Clear Sky Science · en
In situ visualization of Clostridioides difficile phenotypic heterogeneity and single-cell morphology during gut infection
Why Gut Germs Don’t All Behave the Same
Clostridioides difficile, often called C. diff, is a notorious hospital germ that can cause severe, sometimes life‑threatening diarrhea after antibiotics disturb our gut’s normal microbes. Yet even when these bacteria are genetically identical and share the same environment, individual cells can behave very differently. This study reveals, in unprecedented detail, how single C. diff cells turn toxin production on or off and even change shape while infecting the mouse gut, offering clues to why the disease can be so persistent and difficult to treat.
Tracking a Dangerous Gut Invader in Real Time
To understand how individual C. diff cells act inside the body, the researchers needed a way to see them clearly amidst the dense and diverse community of gut microbes. They engineered C. diff strains that continuously glow in bright colors under the microscope, using special fluorescent proteins that do not interfere with the bacteria’s ability to grow or cause disease. By infecting antibiotic‑treated mice with these glowing strains and then slicing and staining the colon, they could pinpoint the exact locations and behaviors of thousands of single bacterial cells within intact gut tissue. 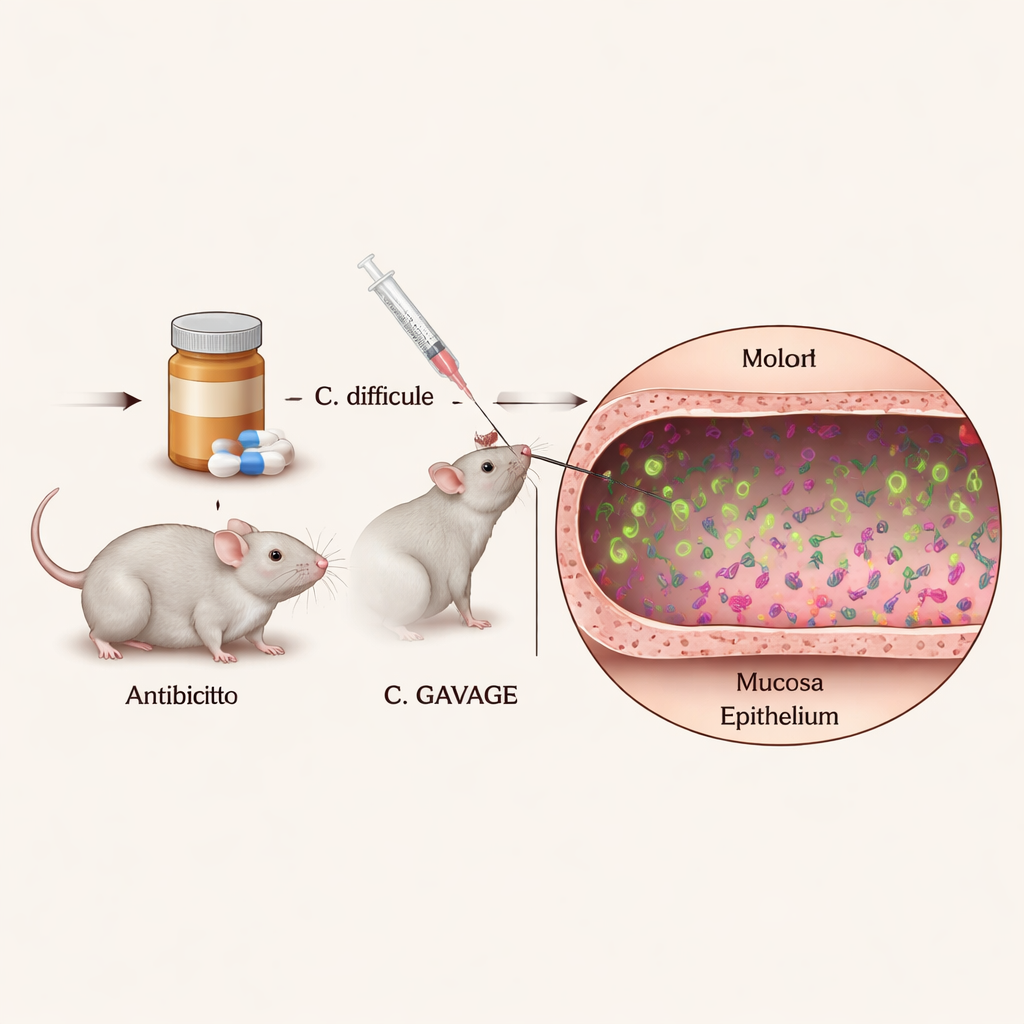
Where the Bacteria Live in the Colon
The images showed that most C. diff cells live in the center of the colon’s contents, known as the lumen, confirming that the organism is mainly a free‑floating “luminal” pathogen. However, a notable minority of cells consistently appeared near the mucus layer and directly next to the epithelial surface—the thin cell layer that lines the colon and acts as a barrier to the outside world. This close‑contact subpopulation had not been clearly seen before in conventional mice with normal immune systems. Importantly, the engineered fluorescent tags did not noticeably weaken the bacteria in animals, which means the pictures likely reflect how C. diff behaves in a realistic infection.
Who Makes Toxin, and Where?
