Clear Sky Science · en
Phosphorothioate DNA modification by BREX type 4 systems in the human gut microbiome
Hidden Chemical Tweaks in Our Gut Bacteria
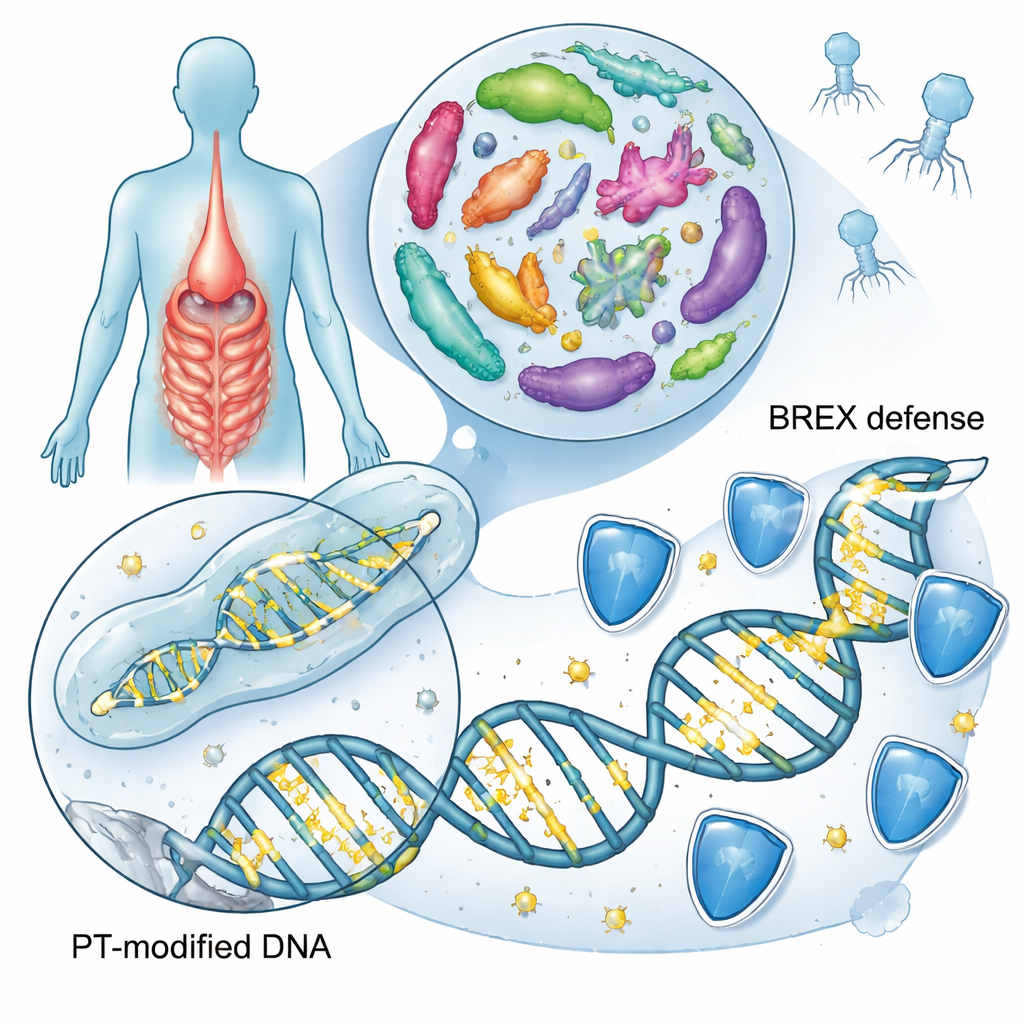
Deep inside the human gut, trillions of bacteria are constantly trading genes, fighting viruses, and reacting to the chemistry of what we eat. This study reveals that many of those microbes quietly rewrite the backbone of their own DNA by swapping in sulfur atoms, creating a special mark called a phosphorothioate. The work uncovers a new version of this system, linked to a bacterial defense toolkit called BREX type 4, and explores how widespread these sulfur marks are in gut bacteria that may influence health and disease.
