Clear Sky Science · en
A single valine to leucine switch disrupts Plasmodium falciparum AP2-G DNA binding and reveals GDV1’s role in ap2-g activation
How Malaria Parasites Make a Critical Life-or-Death Choice
Malaria parasites living in our blood constantly face a life-changing decision: keep multiplying inside one person, or switch into a form that can jump to the next host through a mosquito bite. This study uncovers how a single tiny change in one parasite protein can completely shut down that infectious, mosquito-ready stage, offering new angles for blocking malaria transmission.
The Parasite’s Fork in the Road
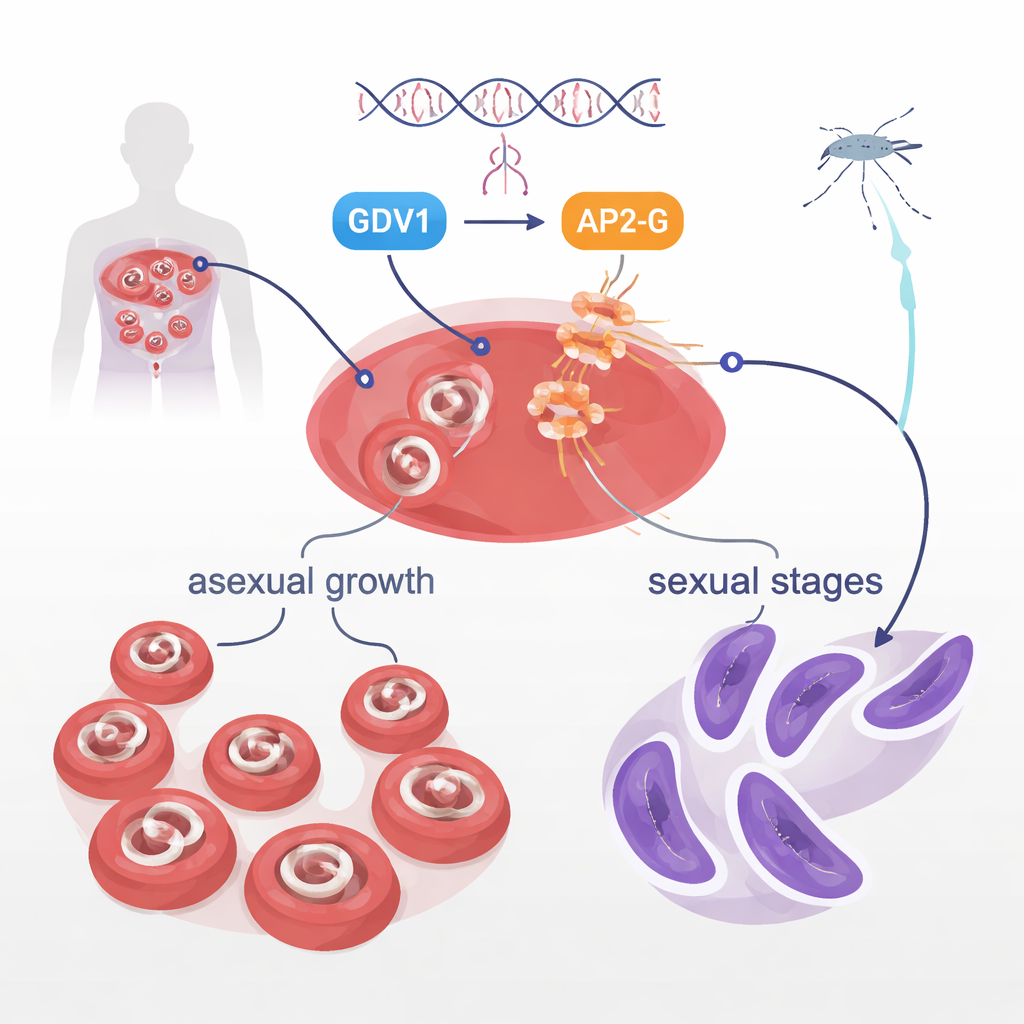
In the bloodstream, Plasmodium falciparum parasites mostly focus on rapid growth, which causes illness. But a small minority transform into sexual forms called gametocytes, the only stages mosquitoes can pick up and pass on. How only some parasites in a crowd make this switch has been a long-standing mystery. Earlier work showed that two parasite proteins, GDV1 and a DNA-binding protein called AP2-G, sit at the heart of this decision. GDV1 helps awaken a normally silent ap2-g gene, and AP2-G in turn turns on a suite of genes that drive sexual development. The current study set out to explore another protein, a kinase called STK2, but instead stumbled onto a much more dramatic finding in AP2-G itself.
