Clear Sky Science · en
Spatial patterning of contractility by a self-organized mechanogen activity gradient underlies Drosophila gastrulation
How tissues sculpt themselves
Early embryos somehow turn simple sheets of cells into intricate three-dimensional forms. This study asks a basic question behind that magic trick: how do cells coordinate where to squeeze and bend so that a tissue folds in the right place and direction? By watching fly embryos at high resolution, the authors uncover a chemical signal that behaves like a “mechanical morphogen,” directly patterning where cells pull on themselves to drive a crucial folding event in development.
A wave that folds the embryo
In the fruit fly Drosophila, a key step in early development is gastrulation, when a flat layer of cells buckles inward to start forming the gut. At the back of the embryo, a patch of cells called the posterior endoderm primordium begins this process by contracting at their upper (apical) sides. This initial bend launches a traveling wave of tissue invagination that sweeps forward across neighboring cells. The authors show that this wave depends on a signal called Fog, a secreted molecule that activates a family of receptors known as GPCRs, which in turn switch on the cell’s contractile machinery built from the motor protein Myosin-II.
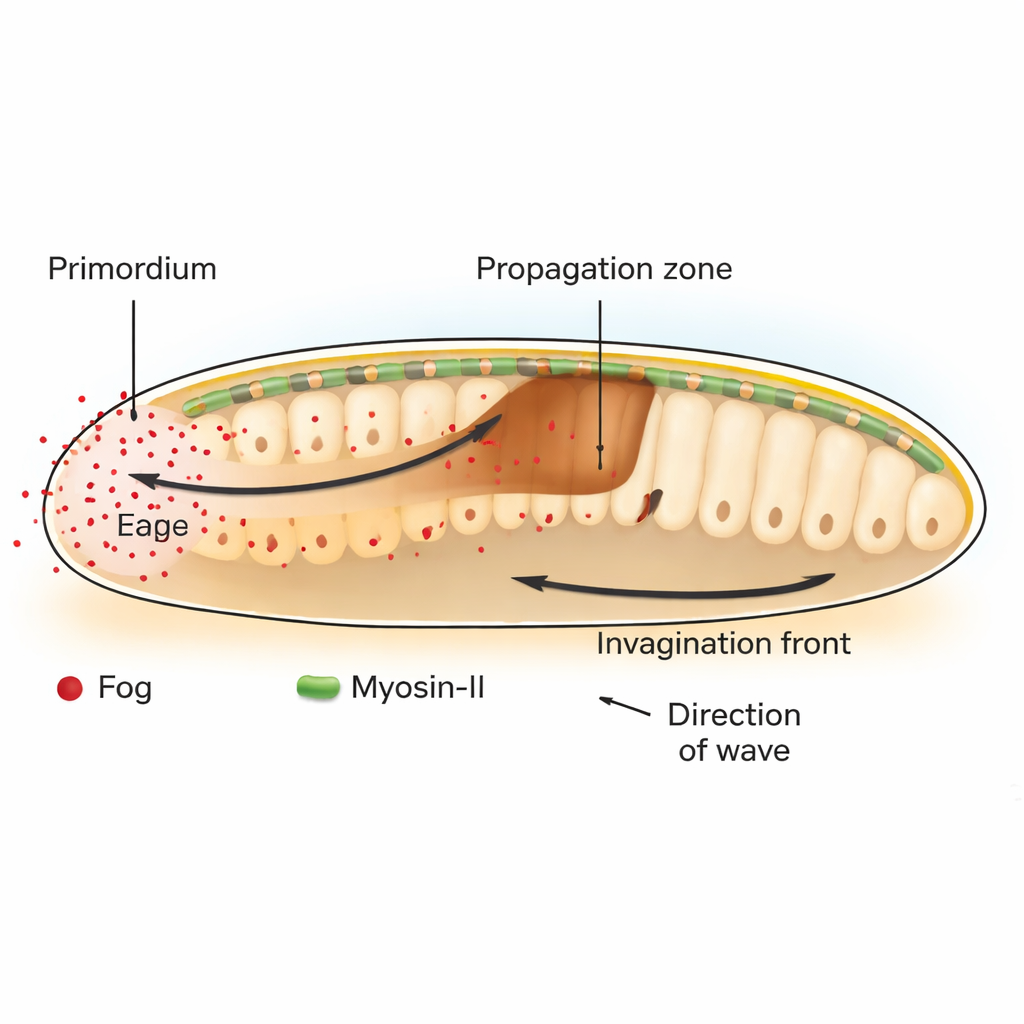
A chemical that patterns force, not fate
Classically, developmental biologists have focused on “morphogens,” diffusing molecules whose concentration gradients tell cells what identity to adopt. Here, Fog behaves differently. Rather than changing what type of cell a region will become, Fog directly adjusts how strongly cells contract. By selectively restoring or blocking Fog and its downstream messenger Gα only in certain regions, the researchers demonstrate that Fog produced in the primordium is both necessary and sufficient to trigger Myosin-II activation and tissue folding at a distance in the adjacent propagation zone. When Fog is tethered so it cannot spread, only the producer cells constrict and the invagination wave stalls after just one or two cell rows.
A hidden gradient on the cell surface
A natural idea is that Fog might form a classic concentration gradient outside the cells. Using a fluorescently tagged version of Fog and sensitive fluorescence measurements, the team finds the opposite: in the thin fluid layer between the tissue and its enclosing shell (the vitelline membrane), Fog is remarkably uniform. Yet Myosin-II activity clearly forms a steep, traveling gradient, strongest right in front of the advancing fold and fading a few cell diameters away. The missing piece is that Fog does not act as a simple bulk concentration cue; instead, a small fraction of Fog becomes briefly trapped at the cell surface, binding to its GPCR receptors and forming a surface-bound activity gradient even though the free Fog molecules remain evenly mixed in the fluid.
