Clear Sky Science · en
Multi-modal AI for opportunistic screening, staging and progression risk stratification of steatotic liver disease
Why liver fat and scarring matter to everyone
Fatty liver disease is no longer rare or niche: about one in three adults worldwide has extra fat in the liver, and that number is climbing. In many people it stays silent, but in others it leads to dangerous scarring (fibrosis), cirrhosis, liver cancer, and heart problems. At the same time, millions of people already receive CT scans for other reasons—chest pain, cancer follow‑up, or routine checkups—without anyone looking closely at their liver. This study asks a simple but powerful question: could artificial intelligence (AI) quietly scan those existing images, flag hidden liver disease, and help doctors act before serious damage occurs?
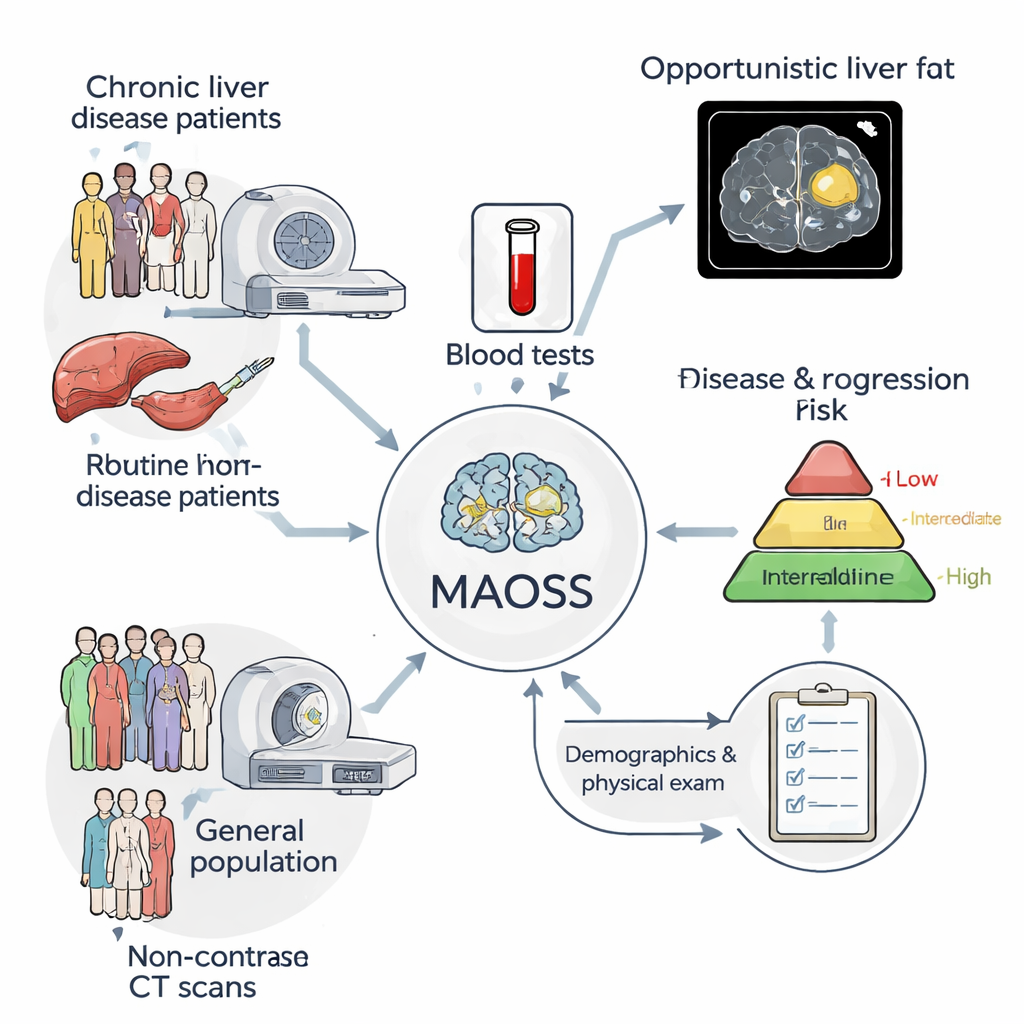
A new AI helper built from real‑world hospital data
The researchers developed a multi‑modal AI system called MAOSS (Multi‑modal AI for Opportunistic hepatic Steatosis Screening). Instead of relying on a single type of information, MAOSS combines three: 3D non‑contrast CT images of the liver, standard blood test results, and basic clinical details such as age and body size. The team trained the system on more than 2,000 patients from a major Chinese hospital, including nearly 1,000 whose liver tissue had been examined under the microscope (the gold standard) and over 1,100 whose scans had detailed radiology reports. This mix allowed the AI to learn from both the most precise labels (biopsies) and the broader, easier‑to‑collect reports used in daily practice.
Teaching the AI to read both fat and scar tissue
MAOSS was designed to answer two key questions from each CT scan: how much fat is in the liver (steatosis) and how advanced any scarring is (fibrosis). To do this, the model treats disease stages as an ordered ladder—from none, through mild and moderate, to severe—and learns to place each patient on the right rung. A special “multimodal” design lets the system flexibly handle missing information; for example, it can still work when some blood tests are unavailable and rely more heavily on imaging. The researchers also added an explanation tool based on “integrated gradients,” which highlights the specific regions and densities within the liver image that most strongly influence the AI’s decision, giving clinicians a heat map of suspected fatty change.