A Single Amino Acid That Stops Transmission
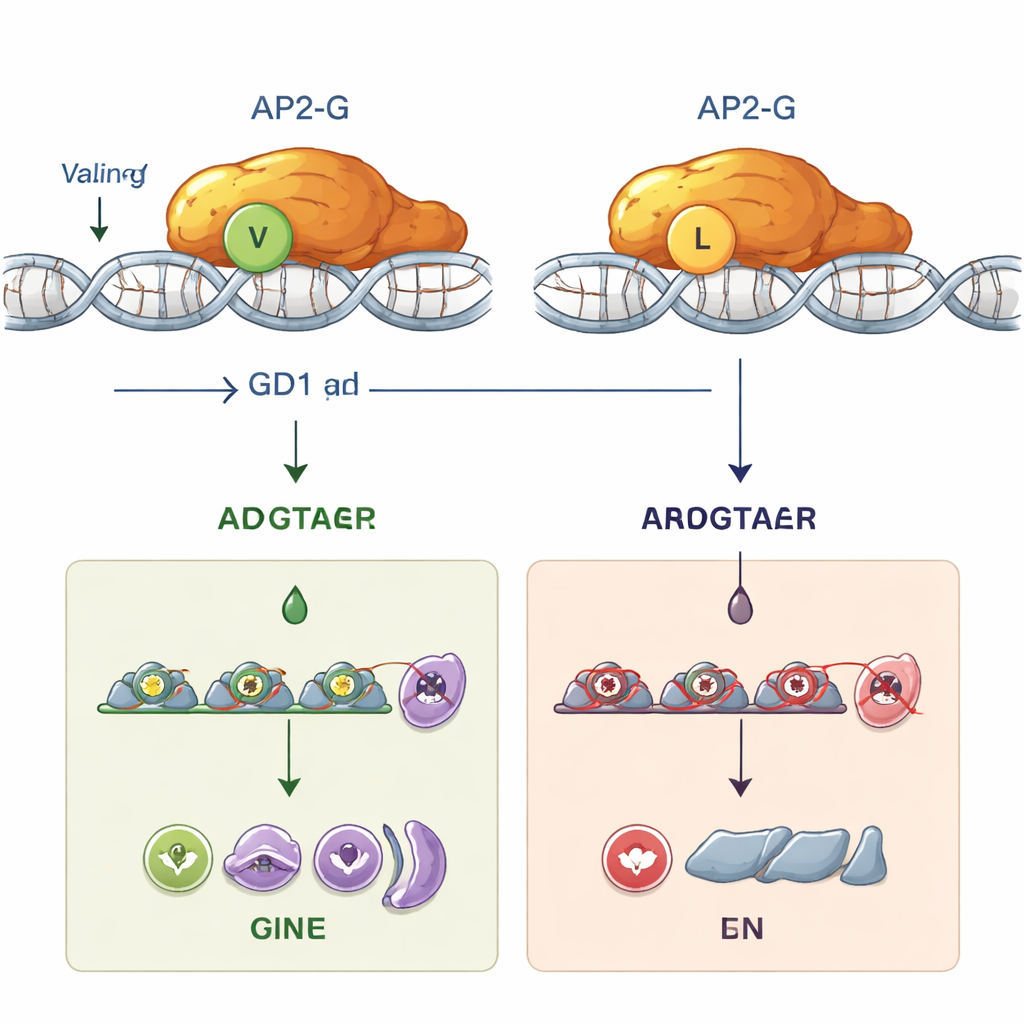
While deleting the stk2 gene in one lab parasite strain, the researchers noticed that these parasites completely lost their ability to produce gametocytes, yet still grew asexually just fine. Surprisingly, when they repeated similar manipulations in another strain, gametocytes formed normally. Whole-genome sequencing revealed the hidden culprit: a single “letter” change in the ap2-g gene, swapping one amino acid, valine, for a very similar one, leucine, at position 2163. This position lies at the very beginning of AP2-G’s DNA-binding region, the part that physically grips specific DNA motifs to turn on genes. This tiny valine-to-leucine switch alone was enough to abolish gametocyte formation. When scientists engineered this mutation into otherwise normal parasites, gametocytes disappeared; when they reversed it back to valine, sexual development was fully restored.