Receptors, endocytosis, and adhesion shape the wave
The authors show that this surface-bound Fog, together with its receptors, self-organizes into a pattern. Where Fog repeatedly encounters receptors, it promotes receptor clustering and activation, which recruits more Myosin-II. A cellular “cleanup” process—GPCR endocytosis—then removes active complexes, preventing runaway activation and helping tune how far the signal spreads. When this endocytosis is genetically slowed, the Myosin-II gradient becomes taller and broader. A second key player is a set of adhesion molecules called integrins. As the tissue bends, cells ahead of the fold press against the vitelline membrane; this contact locally activates integrins, which in turn boost the effectiveness of Fog–GPCR signaling. The region of strong integrin engagement matches the extent of the Myosin-II gradient, and without integrins the wave becomes weak and short-ranged even if Fog signaling is otherwise enhanced.
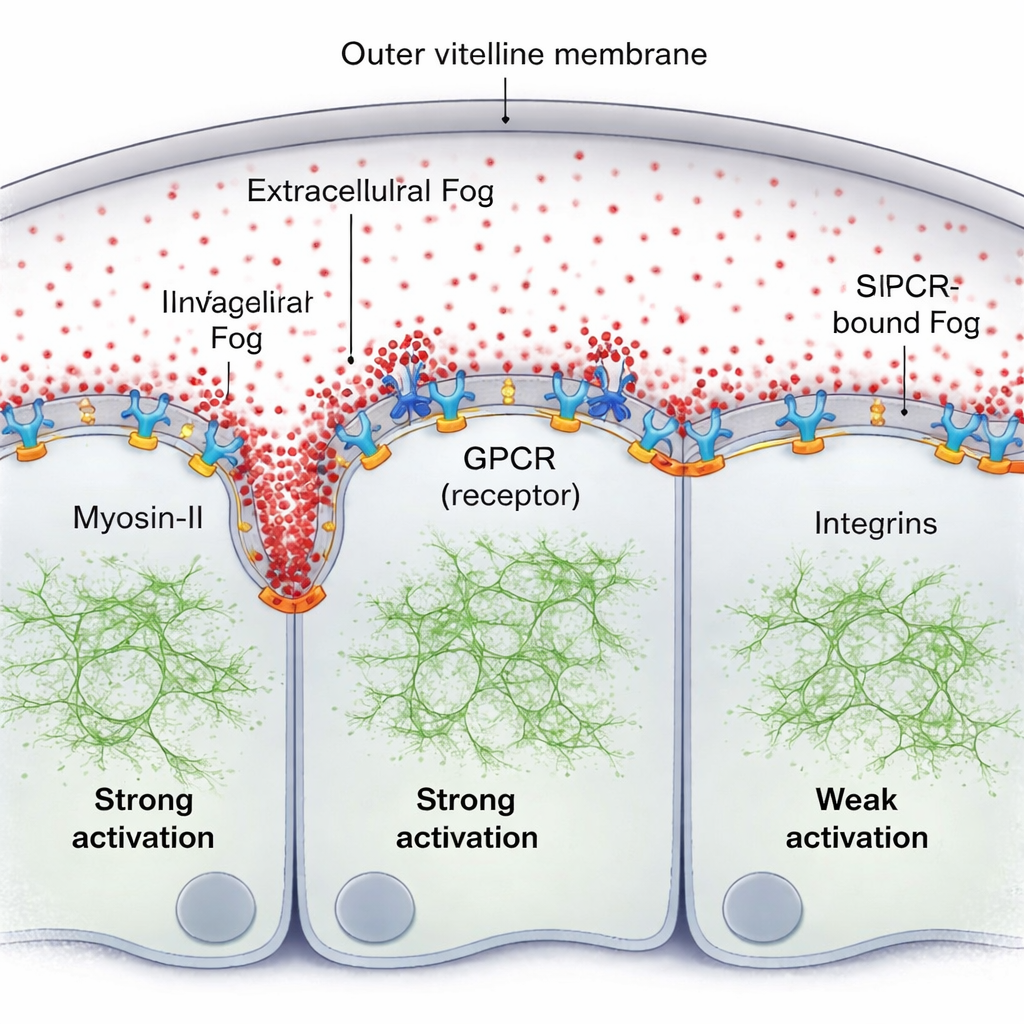
A self-renewing mechanical pattern
To a non-specialist, the main message is that the embryo does not rely solely on pre-drawn chemical maps to decide where to fold. Instead, it builds a moving pattern of mechanical activity on the fly. Fog spreads uniformly, but its effective action is sculpted at the cell surface by receptor clustering, receptor removal, and changing tissue geometry as cells press against their surroundings. This creates a self-organized, traveling gradient of contractile force that pulls the tissue inward in a coordinated wave. The work offers a concrete example of a long-theorized “mechanogen”: a diffusing molecule that directly patterns the physical forces in a tissue, helping explain how living forms so reliably sculpt themselves during development.
Citation: Mundhe, G., Dunsing-Eichenauer, V., Philippe, JM. et al. Spatial patterning of contractility by a self-organized mechanogen activity gradient underlies Drosophila gastrulation. Nat Commun 17, 1721 (2026). https://doi.org/10.1038/s41467-026-68418-z
Keywords: gastrulation, mechanogen, Drosophila embryo, tissue mechanics, Fog signaling