Clear Sky Science · en
Hierarchical mechanisms control the clearance of DNA lesion–stalled RNA polymerase II
When Gene Copying Machines Hit a Roadblock
Every second, millions of tiny molecular machines called RNA polymerase II travel along our DNA, copying genes into RNA. But when the DNA is damaged — for example by ultraviolet (UV) light from the sun — these machines can grind to a halt. If stalled copies pile up, cells can no longer read their genes properly, which is especially dangerous for brain cells and can lead to rare disorders like Cockayne syndrome. This study uncovers, in detail, how our cells sense these blocked machines and either restart or remove them before they cause lasting harm.
A New Way to Watch Cellular Traffic Jams
To understand how cells clear blocked polymerases, the researchers built a time-resolved "traffic camera" for transcription. They used a drug that lets existing RNA polymerase II molecules keep moving but prevents new ones from starting the journey, and then created a tiny patch of UV damage inside each cell nucleus. By following a specific chemical tag on the active form of the polymerase, they could watch how quickly it disappeared from the damaged zone compared to the rest of the genome. In parallel, they developed a companion test that looked at the total amount of active polymerase in cell extracts, revealing not just when polymerases left the DNA, but when they were broken down by the cell’s disposal system.
Tagging the Stalled Machines for Removal
The team focused on transcription-coupled repair, a specialized repair system that fixes lesions on actively read genes. They used a matched set of human cell lines, each missing a different repair factor. Two proteins, CSB and CSA, emerged as decisive gatekeepers. When either was absent, polymerase II accumulated and stubbornly remained at damage sites, and the cell failed to degrade it. Biochemically, these cells also failed to attach small ubiquitin tags to a key spot on the polymerase. In contrast, cells lacking later repair proteins — those that cut out and replace the damaged DNA — still cleared polymerase normally. This showed that the initial ubiquitin tagging of stalled polymerase is the critical trigger for deciding its fate.
Two Cleanup Crews: One Fast, One as Backup
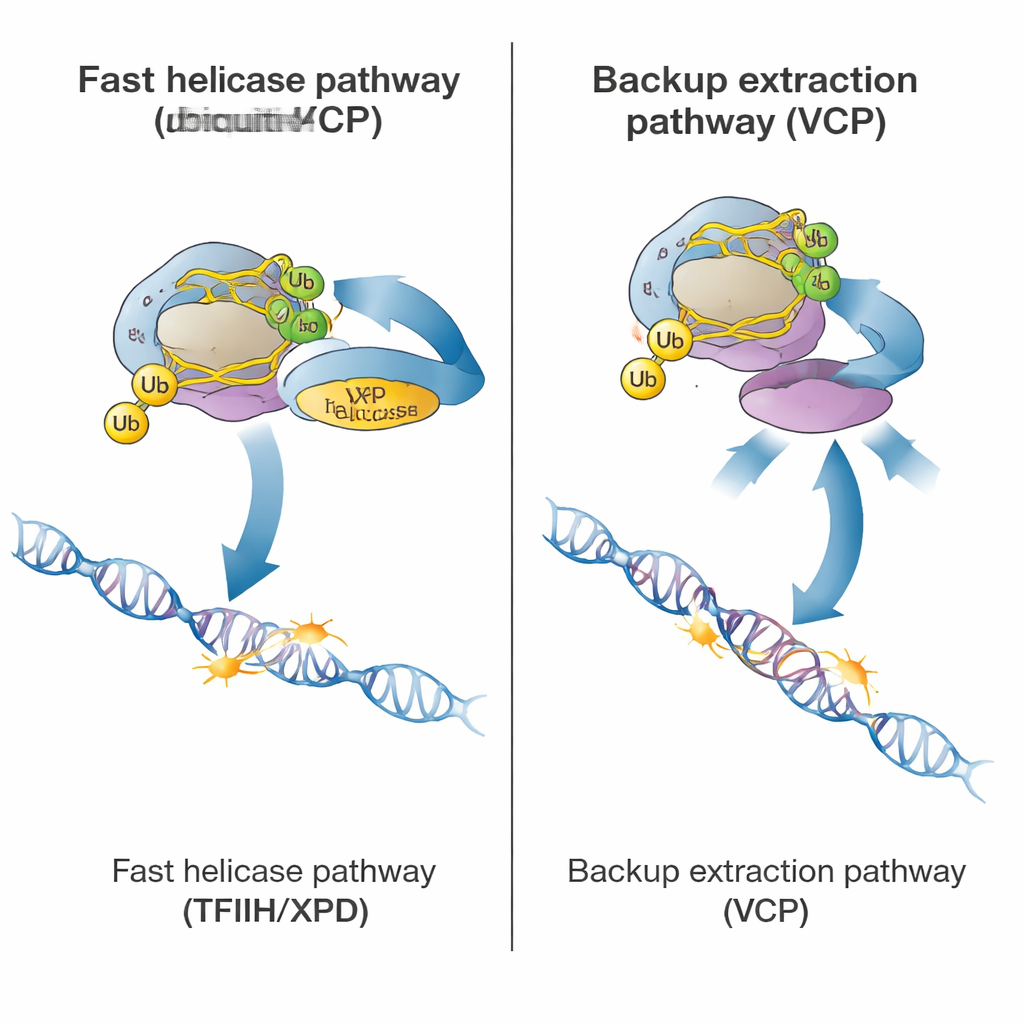
