Clear Sky Science · en
Shigella flexneri evades septin-mediated cell-autonomous immunity via protein ADP-riboxanation
How Some Gut Bacteria Slip Past Our Inner Security System
Severe diarrhea caused by Shigella bacteria remains a major problem worldwide, especially for young children. Our cells are not defenseless: they have built-in tools that can trap and destroy invading microbes. This study reveals how Shigella flexneri, a leading cause of dysentery, uses clever molecular tricks to dismantle one of these tools, helping the bacteria spread from cell to cell inside the gut.
The Cell’s “Do-It-Yourself” Defense
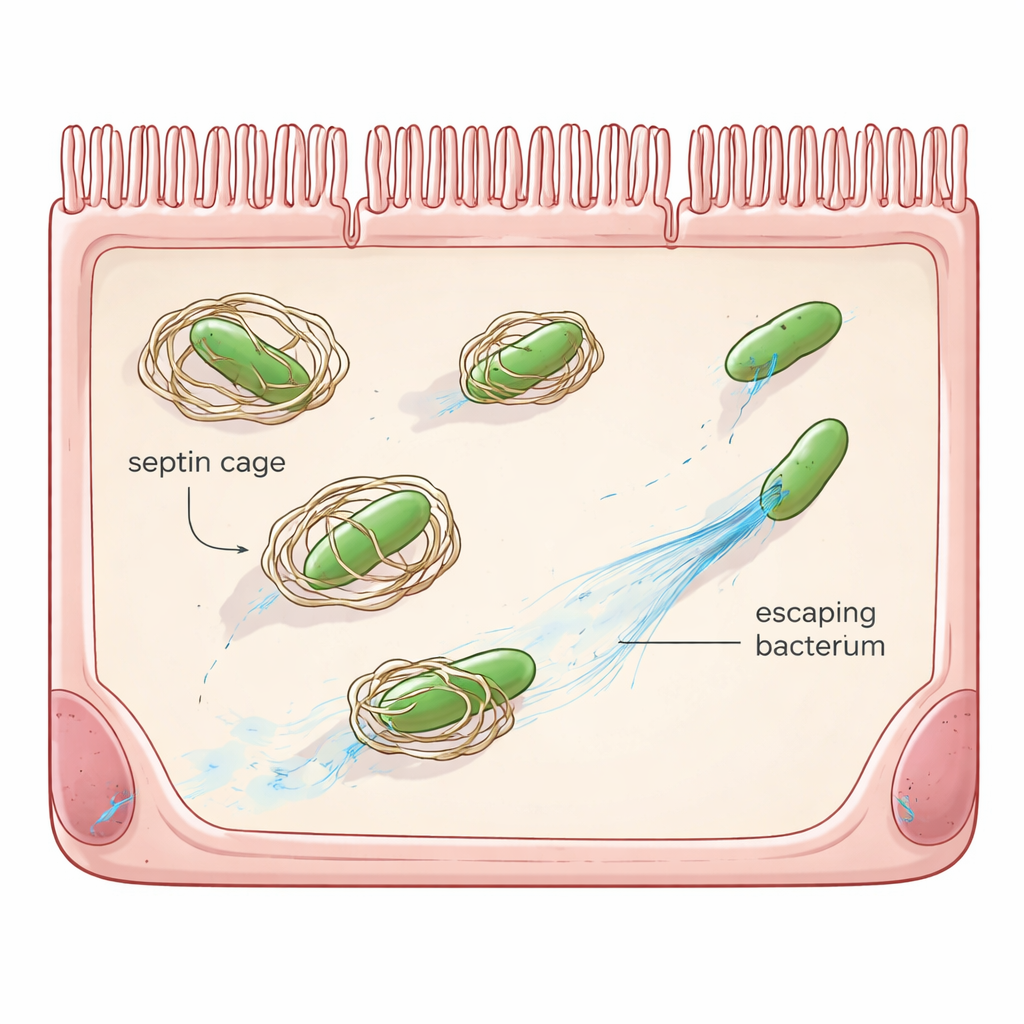
Beyond white blood cells and antibodies, many ordinary cells carry their own emergency security systems, known as cell-autonomous immunity. One such system relies on septins, a family of scaffold proteins that form rings and cage-like structures. When Shigella enters a cell and escapes into the jelly-like interior, septins can assemble around the bacterium, shutting down its ability to move and flagging it for destruction by cellular recycling pathways. Earlier work showed that Shigella already has one countermeasure, a protein called OspG, which tags septins for disposal. Yet even when OspG is missing, most bacteria still avoid being caged, hinting that Shigella has additional tricks.
Unmasking a Second Bacterial Saboteur
The authors turned their attention to another group of Shigella proteins, known as the OspC family. Using a global protein-mapping approach in human cells, they searched for host proteins that became chemically modified when OspC proteins were present. Alongside known targets involved in protein synthesis, they found that several septins, especially one called SEPT9, were consistently marked. Detailed biochemical tests showed that three OspC proteins can directly attach a small energy-rich tag, derived from the molecule NAD, onto many sites in septins. This chemical change, called ADP-riboxanation, is a special, irreversible twist on a more common protein modification.
