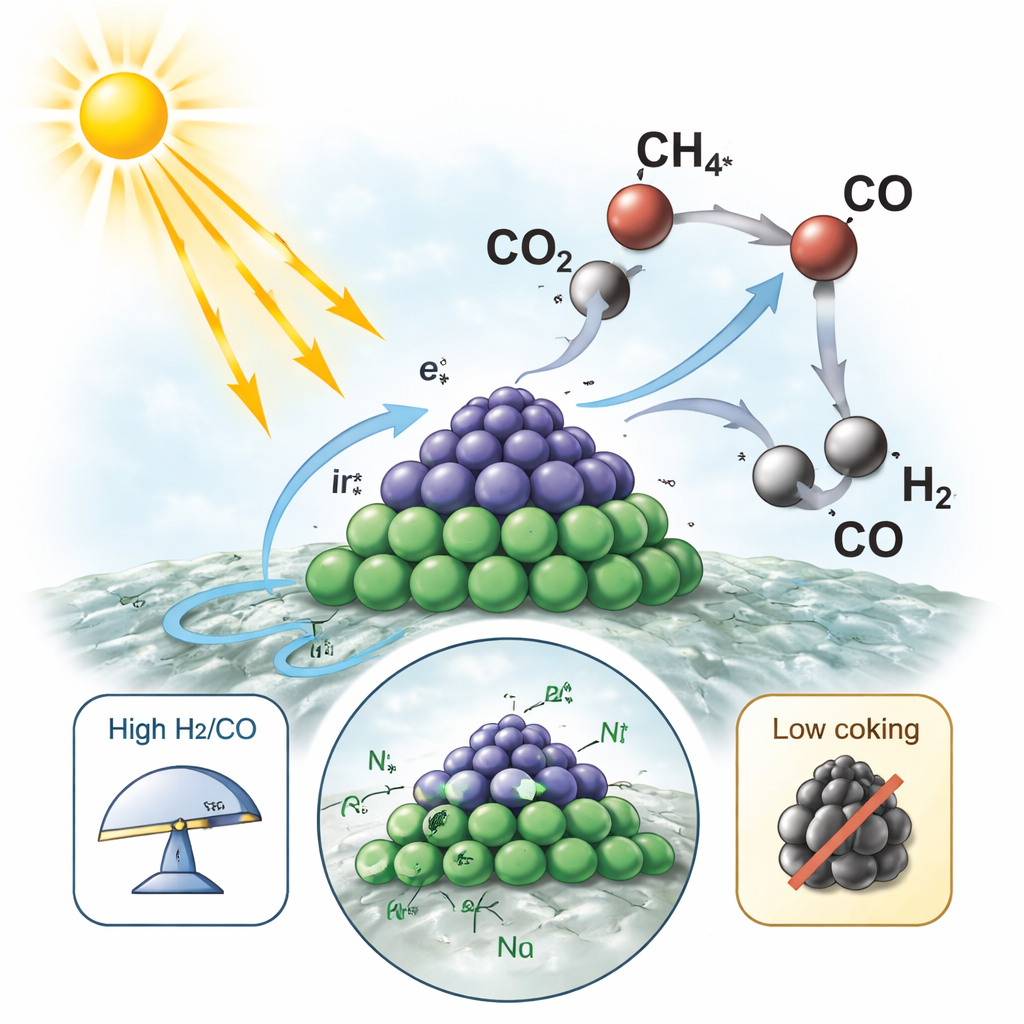
The team builds their catalyst from ultrafine clusters of nickel and iridium—each less than two nanometers across—anchored on sheets of titanium dioxide, a common white pigment that also acts as a light absorber. Rather than simply mixing the metals, they use a stepwise “directional adsorption” method to place iridium where nickel already sits, ensuring the two metals are closely paired. Detailed electron microscopy and X-ray techniques show that, in the dark, these clusters are partly oxidized and strongly connected to the oxide surface. Under illumination, however, the structure reshapes: electrons driven by light move across the interface, allowing iridium atoms to rise and cluster into tiny alloy “islands,” while nickel atoms remain partly oxidized and tethered to the support, acting like linkers that pin the islands in place.
Letting Light Do the Heavy Lifting
When the catalyst is bathed in intense, broad-spectrum light, titanium dioxide and the metal islands absorb photons and create energetic electrons. The authors carefully separate the roles of pure heating and true photoactivity by varying light intensity, cooling the reactor walls with condensation, and comparing with conventional electric heating. They find that photogenerated electrons are responsible for more than half of the syngas production and almost all of the desirable hydrogen-to-carbon monoxide balance, while the warming from light mainly helps molecules move and vibrate. Under optimized conditions, the restructured Ni–Ir nanoislands reach very high reaction rates and a light-to-fuel efficiency of 25 percent—numbers on par with or better than many purely thermal or photothermal systems.
Blocking Carbon Buildup While Steering the Chemistry
To understand why the catalyst stays active, the team tracks the molecules and fragments that land on the surface in real time using infrared spectroscopy, and measures how charges move with ultrafast laser techniques. On the illuminated nanoislands, methane and carbon dioxide are both strongly activated at neighboring nickel and iridium sites, forming short-lived CHxO* species that quickly decompose into hydrogen and carbon monoxide instead of solid carbon. Computer simulations back this picture, showing that the asymmetric pairing of nickel and iridium lowers the energy needed to break the first C–H and C=O bonds and stabilizes oxygen-containing intermediates just enough to keep the reaction moving. In contrast, conventional nickel surfaces tend to crack methane directly into carbon, while pure iridium favors side reactions that skew the gas balance.
From Lab Light to Sunlight
Finally, the researchers take their system outside, using a Fresnel lens to concentrate natural sunlight onto the catalyst. Even under these less controlled conditions, the material maintains high syngas production and good carbon dioxide conversion, and a simple color-changing indicator confirms that carbon monoxide is being produced in real time. For non-specialists, the key takeaway is that carefully designed, light-responsive nanoislands of nickel and iridium can transform waste greenhouse gases into useful building blocks for fuels and chemicals, using the Sun as the main energy input while avoiding the carbon-clogging that usually dooms such catalysts.
Citation: He, C., Yang, R., Zhong, C. et al. Light-driven restructuring generates nanoisland NiIr alloy for efficient methane dry reforming. Nat Commun 17, 1730 (2026). https://doi.org/10.1038/s41467-026-68429-w
Keywords: dry reforming of methane, photocatalysis, syngas, greenhouse gas conversion, NiIr nanoisland catalyst