Once polymerase is tagged, the cell can choose between two ways to clear it. The primary, fast route relies on a large repair complex called TFIIH, and in particular its XPD helicase subunit, which uses energy to unwind DNA. Helper proteins, including ELOF1, UVSSA, and STK19, bring TFIIH to the stalled polymerase and position XPD on the DNA just ahead of it. Using new tests in patient cells that carry a helicase-dead XPD mutation, the authors showed that when XPD cannot unwind DNA, polymerase clearance slows dramatically, even though the rest of the repair machinery is present. This indicates that the mechanical pulling action of XPD is what normally "shakes" the polymerase loose from the lesion so repair enzymes can reach the damaged spot.
A Slow but Essential Plan B
The study also revealed a slower, emergency route for clearing stalled polymerase. This pathway depends on VCP (also known as p97), a protein that recognizes ubiquitin tags and can forcibly extract proteins from chromatin. In healthy cells with fully working TFIIH, blocking VCP had only a minor effect. But in cells where TFIIH was missing, mispositioned, or helicase-dead, polymerase clearance became almost entirely dependent on VCP. In these situations, VCP could still pull the tagged polymerase away from DNA, even when normal repair could not proceed. Crucially, this backup route still required some ubiquitin tagging, explaining why cells lacking CSB or CSA — and thus lacking ubiquitin marks — failed in both the main and backup pathways.
Why This Matters for Health and Disease
Put together, the work outlines a hierarchical safety program that cells use whenever RNA polymerase II runs into DNA damage. First, CSB and CSA flag the stalled machine with ubiquitin. If everything is working properly, TFIIH and its XPD helicase then quickly dislodge the polymerase so the lesion can be cut out and repaired. If TFIIH cannot do its job, VCP steps in to extract the polymerase and send it for degradation, preventing it from clogging gene expression even though the DNA damage itself remains. This framework helps explain why inherited defects in CSB or CSA cause particularly severe neurological problems: without ubiquitin tagging, cells lose both the primary repair-driven route and the backup extraction route, leaving stalled polymerases stuck on damaged genes and transcription chronically blocked.
Citation: van der Meer, P.J., Yakoub, G., Tsukada, K. et al. Hierarchical mechanisms control the clearance of DNA lesion–stalled RNA polymerase II. Nat Commun 17, 1647 (2026). https://doi.org/10.1038/s41467-026-68413-4
Keywords: transcription-coupled DNA repair, RNA polymerase II, UV-induced DNA damage, protein ubiquitylation, Cockayne syndrome