Breaking the Protein Scaffold from Within
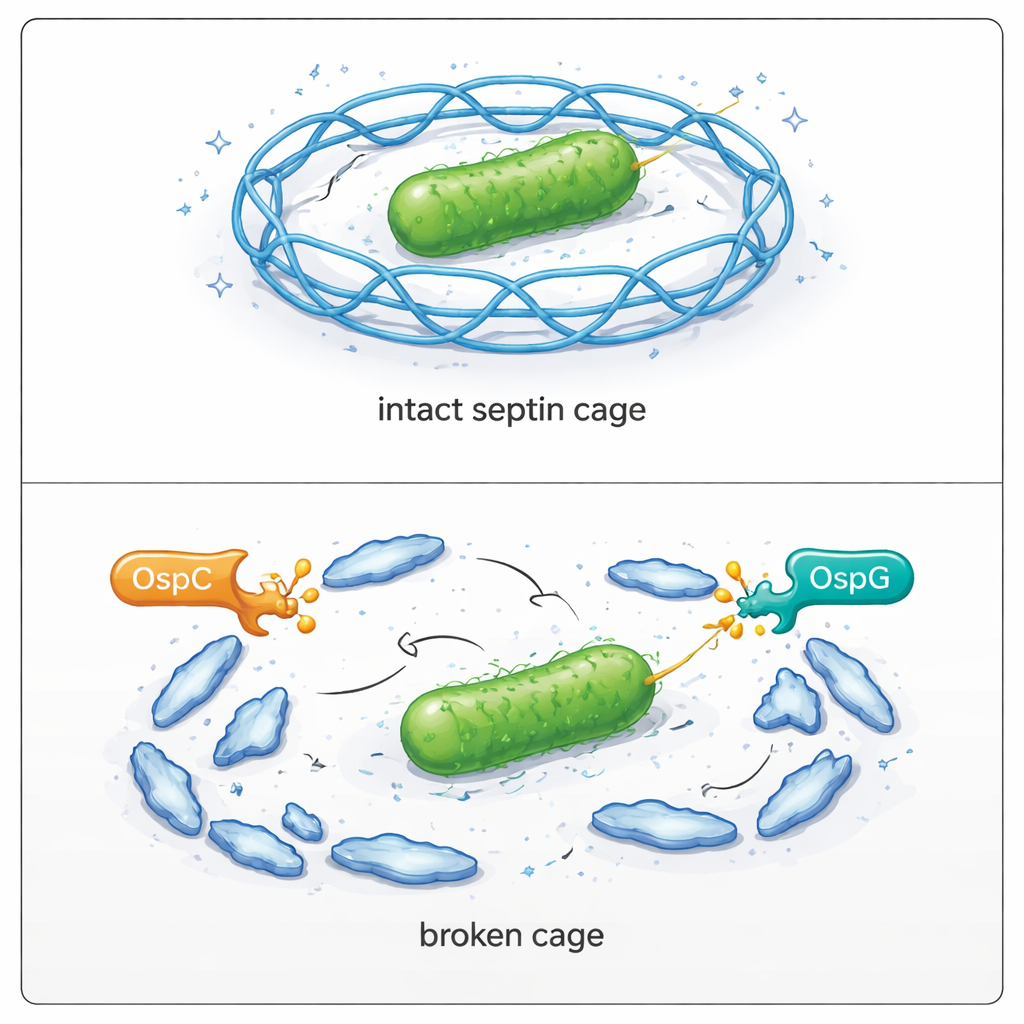
To understand how this chemical tag weakens the cell’s defenses, the team zoomed in on SEPT9. Septins normally form orderly chains of eight units, which then line up into filaments and finally into full cages around bacteria. The researchers discovered that one particular position in SEPT9, an amino acid called arginine 561, is the main site for ADP-riboxanation during infection. Structural work and reconstitution experiments with purified proteins showed that this site lies at a critical contact surface where two SEPT9 units lock together. When that arginine is chemically modified—or replaced with a residue that mimics the added negative charge—the octameric chains fall apart into smaller pieces and can no longer build stable filaments.
Watching Cages Fail Inside Infected Cells
The team then moved back into living cells to see the consequences. In human cells infected with normal Shigella, only a minority of bacteria ended up inside septin cages. When all OspC proteins were genetically deleted, the fraction of caged bacteria nearly doubled. Removing OspG had a similar effect, and deleting both OspC and OspG together raised cage formation even further and severely impaired the bacteria’s ability to spread across a layer of cells. Re-introducing active OspC proteins into the mutant strains restored their escape from cages and their capacity to replicate and form large plaques. Microscopy confirmed that when SEPT9 cannot be modified at arginine 561, it joins cages efficiently; when it is altered by OspC, cage formation around bacteria drops sharply.
What This Means for Fighting Shigella
In accessible terms, this work shows that Shigella carries a matched pair of molecular tools designed to break open the bars of an intracellular “jail.” OspC directly weakens the protein building blocks needed to construct septin cages, while OspG promotes their disposal through another chemical route. Together, these strategies let the bacteria slip free, move within cells, and invade neighboring ones. Understanding these precise escape mechanisms not only deepens our picture of how cell-intrinsic immunity works, but also points to new therapeutic angles: drugs that block OspC or protect key septin contacts could help restore the cell’s natural ability to trap and clear this dangerous pathogen.
Citation: Tang, Z., Xian, W., Özbaykal Güler, G. et al. Shigella flexneri evades septin-mediated cell-autonomous immunity via protein ADP-riboxanation. Nat Commun 17, 1727 (2026). https://doi.org/10.1038/s41467-026-68425-0
Keywords: Shigella, septin cages, cell-autonomous immunity, bacterial virulence factors, ADP-ribosylation