The team then layered on a second fluorescent signal that turns on only when C. diff activates its toxin genes. Toxins are the damaging proteins that injure the gut lining and drive disease symptoms, and they are also what doctors measure in stool tests to diagnose infection. Surprisingly, the bacteria did not all produce toxins at once. Instead, only a fraction of cells lit up as “toxin‑ON” at any given time, both early and late in infection. This fraction was higher in a mutant strain that is genetically wired to overproduce toxin, but even there not every cell joined in. Just as striking, a cell’s position—floating in the lumen, sitting in mucus, or touching the epithelium—did not strongly change either how often toxin was made or how strongly those genes were turned on. 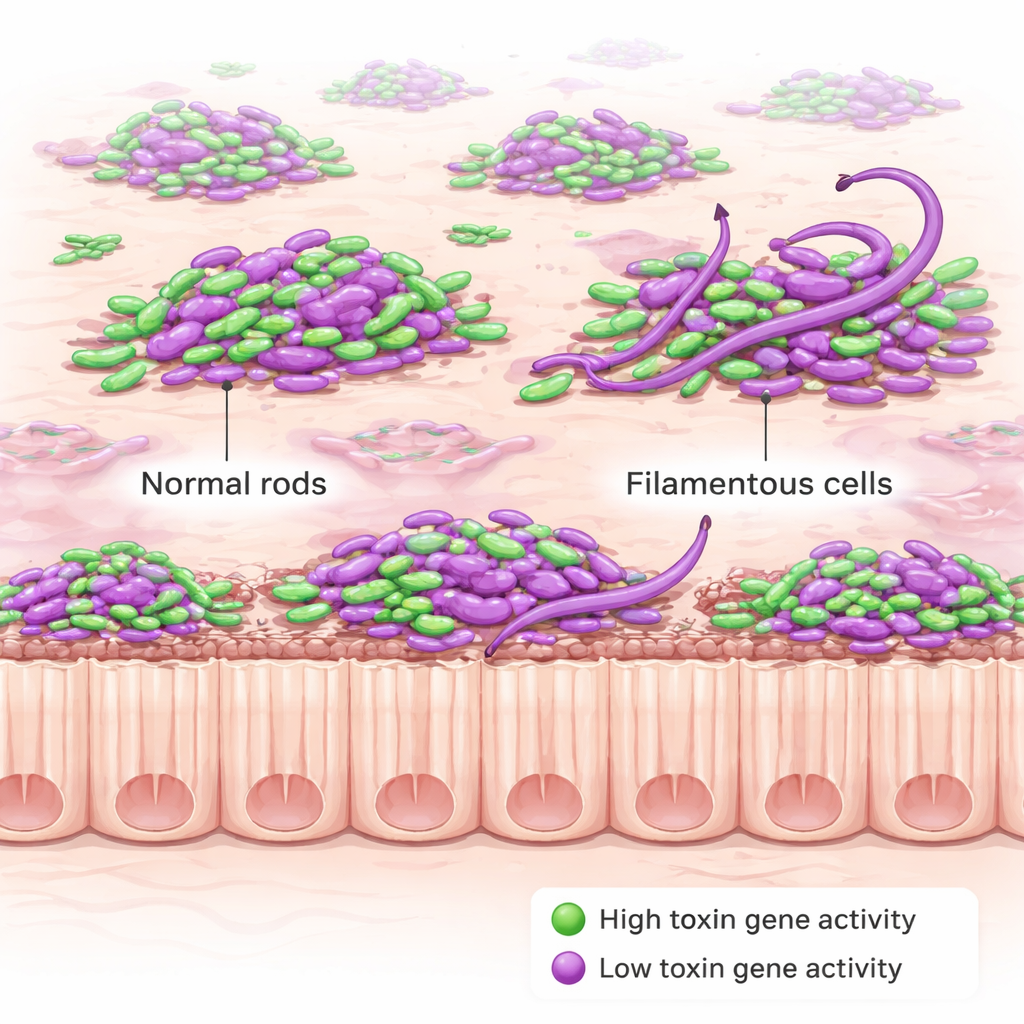
Shape‑Shifting Cells Under Stress
While mapping toxin production, the researchers stumbled upon a surprising new behavior. In mice infected with the toxin‑overproducing mutant, many C. diff cells during the acute phase of disease became unusually long, curved filaments, some more than ten times the length of normal short rods. This “string‑like” morphology appeared largely during early, severe infection and faded later, and it did not show up when the same strain was grown in laboratory broth. That suggests the stressful conditions inside the inflamed gut—rather than just the genetic changes alone—trigger this dramatic reshaping. Follow‑up experiments showed that losing a regulatory protein called RstA, which controls both toxin genes and other stress responses, helps drive this filament formation.
What This Means for C. diff Disease
For non‑specialists, the key message is that even a single strain of C. diff behaves like a divided workforce. Some cells pay the energy cost to make toxin and damage the gut, releasing nutrients that their toxin‑free neighbors can exploit. This shared “division of labor” may help the infection persist and rebound, because not every cell is equally vulnerable to treatments that target toxin or rapidly growing bacteria. The newly developed glowing reporter system gives scientists a powerful way to watch, cell by cell, how C. diff and related gut microbes adapt over time and space inside the body. That insight could ultimately guide therapies aimed not just at killing bacteria, but at disrupting the harmful roles of particular subpopulations that drive disease and recurrence.
Citation: DiBenedetto, N.V., Donnelly-Morell, M.L., Kumamoto, C.A. et al. In situ visualization of Clostridioides difficile phenotypic heterogeneity and single-cell morphology during gut infection. Nat Commun 17, 1716 (2026). https://doi.org/10.1038/s41467-026-68411-6
Keywords: Clostridioides difficile, gut microbiome, bacterial toxins, phenotypic heterogeneity, fluorescent imaging