Clear Sky Science · en
Dynamic allele usage of X-linked genes ameliorates neurodevelopmental disease phenotypes in brain organoids
How the “Quiet” X Chromosome Helps Protect the Brain
Each of a woman’s cells carries two X chromosomes, but for decades biology textbooks have said that one of them is largely shut down—like a spare book kept closed on a shelf. This study shows that, especially in the developing brain, that “silent” X is more like a dynamic backup library. It can be tapped when needed, and this extra flexibility may help explain why many brain development disorders hit males harder than females.
A Hidden Backup System on the X Chromosome
In female mammals, one X chromosome in each cell is switched off early in development to prevent having a double dose of X-linked genes. Yet scientists have long known that some genes can slip through this shutdown. The new work asks a deeper question: is this escape fixed, or can it change as cells develop? Using human stem cells and mini–brain tissues called organoids, the researchers tracked which copy of each X-linked gene—maternal or paternal—was used as cells matured from stem cells into nerve cells. They found that many genes on the supposedly inactive X turn on and off in a stage‑ and cell type–specific way, rather than following a simple on/off rule.
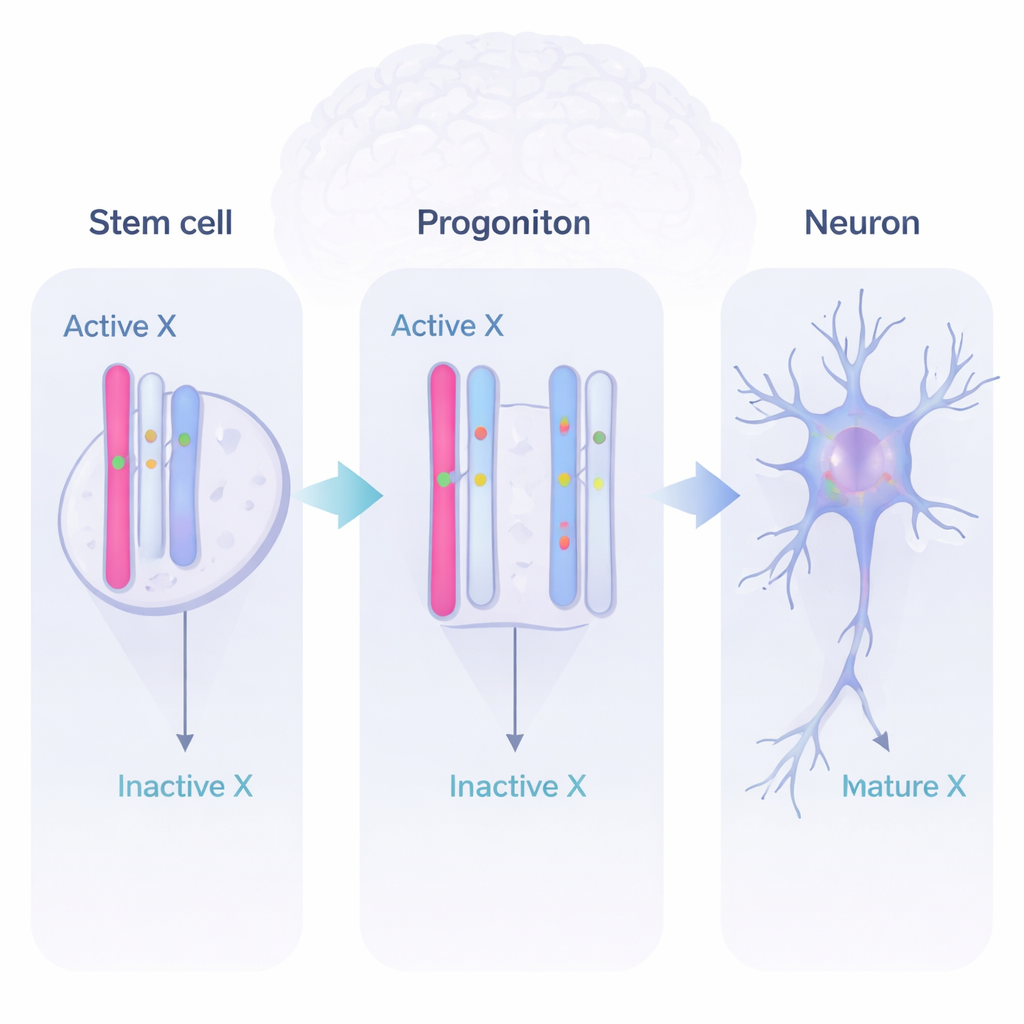
Dynamic Gene Use During Brain Development
By reading RNA (the messages produced from genes) and distinguishing between the two parental copies, the team discovered three major behaviors. Some genes stayed mostly off on the inactive X, as expected. Others were “full escapees,” active from both X copies at all stages. Most intriguingly, a sizable group of genes were dynamic. These genes were silent on the inactive X in stem cells, switched on from both X chromosomes in neural progenitor cells and early neurons, and then some switched off again later. This shows that the inactive X is not a static graveyard of genes but a flexible reservoir that the developing brain can draw on when extra gene activity is useful.
Conserved Patterns and Links to Brain Disorders
When the researchers compared their human data to mouse studies and to chemical marks on DNA from fetal brain tissue, they found that this dynamic escape from X inactivation is seen in other mammals and is tied to chromatin states—features of DNA packaging that control how easily genes can be turned on. The reactivating genes tended to cluster in certain regions of the X chromosome and carried epigenetic signatures of active DNA specifically in female brain tissue. Importantly for medicine, these dynamically reactivated genes were unusually rich in known risk genes for neurodevelopmental disorders such as intellectual disability and autism‑spectrum conditions, suggesting that this flexibility evolved as a protective mechanism.