How the Switch Breaks the Molecular Lock
To understand why this subtle change is so devastating, the team combined computer modeling with lab experiments. Structure predictions suggested that replacing valine with leucine shifts and destabilizes the precise shape of the AP2 DNA-binding domain. In test-tube assays using purified proteins, normal AP2-G bound strongly to its preferred DNA sequence, a short code containing the motif “GnGTAC.” In contrast, the mutant AP2-G with leucine at position 2163 failed to bind this sequence at all. Without stable DNA binding, AP2-G could no longer turn on its own gene (a positive feedback loop known as autoregulation) or activate dozens of downstream genes needed to push parasites into the sexual pathway. Functionally, the mutant parasites became “sterile”: they could not make the forms that infect mosquitoes, even though they still flourished in red blood cells.
GDV1’s Hidden Role Before AP2-G Takes Over
The mutation also gave researchers a unique tool: parasites in which the ap2-g gene can be switched on, but AP2-G cannot bind DNA. Using glowing and light-emitting reporters, they tracked when different players act during the parasite’s 48-hour cycle in red blood cells. They found that GDV1 protein appears first, early in a stage called schizogony, and is essential to kick-start ap2-g expression from its silenced state. This early activation happened whether ap2-g encoded normal AP2-G or the mutant version. Only later, once enough normal AP2-G built up, did the strong self-boosting loop and the activation of other “sexual” genes occur. A key marker, a protein called MSRP1, lit up only in parasites with functional AP2-G, providing a handy way to distinguish early and late sexually committed parasites. In mutant lines with the leucine switch, GDV1 could still wake up ap2-g, but the broken AP2-G protein could not carry the process forward, so sexual development stalled.
Implications for Stopping Malaria Spread
For a general reader, the punchline is simple: malaria parasites rely on an extremely sensitive molecular lock to decide whether they will be transmissible. This study shows that changing just one “tooth” in that lock—a single valine in AP2-G’s DNA-gripping region—prevents the parasite from ever making mosquito-infectious stages. At the same time, it clarifies that another protein, GDV1, acts earlier as the key to first unlock the silent ap2-g gene, before AP2-G strengthens its own production and turns on a wider sexual program. By mapping this sequence of events and creating reporter parasite lines that glow when each step occurs, the work provides powerful tools to screen drugs or human factors that interfere with sexual commitment. In the long run, targeting AP2-G’s DNA-binding region or the GDV1-driven activation step could form the basis of new strategies that don’t just cure malaria in one patient, but cut the chain of transmission altogether.
Citation: Prajapati, S.K., Dong, J.X., Morahan, B.J. et al. A single valine to leucine switch disrupts Plasmodium falciparum AP2-G DNA binding and reveals GDV1’s role in ap2-g activation. Nat Commun 17, 1719 (2026). https://doi.org/10.1038/s41467-026-68416-1
Keywords: malaria transmission, Plasmodium falciparum, gametocyte development, AP2-G, GDV1