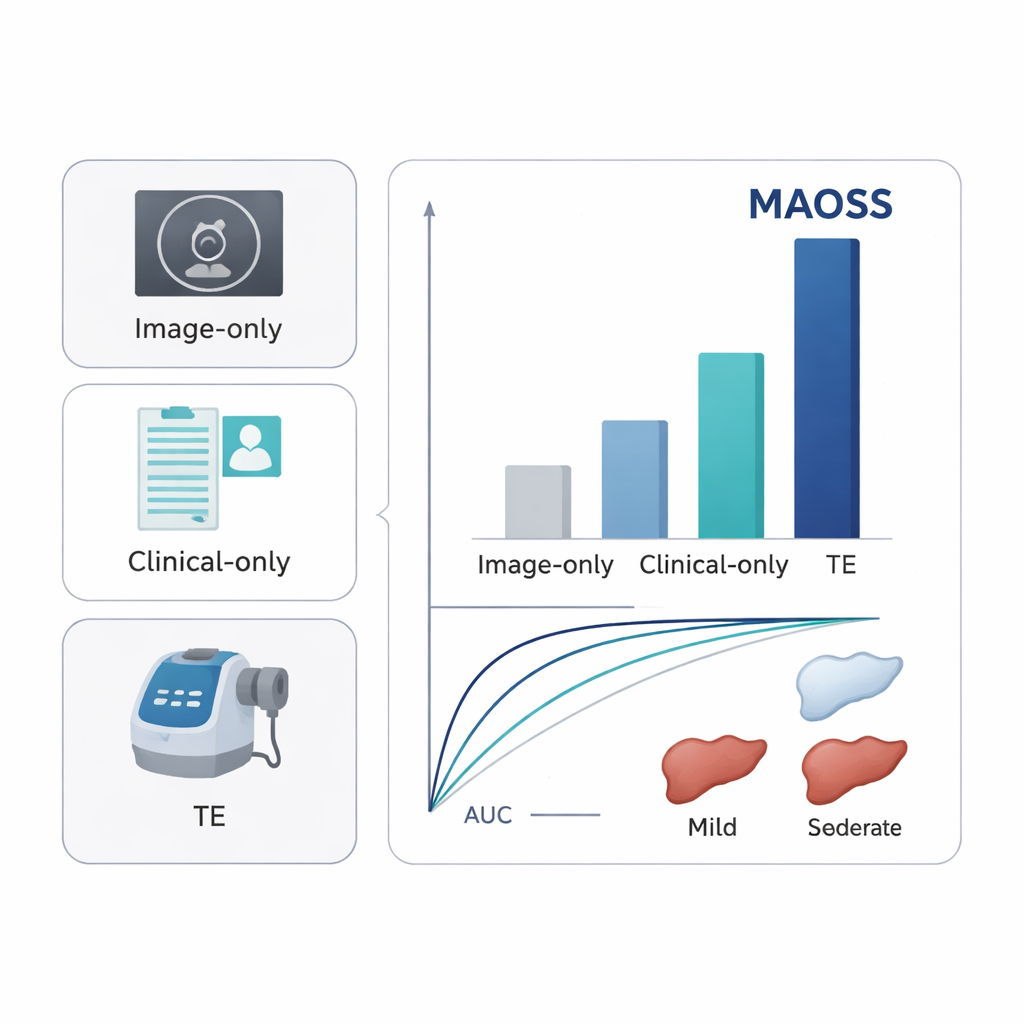
How well MAOSS performs compared with current tools
When tested on separate groups of patients from several hospitals—including an external cohort and a group with MRI‑based liver fat measurements—MAOSS showed high accuracy for detecting even mild liver fat, with areas under the ROC curve (AUCs) around 0.90–0.93. It also performed strongly in identifying clinically important fibrosis, with AUCs around 0.82–0.89. These scores were consistently better than models that used only images, only clinical data, or standard ultrasound‑based measures such as transient elastography. In a reader study with 11 radiologists, MAOSS served as an assistant: when doctors saw the AI score alongside the CT, their ability to pick up early‑stage fatty liver improved noticeably, especially in distinguishing normal livers from those with subtle disease.
Turning routine scans into an early‑warning network
The team then asked how MAOSS might work in the messy reality of everyday medicine. They applied the system to more than 18,000 real‑world CT scans from emergency rooms, inpatients, outpatients, and health‑check centers, most of which were originally ordered for reasons unrelated to the liver. MAOSS’s assessments of “fatty liver” versus “no fatty liver” agreed closely with radiologists’ reports, particularly in large physical‑examination cohorts. Next, they plugged MAOSS into an established clinical guideline that decides which patients with fatty liver should be referred for specialist care. In a biopsy‑confirmed group of 1,192 people, the MAOSS‑enhanced pathway identified about one‑third more patients at risk of progressing to steatohepatitis or advanced fibrosis than the standard approach based on ultrasound measures alone, while still safely ruling out low‑risk individuals.
What this means for patients and future care
For a layperson, the key message is that the same CT scans already being done for other health issues could double as a quiet screening system for liver disease, without extra appointments or invasive procedures. By automatically reading CT images alongside routine blood tests, MAOSS can catch fatty liver and worrisome scarring earlier than traditional methods, help radiologists see subtle disease they might otherwise miss, and more accurately sort patients into low, intermediate, and high risk for progression to cirrhosis. While the authors note that larger, longer‑term studies are needed and the AI is not perfect, their results suggest that multi‑modal AI could become an important part of preventing severe liver disease before it silently advances.
Citation: Gao, Y., Li, C., Chang, W. et al. Multi-modal AI for opportunistic screening, staging and progression risk stratification of steatotic liver disease. Nat Commun 17, 1562 (2026). https://doi.org/10.1038/s41467-026-68414-3
Keywords: fatty liver disease, medical AI, CT imaging, liver fibrosis, opportunistic screening