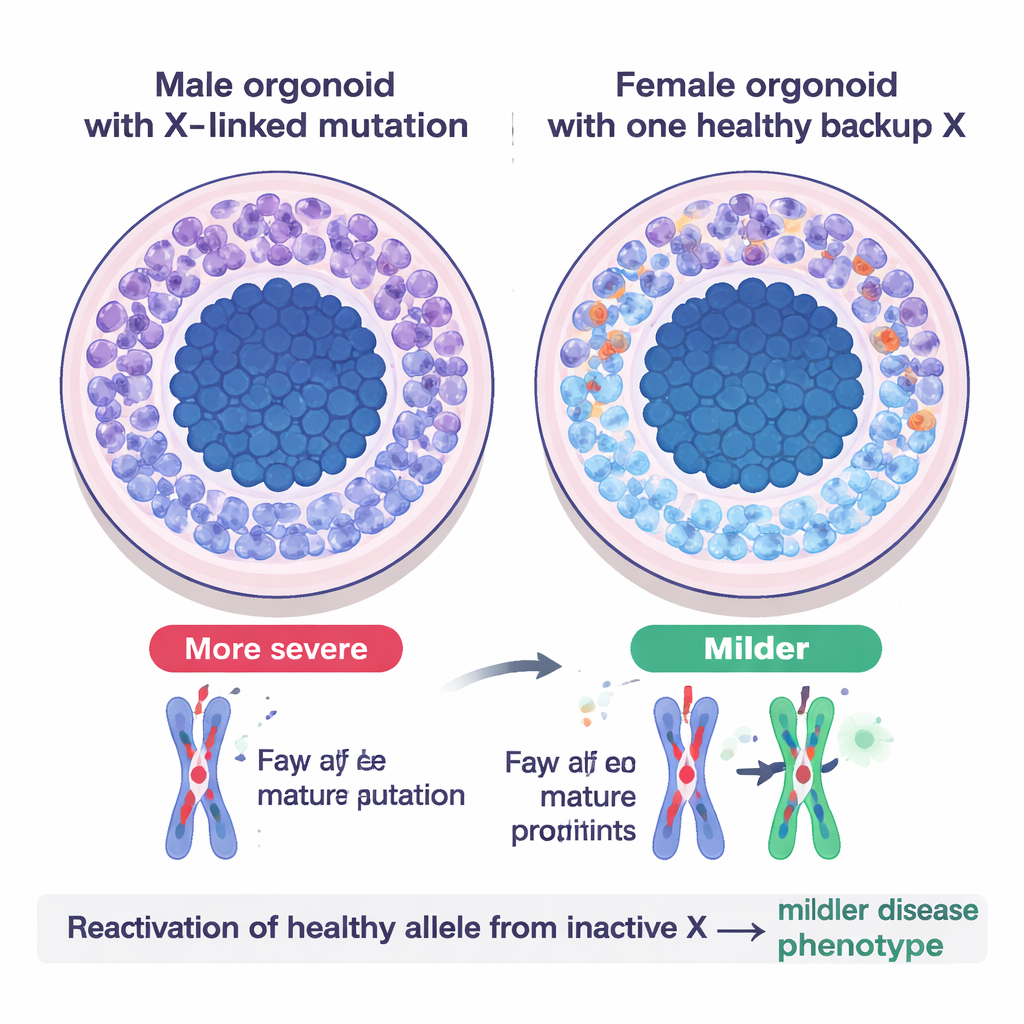
A Real-World Test: Opitz BBB/G Syndrome
To see how this backup system plays out in disease, the team modeled Opitz BBB/G syndrome, a rare X‑linked condition that affects midline brain structures and often causes developmental delay. The disorder is caused by damaging mutations in a gene called MID1 on the X chromosome. In brain organoids grown from male patient cells—where there is only one X copy—neurons were scarce and neural stem cells lingered too long in a dividing state, echoing the brain undergrowth seen in patients. Female organoids with the same mutation on their active X, but a healthy copy on the inactive X, looked noticeably healthier: they produced more neurons and had milder defects. When scientists engineered female lines so that both X copies carried the mutation, the organoids developed severe, male‑like problems, confirming that the previously inactive healthy allele had been reactivated and was helping to compensate.
What This Means for Sex Differences in Brain Disease
The study reveals that the second, “silent” X chromosome in females is not merely genetic ballast. During key windows of brain development, selected genes on this inactive X switch on in specific cell types, expanding the pool of working gene copies. For genes like MID1 that are crucial for building the brain, this dynamic reactivation can soften the impact of damaging mutations and produce milder, more variable symptoms in females. In other words, female brains possess an internal backup system that can be dialed up or down over time, helping to explain why many X‑linked neurodevelopmental disorders are less frequent or less severe in girls than in boys.
Citation: Bertin, M., Todorov, H., Frank, S. et al. Dynamic allele usage of X-linked genes ameliorates neurodevelopmental disease phenotypes in brain organoids. Nat Commun 17, 599 (2026). https://doi.org/10.1038/s41467-026-68428-x
Keywords: X chromosome inactivation, brain organoids, neurodevelopmental disorders, gene dosage, sex differences in disease