A Different Kind of DNA Mark
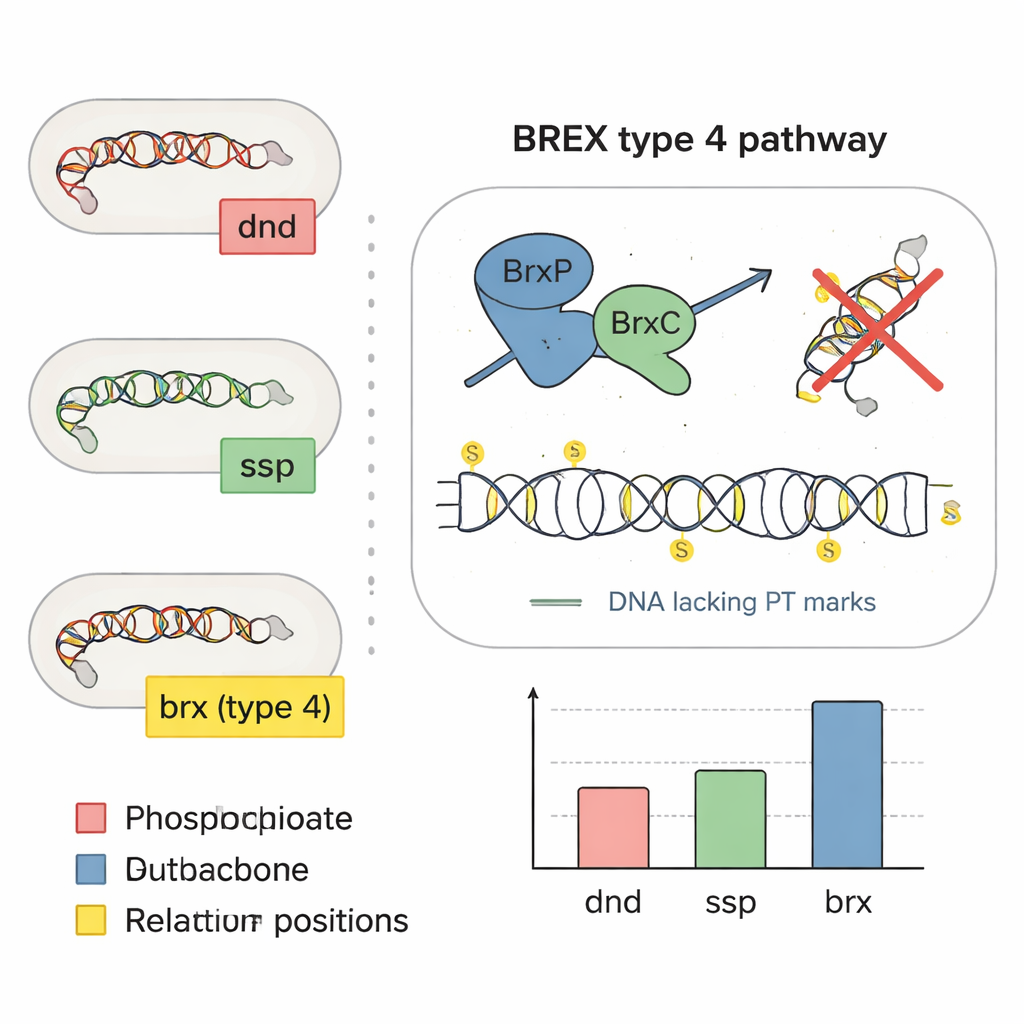
Most people have heard of DNA letters—A, T, C, and G—being modified by tiny chemical tags such as methyl groups, a key part of epigenetics. Here the focus is on something far more radical: some bacteria replace one of the oxygen atoms in the DNA backbone itself with sulfur. This backbone editing, known as phosphorothioation, changes how DNA behaves without altering the genetic code. Earlier studies had shown two main families of genes, called dnd and ssp, that install these sulfur marks in about one in ten bacterial and archaeal species. These marks help bacteria recognize their own DNA, fend off invading viruses called phages, and potentially respond to oxidative stress and inflammation.
Searching Thousands of Gut Genomes
To see how common sulfur-marked DNA is in our intestines, the researchers scanned 13,663 bacterial genomes drawn from three large collections of human gut microbes. They hunted for key genetic signatures that are sufficient to build sulfur marks: sets of dnd, ssp, or a newer candidate set of genes called brx that are part of BREX, a known anti-phage system. About 6.3% of gut genomes carried at least one of these systems, mainly in the common gut groups Bacteroidota, Bacillota, and Pseudomonadota. Compared with a broader catalog of bacteria from many environments, the gut showed a clear enrichment of the BREX type 4 version, suggesting that this sulfur-based defense is especially favored in the intestinal ecosystem.
A New Sulfur-Based Defense System
By examining how genes sit next to each other on bacterial chromosomes, the team noticed dnd-like genes nestled among BREX defense genes, hinting that some BREX systems might directly install sulfur marks. They zeroed in on a set of four core genes, brxP, brxC, brxZ, and brxL, and used sequence analysis to show that BrxP and BrxC resemble the sulfur-handling proteins from ssp systems. Experiments in the human gut bacterium Bacteroides salyersiae confirmed the suspicion: when the researchers deleted the brxC gene, sulfur-modified DNA vanished; when they restored brxC on a plasmid, the sulfur marks came back. They also moved key BREX genes into another gut bacterium that normally lacks these systems and showed it began making the same sulfur-patterned DNA, demonstrating that the BREX type 4 machinery can by itself create phosphorothioate marks.
Mapping Where Sulfur Lands on the Genome
Finding sulfur marks is only half the story; knowing exactly where they appear along the DNA is crucial for understanding their function. The researchers combined sensitive mass spectrometry with a custom sequencing method called PT-seq, which selectively cuts DNA at sulfur-modified sites and then reads out the surrounding sequence. Across 226 gut bacterial isolates, they identified eight distinct sulfur-containing dinucleotide “building blocks,” and from representative strains worked out full short sequence motifs where sulfur tends to land. Intriguingly, bacteria with dnd, ssp, or BREX systems produced different sets of sulfur patterns, like different dialects of the same chemical language. The marks were not scattered randomly: they were enriched in ribosomal RNA genes and generally avoided the beginnings and ends of protein-coding genes, suggesting that bacteria may be steering sulfur away from sensitive control regions.
What This Means for Our Health
To a non-specialist, these findings show that gut bacteria do more than just live off our food—they actively rewrite the chemistry of their own DNA in ways that shape how they battle viruses and respond to the harsh, oxidizing conditions that often accompany inflammation. By identifying BREX type 4 as a new sulfur-marking system and showing that about one in thirteen gut microbes carries some form of phosphorothioate machinery, this work lays the groundwork for exploring how these unusual DNA marks influence microbiome stability, resistance to infection, and possibly the course of diseases like inflammatory bowel disease. In the long run, understanding and perhaps manipulating these sulfur-based epigenetic systems could offer new strategies for tuning the gut microbiome to support human health.
Citation: Yuan, Y., DeMott, M.S., Byrne, S.R. et al. Phosphorothioate DNA modification by BREX type 4 systems in the human gut microbiome. Nat Commun 17, 1717 (2026). https://doi.org/10.1038/s41467-026-68412-5
Keywords: gut microbiome, bacterial epigenetics, DNA phosphorothioation, BREX defense system, bacteriophages